Chemical reactions review
advertisement

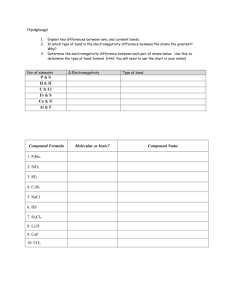
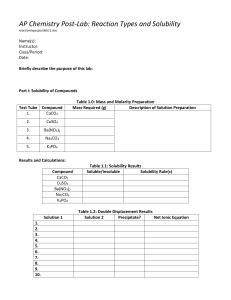
Name _______________________________________________ period _____ date ___________________ Chemical reactions review I. Fill in the blanks with the most appropriate term: A _____________________ ________________ tells the story of a chemical reaction. ________________ are the starting substances in the reaction while ________________ are the new substances that are formed. The large numbers in front of some of the formulas are called ________________. These numbers are used to ________________ the equation because chemical reactions must obey the Law of __________________ of Matter. The number of atoms of each element on both sides of the equation must be ________________ because matter cannot be ________________ or ________________. When balancing equations, the only numbers that can be changed are _____________________; remember that __________________________ must never be changed in order to balance an equation. II. Balance and classify the following equations: 1. ____Al + _____O2 → _____ Al2O3 2. ____C3H8 + _____ O2 → ____ CO2 3. ____KNO3 → ____ KNO2 + ____ O2 4. ____KOH + ____H3PO4 → ____ K3PO4 5. ____BaF2 + ____K3PO4 → ____ Ba3(PO4)2 + + ____ H2O + ____ H2O ____ KF III. Predict the products in each of the following synthesis, decomposition or combustion reactions. 1. Ba + N2 → 2. H2CO3 → 3. C3H8 + O2 → 4. LiCl + O2 → 5. CuF → IV. Predict the products in each of the following single replacement reactions. If no reaction occurs, write NR. 1. Pt + MgBr2 → 2. Sn + CaS → 3. NaI + F2 → 4. Zn + AgNO3 → 5. Fe2SO4 + Cu → 6. H2CO3 → + Li V. Predict the products in each of the following reactions. Using the solubility chart, determine if a precipitate forms (insoluble compound) and circle it. If no reaction occurs, write NR. 1. Ba(NO3)2 + H2SO4 → 2. FeCl2 + (NH4)3PO4 → 3. NaClO3 + K2S → 4. AlBr3 + MgI2 → 5. K2CO3 + SrO → V. Choose two of the reactions from the previous section (IV) and write the complete balanced equation, complete ionic equation and net ionic equation for each.