Thermodynamics and specific heat
advertisement

THERMOCHEMISTRY
Thermodynamics
The study of Heat and Work
and State Functions
1
Energy & Chemistry
ENERGY is the capacity to
do work or transfer heat.
HEAT is the form of energy
that flows between 2
objects because of their
difference in temperature.
Other forms of energy —
• light
• electrical
• kinetic and potential
2
Energy & Chemistry
• Burning peanuts
supply sufficient
energy to boil a cup
of water.
• Burning sugar
(sugar reacts with
KClO3, a strong
oxidizing agent)
3
Energy & Chemistry
• These reactions are PRODUCT
FAVORED
• They proceed almost completely
from reactants to products, perhaps
with some outside assistance.
4
Energy & Chemistry
2 H2(g) + O2(g) -->
2 H2O(g) + heat and light
This can be set up to
provide ELECTRIC
ENERGY in a fuel cell.
Oxidation:
2 H2 ---> 4 H+ + 4 e-
Reduction:
4 e- + O2 + 2 H2O ---> 4 OH-
CCR, page 845
5
Potential & Kinetic Energy
Potential energy
— energy a
motionless
body has by
virtue of its
position.
6
Potential Energy
on the Atomic Scale
• Positive and
negative particles
(ions) attract one
another.
• Two atoms can
bond
• As the particles
attract they have a
lower potential
energy
NaCl — composed of
Na+ and Cl- ions.
7
Potential Energy
on the Atomic Scale
• Positive and
negative particles
(ions) attract one
another.
• Two atoms can
bond
• As the particles
attract they have a
lower potential
energy
8
Potential & Kinetic Energy
Kinetic energy
— energy of
motion
• Translation
9
Potential & Kinetic Energy
Kinetic energy —
energy of
motion.
rotate
vibrate
translate
10
Internal Energy (E)
• PE + KE = Internal energy (E or U)
• Int. E of a chemical system
depends on
• number of particles
• type of particles
• temperature
11
12
Internal Energy (E)
• PE + KE = Internal energy (E or U)
QuickTime™ and a
Graphics decompressor
are needed to see this picture.
Internal Energy (E)
• The higher the T
the higher the
internal energy
• So, use changes
in T (∆T) to
monitor changes
in E (∆E).
13
Thermodynamics
• Thermodynamics is the science of heat
(energy) transfer.
Heat energy is associated
with molecular motions.
Heat transfers until thermal equilibrium is
established.
14
Directionality of Heat Transfer
15
• Heat always transfer from hotter object to
cooler one.
• EXOthermic: heat transfers from SYSTEM to
SURROUNDINGS.
T(system) goes down
T(surr) goes up
Directionality of Heat Transfer
• Heat always transfer from hotter object to
cooler one.
• ENDOthermic: heat transfers from
SURROUNDINGS to the SYSTEM.
T(system) goes up
T (surr) goes down
16
Energy & Chemistry
All of thermodynamics depends
on the law of
CONSERVATION OF ENERGY.
• The total energy is unchanged
in a chemical reaction.
• If PE of products is less than
reactants, the difference must
be released as KE.
17
Energy Change in
Chemical Processes
PE
Reactants
Kinetic
Energy
Products
PE of system dropped. KE increased. Therefore,
you often feel a T increase.
18
UNITS OF ENERGY
1 calorie = heat required to
raise temp. of 1.00 g of
H2O by 1.0 oC.
1000 cal = 1 kilocalorie = 1
kcal
1 kcal = 1 Calorie (a food
“calorie”)
But we use the unit called
the JOULE
1 cal = 4.184 joules
James Joule
1818-1889
19
20
HEAT CAPACITY
The heat required to raise an
object’s T by 1 ˚C.
Which has the larger heat capacity?
Specific Heat Capacity
How much energy is transferred
due to T difference?
The heat (q) “lost” or “gained” is
related to
a)
b)
sample mass
change in T and
c)
specific heat capacity
Specific heat capacity =
heat lost or gained by substance (J)
(mass, g)(T change, K)
21
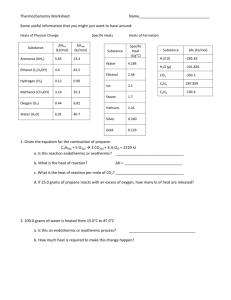
Specific Heat Capacity
Substance
Spec. Heat (J/g•K)
H2O
4.184
Ethylene glycol
2.39
Al
0.897
glass
0.84
Aluminum
22
23
Specific Heat Capacity
If 25.0 g of Al cool from
310 oC to 37 oC, how
many joules of heat
energy are lost by
the Al?
Specific heat capacity =
heat lost or gained by substance (J)
(mass, g)(T change, K)
Specific Heat Capacity
If 25.0 g of Al cool from 310 oC to 37 oC, how many
joules of heat energy are lost by the Al?
heat gain/lose = q = (sp. ht.)(mass)(∆T)
where ∆T = Tfinal - Tinitial
q = (0.897 J/g•K)(25.0 g)(37 - 310)K
q = - 6120 J
Notice that the negative sign on q signals
heat “lost by” or transferred OUT of Al.
24
Heat Transfer
No Change in State
q transferred = (sp. ht.)(mass)(∆T)
25
Heat Transfer with
Change of State
Changes of state involve energy (at constant T)
Ice + 333 J/g (heat of fusion) -----> Liquid water
q = (heat of fusion)(mass)
26
27
Heat Transfer and
Changes of State
Liquid ---> Vapor
Requires energy (heat).
This is the reason
a) you cool down after
swimming
b) you use water to put
out a fire.
+ energy
Heating/Cooling Curve for Water
Evaporate water
Heat water
Melt ice
Note that T is
constant as ice melts
28
Heat & Changes of State
What quantity of heat is required to melt
500. g of ice and heat the water to
steam at 100 oC?
Heat of fusion of ice = 333 J/g
Specific heat of water = 4.2 J/g•K
Heat of vaporization = 2260 J/g
+333 J/g
+2260 J/g
29
Heat & Changes of State
How much heat is required to melt 500. g of ice
and heat the water to steam at 100 oC?
1.
To melt ice
q = (500. g)(333 J/g) = 1.67 x 105 J
2.
To raise water from 0 oC to 100 oC
q = (500. g)(4.2 J/g•K)(100 - 0)K = 2.1 x 105 J
3.
To evaporate water at 100 oC
q = (500. g)(2260 J/g) = 1.13 x 106 J
4. Total heat energy = 1.51 x 106 J = 1510 kJ
30
Chemical Reactivity
What drives chemical reactions? How do they
occur?
The first is answered by THERMODYNAMICS and
the second by KINETICS.
Have already seen a number of “driving forces”
for reactions that are PRODUCT-FAVORED.
• formation of a precipitate
• gas formation
• H2O formation (acid-base reaction)
• electron transfer in a battery
31
Chemical Reactivity
But energy transfer also allows us to predict
reactivity.
In general, reactions that transfer energy
to their surroundings are productfavored.
So, let us consider heat transfer in chemical processes.
32
Heat Energy Transfer in
a Physical Process
CO2 (s, -78 oC) ---> CO2 (g, -78 oC)
Heat transfers from surroundings to system in endothermic process.
33
Heat Energy Transfer in
a Physical Process
• CO2 (s, -78 oC) --->
CO2 (g, -78 oC)
• A regular array of
molecules in a solid
-----> gas phase
molecules.
• Gas molecules have
higher kinetic
energy.
34
Energy Level Diagram
for Heat Energy
Transfer
CO2 gas
∆E = E(final) - E(initial)
= E(gas) - E(solid)
CO2 solid
35
Heat Energy Transfer in
Physical Change
CO2 (s, -78 oC) ---> CO2 (g, -78 oC)
Two things have happened!
• Gas molecules have higher
kinetic energy.
• Also, WORK is done by the
system in pushing aside the
atmosphere.
36
FIRST LAW OF
THERMODYNAMICS
heat energy transferred
∆E = q + w
energy
change
work done
by the
system
Energy is conserved!
37
38
heat transfer in
(endothermic), +q
heat transfer out
(exothermic), -q
SYSTEM
∆E = q + w
w transfer in
(+w)
w transfer out
(-w)
ENTHALPY
Most chemical reactions occur at constant P, so
Heat transferred at constant P = qp
qp =
∆H
where H
= enthalpy
and so ∆E = ∆H + w (and w is usually small)
∆H = heat transferred at constant P ≈ ∆E
∆H = change in heat content of the system
∆H = Hfinal - Hinitial
39
ENTHALPY
∆H = Hfinal - Hinitial
If Hfinal > Hinitial then ∆H is positive
Process is ENDOTHERMIC
If Hfinal < Hinitial then ∆H is negative
Process is EXOTHERMIC
40
USING ENTHALPY
Consider the formation of water
H2(g) + 1/2 O2(g) --> H2O(g) + 241.8 kJ
Exothermic reaction — heat is a “product”
and ∆H = – 241.8 kJ
41
USING ENTHALPY
42
Making liquid H2O from H2 +
O2 involves two exothermic
steps.
H2 + O2 gas
H2O vapor
Liquid H2O
USING ENTHALPY
Making H2O from H2 involves two steps.
H2(g) + 1/2 O2(g) ---> H2O(g) + 242 kJ
H2O(g) ---> H2O(liq) + 44 kJ
-----------------------------------------------------------------------
H2(g) + 1/2 O2(g) --> H2O(liq) + 286 kJ
Example of HESS’S LAW—
If a rxn. is the sum of 2 or more others,
the net ∆H is the sum of the ∆H’s of
the other rxns.
43
Hess’s Law
& Energy Level Diagrams
Forming H2O can occur in a
single step or in a two
steps.
∆Htotal is the same no matter
which path is followed.
44
Hess’s Law
& Energy Level Diagrams
45
Forming CO2 can occur in a
single step or in a two steps.
∆Htotal is the same no matter
which path is followed.
46
∆H along one path =
∆H along another path
• This equation is valid because
∆H is a STATE FUNCTION
• These depend only on the state
of the system and not on how
the system got there.
• V, T, P, energy — and your bank
account!
• Unlike V, T, and P, one cannot
measure absolute H. Can only
measure ∆H.
Standard Enthalpy Values
Most ∆H values are labeled ∆Ho
Measured under standard conditions
P = 1 bar = 105 Pa = 1 atm /1.01325
Concentration = 1 mol/L
T = usually 25 oC
with all species in standard states
e.g., C = graphite and O2 = gas
47
Enthalpy Values
Depend on how the reaction is written and on phases
of reactants and products
H2(g) + 1/2 O2(g) --> H2O(g)
∆H˚ = -242 kJ
2 H2(g) + O2(g) --> 2 H2O(g)
∆H˚ = -484 kJ
H2O(g) ---> H2(g) + 1/2 O2(g)
∆H˚ = +242 kJ
H2(g) + 1/2 O2(g) --> H2O(liquid)
∆H˚ = -286 kJ
48
Standard Enthalpy Values
NIST (Nat’l Institute for Standards and
Technology) gives values of
∆Hfo = standard molar enthalpy of
formation
— the enthalpy change when 1 mol of
compound is formed from elements under
standard conditions.
See Table 6.2
49
o,
∆Hf standard molar
enthalpy of formation
Enthalpy change when 1 mol of
compound is formed from the
corresponding elements under
standard conditions
H2(g) + 1/2 O2(g) --> H2O(g)
∆Hfo (H2O, g)= -241.8 kJ/mol
By definition,
∆Hfo = 0 for elements in their
standard states.
50
Using Standard Enthalpy Values
Use ∆H˚’s to calculate enthalpy change for
H2O(g) + C(graphite) --> H2(g) + CO(g)
(product is called “water gas”)
51
Using Standard Enthalpy Values
52
H2O(g) + C(graphite) --> H2(g) + CO(g)
From reference books we find
• H2(g) + 1/2 O2(g) --> H2O(g) ∆Hf˚ = - 242 kJ/mol
• C(s) + 1/2 O2(g) --> CO(g)
∆Hf˚ = - 111 kJ/mol
Using Standard Enthalpy Values
H2O(g) --> H2(g) + 1/2 O2(g) ∆Ho = +242 kJ
C(s) + 1/2 O2(g) --> CO(g) ∆Ho = -111 kJ
--------------------------------------------------------------------------------
H2O(g) + C(graphite) --> H2(g) + CO(g)
∆Honet = +131 kJ
To convert 1 mol of water to 1 mol each of H2
and CO requires 131 kJ of energy.
The “water gas” reaction is ENDOthermic.
53
54
Using Standard Enthalpy Values
Calculate ∆H of
reaction?
In general, when ALL
enthalpies of formation are
known:
∆Horxn = ∆Hfo (products) - ∆Hfo (reactants)
Remember that ∆ always = final – initial
55
Using Standard Enthalpy Values
Calculate the heat of combustion of
methanol, i.e., ∆Horxn for
CH3OH(g) + 3/2 O2(g) --> CO2(g) + 2 H2O(g)
∆Horxn = ∆Hfo (prod) - ∆Hfo (react)
Using Standard Enthalpy Values
CH3OH(g) + 3/2 O2(g) --> CO2(g) + 2 H2O(g)
∆Horxn = ∆Hfo (prod) - ∆Hfo (react)
∆Horxn = ∆Hfo (CO2) + 2 ∆Hfo (H2O)
- {3/2 ∆Hfo (O2) + ∆Hfo (CH3OH)}
= (-393.5 kJ) + 2 (-241.8 kJ)
- {0 + (-201.5 kJ)}
∆Horxn = -675.6 kJ per mol of methanol
56
CALORIMETRY
57
Measuring Heats of Reaction
Constant Volume
“Bomb” Calorimeter
• Burn combustible
sample.
• Measure heat evolved
in a reaction.
• Derive ∆E for
reaction.
Calorimetry
Some heat from reaction warms
water
qwater = (sp. ht.)(water mass)(∆T)
Some heat from reaction warms
“bomb”
qbomb = (heat capacity, J/K)(∆T)
Total heat evolved = qtotal = qwater + qbomb
58
Measuring Heats of Reaction
CALORIMETRY
Calculate heat of combustion of octane.
C8H18 + 25/2 O2 --> 8 CO2 + 9 H2O
• Burn 1.00 g of octane
• Temp rises from 25.00 to 33.20 oC
• Calorimeter contains 1200 g water
• Heat capacity of bomb = 837 J/K
59
Measuring Heats of Reaction
CALORIMETRY
Step 1
Calc. heat transferred from reaction to water.
q = (4.184 J/g•K)(1200 g)(8.20 K) = 41,170 J
Step 2
Calc. heat transferred from reaction to bomb.
q = (bomb heat capacity)(∆T)
= (837 J/K)(8.20 K) = 6860 J
Step 3
Total heat evolved
41,170 J + 6860 J = 48,030 J
Heat of combustion of 1.00 g of octane =
- 48.0 kJ
60