Periodic table
advertisement

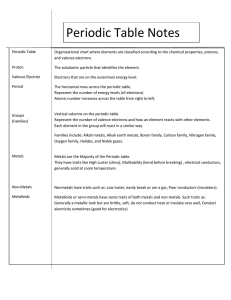
Periodic table The periodic table can be understood in terms of the possible electron states, as predicted by the solution of the Schrödinger equation, and the Pauli principle. for a given kind of atom (i.e. given total number of electrons), the ground state of the atom is that in which electrons occupy the states with the lowest possible energy. the electron configuration in the outer subshells of an atom determines its chemical properties. the beginning of every “period” of the periodic table corresponds to the beginning of a new shell the end of every period corresponds to the “noble gases”, in which the outer electrons are in a particularly stable configuration, called “noble gas configuration”or “octet structure”. the noble gas configuration is ns2np6 , i.e. 2 electrons in the s subshell, and 6 electrons in the p subshell of the shell n (except for n = 1, where there is no l = 1 subshell). plot of ionization energy vs atomic number Z shows minima at the Z values corresponding to the beginning of periods (hydrogen, and the “alkali metals” Li, Na, K, Rb, Cs), and maxima at the end of the periods (the noble gases He, Ne, Ar, Kr, Xe, Rn). (ionization energy = energy necessary to liberate loosest bound electron of the atom). Periodic table and electron configuration 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 1 H He 1 2 2 Li Be B C N O F Ne 2,1 2,3 2,4 2,5 2,6 2,7 Mg Al Si P S Cl Ar 2,8,2 2,8,3 2,8,4 2,8,5 2,8,6 2,8,7 2,8,8 Na 3 2,8,1 2,2 K Ca Sc Ti 42,8,8, 1 Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 2,8,8, 2,8,9 2,8,10, 2,8,11, 2,8,13, 2,8, 2,8,1 2,8,15 2,8,1, 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,1, 2 2,8,1, 1 2,8,1, 2 2 ,2 2 2 1 13,2 4,2 2 3 4 5 6 7 8 Rb Sr Y 5 V 2,8 Zr Nb Mo Tc Ru Rh Pd 2,8,1 2,8, 2,8, 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 8 18 18 8,1 8,2 10,2 12,1 13,1 16,1 18,0 9,2 14,1 15,1 Cs Ba * Hf Ta W Re Os Ir Pt Ag Cd In Sn Sb Te I Xe 2,8,18 18,1 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 18,2 18,3 18,4 18,5 18,6 18,7 18,8 Au Hg Tl Pb Bi Po At Rn 2,8,1 2,8,1 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 8 8 32,11, 32,12, 32,15, 32,18, 32,18, 32,18, 32,18, 32,18, 32,18, 32,10,2 32,13 32,1 32,17,1 32,18,1 32,18,2 2 2 2 3 4 5 6 7 8 ,2 4,2 6 2,8,18 2,8,18 18,8,1 18,8,2 Fr Ra ** Rf Db Sg Bh Hs Mt Uun Uuu Uub 2,8,18, 2,8,18, 2,8,1 2,8,1 2,8,18 2,8,18, 2,8,183 2,8,183 32 32 8,32 8,32 32 2,8,1832 32 2 2 32,11, 32,12, 32,13 32,1 32,15, 32,18,1 32,10,2 32,17,1 32,18,2 2 2 2 4,2 2 2,8,18 72,8,18 32 32 18,8,1 18,8,2 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 2,8,1 2,8,1 2,8,18 2,8,18 2,8,18 8 8 2,8,18 2,8,18 18,9,2 20,8,2 21,8,2 22,8, 23,8, 24,8,2 25,8,2 2 2 2,8,18 25,9,2 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 27,8,2 28,8,2 29,8,2 30,8,2 31,8,2 32,8,2 32,9,2 Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr * ** 2,8,18, 2,8,1 2,8,1 2,8,18, 2,8,18, 2,8,18 2,8,18,3 2,8,18, 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 2,8,18 32 8,32 8,32 2,8,18,32 32 32 ,32 2 32 ,32 ,32 ,32 ,32 ,32 ,32 18,10, 21,9, 23,8, 25,9,2 18,9,2 20,9,2 24,8,2 25,8,2 26,9,2 28,8,2 29,8,2 30,8,2 31,8,2 32,8,2 32,9,2 2 2 2 Element Groups (Families) Alkali Earth Rare Earth Non-Metals Alkaline Earth Other Metals Halogens Transition Metals Metalloids Noble Gases Quantum numbers in periodic table • The period indicates the value of principal quantum number for the valence shell; • the lanthanides and actinides are in periods 6 and 7, respectively. • The block indicates value of azimuthal (orbital) quantum number l for the last subshell that received electrons in building up the electron configuration. • blocks are named for subshells (s, p, d, f) • Each block contains a number of columns equal to the number of electrons that can occupy that subshell • The s-block (in orange) has 2 columns, because a maximum of 2 electrons can occupy the single orbital in an s-subshell. • The p-block (in violet) has 6 columns, because a maximum of 6 electrons can occupy the three orbitals in a p-subshell. • The d-block (in green) has 10 columns, because a maximum of 10 electrons can occupy the five orbitals in a d-subshell. • The f-block (in dark blue) has 14 columns, because a maximum of 14 electrons can occupy the seven orbitals in a f-subshell. Periodic table and phases of elements l http://www.intercorr.com/periodic/ Periodic table Ionization energy Atomic radius Electron affinity Periodic table l http://library.thinkquest.org/3659/pertable/ “representative” metals: alkali metals alkaline earth metals, post-transition metals metals l l Transition metals inner transition metals (rare earth metals) (Lanthanide, Actinide series) Metalloids, non-metals l Metalloids l Non-metals H halogens noble gases