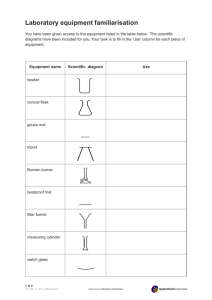
. KNO Lab Materials:
advertisement

. KNO3 Lab Materials: hot plate 600 ml beaker 10 ml grad cylinder squirt bottle of tap water glass stir rods thermometers or labpro setup beaker tongs 1. 2. Each Lab Table will have 2 hot plates, upon entering the class, go to lab tables and fill each 600 ml beaker ½ full, plug in hot plate, set beaker with water onto the hot plate and turn on. 3. Obtain from the metal cart near hood one test tube 2,4,6 or 8 g. I will tell you which one to pick. Return to your table and using the graduated cylinders in the baskets on the table and the squirt bottles of water, add exactly 5.0 ml of water to the test tube of your choice. Using the glass stir rod, stir while heating until dissolved. 4. Then, remove beaker from hot plate with beaker tongs (only 2 in classroom – you will have to share), return tongs to lab tables at the front of the room for others to use. Remove stir rod from test tube 5. Unplug hot plates. 6. Obtain thermometer (you will use the Labpros and temperature sensor in your top drawer on each side of the lab table), place thermometer into the water bath containing the test tube with your salt. 7. Do not disturb your test tube. Wait and watch until the first crystal starts to form. This is your temperature at which your amount of KNO3 is saturated. 8. Pooling class data, construct a solubility curve. Converting 2,4,6,8 g per 5.0 ml to the amount per 100 g of water. 9. Clean up by rinsing test tubes down the sink with plenty of water and return equipment originally on the cart back to the cart. The other supplies stay at the lab tables, rinsed well with tap water. This will be later next week Mar. 18 2014, don not copy this part Solution Dilution While waiting for your KNO3 to dissolve and cool to the saturation temperature, dilute the solutions of Nickel II sulfate and Copper II sulfate into appropriate volumetric flasks. Show me and you are done. At least one dilution per 2 people.