Solubility of a salt
advertisement

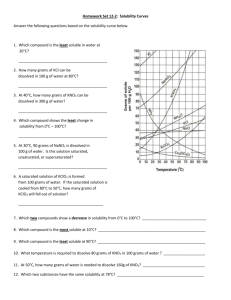
Title: Solubility of a salt Objective: How does temperature affect solubility? Procedure: Test tube 1 2 3 4 Grams of KNO3/5 mL H 2O 2g/5ml 4g/5ml 6g/5ml 8g/5ml Conversion based on a 100 ml Crystallization temperature (OC) Using the conversion scale, graph the affects of solubility versus temperature. 1. According to your graph, how many grams of KNO3 can be dissolved in 100mL of H2O at the following temperature? a. 30 OC b. 40OC c. 60OC d. 70OC 2. Using the reference tables, find the actual amount and compare the results to question one. 3. Define the terms saturated, unsaturated, supersaturated, and equilibrium. 4. Using the reference table, classify the following KNO3 solutions as saturated, unsaturated, and supersaturated. a. 75 g KNO3/100 ml H2O at 40OC b. 60 g KNO3/100 ml H2O at 50OC c. 60 g KNO3/200 ml H2O at 30OC 5. How does the solubility of a gas change with increasing temperature? Draw a rough sketch showing the general shape of the solubility curve of a gas. 6. Based of the reference tables, which of the compounds represented become less soluble as temperature increases?