Zumdahl -1411 Chapter 10 Practice Problems.doc
advertisement

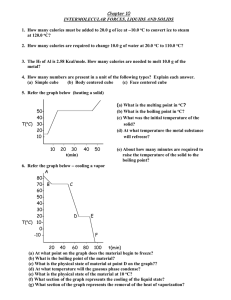
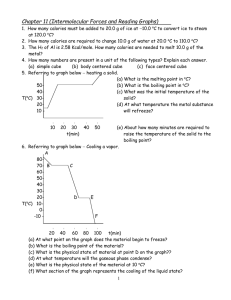
CHAPTER 10 LIQUIDS AND SOLIDS 1. How many calories must be added to 20.0 g of ice at 10.0 oC to convert ice to steam at 120.0 oC? 2. How many calories are required to change 10.0 g of water at 20.0 oC to 110.0 oC? 3. The Hf of Al is 2.58 Kcal/mole. How many calories are needed to melt 10.0 g of the metal? 4. How many numbers are present in a unit of the following types? Explain each answer. (a) Simple cube (b) Body centered cube (c) Face centered cube 5. Refer the graph below (heating a solid) (a) What is the melting point in oC? (b) What is the boiling point in oC? (c) What was the initial temperature of the solid? (d) At what temperature the metal substance refreeze? 5040o T( C) 302010 l 10 l l 20 30 t(min) l l 40 50 (e) About how many minutes are required to raise the temperature of the solid to the boiling point? 6. Refer the graph below cooling a vapor A 8070- B C 6050403020D E o T( C) 100-10 F l l l l l 20 40 60 80 100 t(min) (a) At what point on the graph does the material begin to freeze? (b) What is the boiling point of the material? (c) What is the physical state of material at point D on the graph?? (d) At what temperature will the gaseous phase condense? (e) What is the physical state of the material at 10 oC? (f) What section of the graph represents the cooling of the liquid state? (g) What section of the graph represents the removal of the heat of vaporization? 7. Refer the graph below phase diagram of compound. A 2.52.0P(atm) 1.51.00.5- l 0 l 60 B C l 80 l l l 120 160 200 T(oC) (a) For this substance, in what physical state would it exist at 10 oC and 760 mm Hg? (b) What is the pressure and temperature at the triple point? (c) What are the critical temperature and pressure? (d) What is the vapor pressure of the liquid phase at 100 oC? (e) What is the maximum temperature to which the substance can be raised and still undergo sublimation? 8. Define the following terms: a) Triple point b) Critical temperature e) Melting point f) Boiling point i) Freezing point j) Hydrogen bonding c) Sublimation g) Condensation d) Evaporation h) Deposition 9. An element having atomic mass 52 occurs in body centered cubic lattice. The density of the element is 7.2 g/cc. Calculate the edge length of the unit cell. 10. An element having atomic mass 60 has face centered cubic unit cells. The edge length of the unit cell is 400 pm. Find out density of the element.