1 Unit 5 Gases and Atmospheric Chemistry Day 1
advertisement

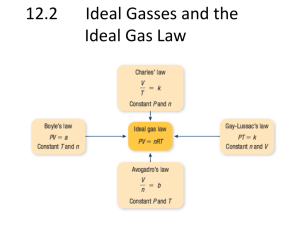
1 Unit 5 Gases and Atmospheric Chemistry Day 1 1. Complete table below :( Use checkmarks )(pg 418) Property Solid Liquid Gas definite volume Definite shape Indefinite volume Indefinite shape Ability to flow Vibrational motion Rotational motion Translational motion Compressible Degree of disorder ( low, medium, high) Characteristics of Gases The empirical properties of the three states of matter—solid, liquid, gas—provide important clues about the nature and structure of matter. At the same temperature some substances are solids, some are liquids, and some are gases. This suggests that the forces between the particles that make up the substance vary in strength. kinetic molecular theory: the idea that all substances contain particles that are in constant, random motion 3 types of motion : 1. translational (straight line) 2. rotational (spinning) 3. vibrational (back & forth motion of atoms in a molecule) solid (vibrational)• solids have definite shape and volume • are virtually incompressible • do not flow easily Liquid (vibrational, rotational and translational) • liquids assume the shape of the container but have a definite volume • are virtually incompressible • flow readily Gas( translational) • gases assume the shape and volume of the container • are highly compressible • flow readily •Gases form homogeneous mixtures. The differences in physical states of elements at SATP demonstrate the difference between intermolecular forces: dipole–dipole, London, and hydrogen bonding are not as strong as ionic, covalent, or metallic bonds. Atomic theory predicts that noble gases are composed of monatomic molecules e.g.,Ne (g). diatomic molecules of elements, for example, H2(g) and Cl2(g). Properties (continued) : Gases have no definite shape or volume Most gases are colourless and odourless Gases have ___________ density e.g. NH3 = 0.771 g/L H2 = 0.0899 g/L O2 = 1.429 g/L Gases can be mixed together to form ___________________ Gases have the ability to diffuse Gases are able to exert pressure Collisions of Gas Particles 2. Summarize the 5 major assumptions of the kinetic molecular theory of gases.(pg 421) __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ 2 3 Kinetic Theory 3. 4. 5. For a gas to be considered an ideal gas ; what 2 major assumptions must we make ?(422) __________________________________________________________________ __________________________________________________________________ Answer questions page 423 ( 1-7 ) Define pressure. What is the SI unit for pressure ?Define atmospheric pressure and state quantitative value in mm mercury , kilopascals (kPa ), atmospheres, and torrs. Convert from one unit to another. Understand how a manometer works ( see page 429 ) Do question # 1 on page 435 Measurement of gas pressure : Pressure - - defined as _____per unit area ; a pressure of 1.0 N/m2 = 1.0 _________ to illustrate : consider a long glass tube filled with mercury which is inverted into a container of mercury; level in the tube decreases to height that can be just supported by the atmosphere; at sea level the height is 760 mm Hg = 1.0 atmosphere of pressure = 101.3 kPa. See overhead average air pressure at sea level = _________kPa ---> atmospheric pressure 4 1. 2. 3. 4. 5. 6. 7. 1. The tube is filled with mercury. It is inverted into a dish of mercury. The mercury will fall down in the tube due to the force of __________________ _____________ pressure pushes the column of mercury up. Air pressure is high enough to keep the column of mercury at a height of approx ________ mm. The space above the mercury is a ____________ since no air was allowed to enter the tube. If water were used instead of mercury, then air pressure would be able to hold up a column of approx 10 m. Manometer ( see pg. 429) See overhead Open End - air exerts pressure on the column of liquid ( mercury ) on one am of the tube and the gas sample exerts pressure on the other arm ---> Patm. is < Pgas : Patm = Pgas - H Pgas = Patm + H = _________________ = _________________ Convert to kPa 101.3 kPa x --------------- = ---------760 mm Hg _______ mm Hg x = ____________ kpa -----> Pgas = _______________________ Do question # 1 on page 435 5 Gas Pressure Conversions To be able to convert between pressure units. Convert the pressure from the units in the left hand column to the units in the last column. In the middle column show all work with unit cancellations. Use the following equivalences to help. 101.3 kPa = 760.0 mm Hg = 760.0 torr = 1.00 atm =14.7 p.s.i. 100.0 kPa = 1.00 bar = 750.0 mm Hg = 14.5 p.s.i. Convert this Pressure e.g. 741 mm Hg 20.0 kPa 0.83 atm 1.2 bar 1250.0 mm Hg Showing Calculations to x kPa____ = 101.3 kPa 741 mm Hg 760mm Hg x kPa = 741 mm Hg x 101.3 kPa 760 mm Hg Final Units 98.8 kPa ______ mm Hg _______ ______ kPa kPa ________ atm 225 kPa ______ torr 982 mbar _______kPa 55 p.s.i. 0.89 atm ______ torr _______ kPa Day 2 Inqury Lab Gas Laws 6 In solids and liquids, the atoms or molecules are very close to each other leaving no room between them. For this reason, solids and liquids cannot be measurably compressed. Gases, on the other hand, have relatively large distances between the atoms or molecules as they bounce around into each other and the walls of their container. This fact allows gases to be compressed. In this experiment you will use modern equipment to recreate the experiment first done by Robert Boyle in 1661. Boyle discovered that there is a mathematical relationship between gas pressure and volume. Gas pressure is related to how often gas molecules bounce into surfaces. When the volume of a container of gas is changed, the distance between the walls changes and the amount of time it takes for a particle to get from one wall to another changes, which results in a different number of collisions per second and, results in, a different amount of pressure. The pressure of a constant volume of gas will be affected by changes in temperature. Since temperature is a measure of the average kinetic energy (and therefore the average speed) of gas molecules, a change in temperature will change how much time it takes for molecules to move wall to wall in a container. This implies that if the molecules stopped moving, they would no longer hit the walls and the pressure would be zero. The colder something gets, the slower the molecules move, thus the temperature at which all motion stops must be the coldest temperature possible. This temperature is called "absolute zero". In part two of this lab you will attempt to determine the relationship between pressure and temperature and also to determine the numerical value of absolute zero. Purpose In part one of this two part exploration, you will measure how the pressure of a confined gas changes with changes in the volume. The gas will be captured in a syringe which has a volume scale printed on it. The syringe will be attached to a pressure sensor to measure the pressure of the gas within the syringe.In part two, you will use the Chemistry Sensor to measure the pressure of the gas inside a rigid container and use a temperature probe to measure the temperature of the water bath in which the container is immersed. You will then use a graph of Absolute Pressure versus Temperature data to determine the relationship between pressure and temperature and to estimate the value of absolute zero. Materials PASCO & Other Equipment PASPORT Xplorer GLX Right angle Clamp PASPORT Chemistry Sensor Small Tripod Base & Rod Temperature probe 45 cm Steel Rod hot plate beaker, 1-L Aluminum Can tongs quick-release connector (with sensor) Three-finger Clamp 7 tubing (with sensor) Gas law syringe Rubber Stopper, (#8, 1-Hole) protective gear Consumables glycerin (C3H5(OH)3), 1 drop water, 1.0 L rubber band Safety Precautions • Remember, follow the directions for using the equipment. • Keep water away from electrical outlets, the GLX, and the PASPORT equipment. • Be very careful when using the hot plate. • Wear safety glasses and follow all standard laboratory safety procedures. Pre-Lab Question Part 1 1) In general, what change in pressure do you expect to see as the volume changes? Explain. ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ __________________________________________________________ 2) Why is it important to keep the temperature constant during this experiment? ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ Part 2 1) Use the kinetic molecular theory to explain how/why gas pressure changes as temperature changes. ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 2) Why can you use pressure vs. temperature to guess the value of absolute zero? ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 8 Part 1 Procedure Equipment Setup Put a drop of glycerin on the barb end of a quick-release connector. Put the end of the connector into one end of a short piece (about 2.5 cm) of plastic tubing that comes with the Chemistry Sensor. 1) Put a drop of glycerin on the end of the syringe. Connect the end of the syringe to the other end of the small piece of plastic tubing. 2) Adjust the volume of air in the syringe to 20.0 mL. 3) Align the quick-release connector on one end of the plastic tubing with the pressure port of the Chemistry Sensor. Push the connector onto the port, and then turn the connector clockwise until it clicks (about oneeighth turn). Check that the syringe and Chemistry Sensor have a secure seal by adjusting the volume from 20.0 mL down to 10.0 mL. (It should get harder to push the plunger as the volume decreases.) Xplorer GLX Setup Connect the Chemistry Sensor into Port #1 on the GLX. Record Data 1) From the home screen, press the Sensor button, then the Mode button ( 2) Switch the mode from continuous to manual ( , , ). ). 3) In this mode, after you press the record button, a flag appears at the top of the GLX screen instead of a clock. The GLX will only record a data point when you press .Press and enter “volume” and press. 4) Scroll down to Measurement Unit, press , enter “ml”, then press 5) Scroll down to Number of Digits and press . for one digit past the decimal place, then press . 6) Press 7) Press , to open the graph display. and use the arrow keys to highlight the X axis label. Press select volume and press 8) Press record ( . ). When you press Enter the volume and press then use the arrow keys to you will be prompted to enter the volume on the syringe. . The GLX will assign the pressure reading to this value. 9) Change the volume in the syringe by compressing to about 16 ml, hold the syringe at this position and press , enter this volume and press . 10) Continue this process to obtain about 5-6 data points (don’t compress below about 6 ml or the syringe , 9 may leak). 11) when you have collected your data points, press . Analyze Record calculations in your lab notebook as you complete your analysis. Data Make a sketch of your graph of Pressure versus Volume and a sketch of your graph of Pressure versus Inverse Volume (InvVol). Be sure to label the axes. Analysis and Synthesis Questions 1) What was the actual pressure - volume relationship you discovered? ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 2) What could have caused an error in the volume measurement in your experiment? ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 3) What would have happened to your results if the syringe leaked when compressed? ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 10 Part 2 Procedure Equipment Setup 1) Connect the tubing to the Chemistry Sensor, using the directions in the Sensor Information Sheet. 1) Use a rubber band to attach the Temperature Sensor to the air chamber (aluminum can). 2) Set the air chamber/Temperature Sensor into the beaker. 3) Fill the beaker with water until half of the air chamber is submerged. Note: do not let the rubber stopper get wet. 1) Set the beaker/air chamber assembly on the hot plate (but don’t turn on the hot plate yet). 2) Use the three-finger clamp to hold the air chamber in the water. Xplorer GLX Setup 1) Connect the Chemistry Sensor into Port #1 on the GLX. 2) Connect the Temperature probe to the temperature port on the Chemistry Sensor. Record Data Press 1) to begin recording data. Turn on the hot plate to “High” (or its hottest setting). 2) Continue to record data until the water begins to boil (or the rubber stopper is pushed out). Press again to end data recording. 3) Turn off the hot plate.Note: Be very careful not to touch the hot plate, beaker, or hot water. Analyze Record calculations in your lab notebook as you complete your analysis. Use the Graph Display to examine the plot of Absolute Pressure versus Temperature. Press 1) then to select Linear Fit. Use the resulting fit to determine the relationship of pressure and temperature. 2) From the Tools menu ( ), select the Smart Tool ( ) Cursor. Use the Smart Tool Cursor to find where the Linear Fit line crosses the x-axis. You will need to use the ( Scale/Move ) function to move the axis of the graph to find the intersection point. Record the x-coordinate of the point where your line crosses the x-axis as your estimate for absolute zero in your notebook. Analysis and Synthesis Questions 1) Based on your Pressure versus Temperature data, how is the pressure related to the temperature? ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 2) Do your results support your predictions? Why or why not? ______________________________________________________________________________ 11 ____________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ __ Based on your Pressure versus Temperature data, what is your estimate for absolute zero? 3) ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ How does your estimate for absolute zero compare to the accepted value of –273.15ºC? 4) ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ Day 3 - State Boyle’s law in words. Explain using the kinetic molecular theory. Do page 435 (2,3) . State the mathematical relationship and formula used. Solve problems do page 435 ( 4-6 ). Do page 468 ( 16,18 ) Boyle’s Law ( Pressure and Volume ) ( temperature and # of moles constant ) A Gas Sample is Compressed Zumdahl, Zumdahl, DeCoste, World of Chemistry 2002, page 429 - 12 see pg. 432 ----> as the pressure increases the volume ________________ pressure exerted by a gas varies ______________________ as the volume as the volume decreases molecules hit the walls of the container _______ often thus pressure _________ P 1/V ------> P = k/V ( k = constant ) or PV = constant = K General formula : P1V1 = P2V2 - To isolate V2 by itself divide each side by _________________ and cancel thus : V2 = Standard atmospheric conditions : STP ----> P = 101.3 kPa T = Oo C = 273 K STP STP Standard Temperature & Pressure 0°C 1 atm 273 K - OR - 101.325 kPa Ex. 1 Consider a 4.41 L of gas at a pressure of 94.2 kPa. What volume would the gas occupy at a standard pressure of 101.3 kPa? V1 = _______ P1V1 = ___________ V2 = ??? P1 = _______ V2 = ________________ = ________________ P2 = _______ Ex.2 Consider 2.0 L of hydrogen gas at a pressure of 101 kPa. What pressure would the gas exert if volume is increased to 4.0 L. V1 = _______ V2 = _______ P1 = ________ P2 = ??? P1V1 = _____________ Consider the sample data : Sketch a graph of Volume vs Pressure P( kPa ) 200 150 100 50 P2 = _________________ V(mL) 100 _____ 200 _____ do page 435 ( 4-6 ). Do page 468 ( 16,18 ) Sketch a graph of Volume vs. 1/P 13 SCH3U BOYLE’S LAW PROBLEM SET 1. A 2.00 L sample of a gas at a pressure of 1000 kPa is allowed to expand until it drops to 300 kPa. If the temperature remains constant, what will be its new volume? [6.67 L] 2. If a given mass of a gas occupies a volume of 8.4 L at a pressure of 101 kPa, what is its volume at a pressure of 112 kPa and the same temperature? [7.6 L] 3. What is the volume fo 4.8 L of hydrogen gas if the pressure exerted on it increases from 55 kPa to 127 kPa? Assume that the temperature remains constant. [2.1 L] 14 4. The barrel of a bicycle pump has a volume of 108 mL and contains air at 102 kPa pressure. What volume of the barrel would the gas occupy if its pressure increases to 630 kPa? Assume the temperature is constant. [175 mL] 5. A 16.0 L fire extinguisher contains 15.0 L of water and 1.00 L of compressed air. When in use, the fire extinguisher must expel the last bit of water at a pressure of 110 kPa. What should the original compressed air pressure in the fire extinguisher be? Assume that the temperature is constant. 6. A gas has a volume of 725 mL at a pressure of 200 kPa. Determine the pressure required to decrease the volume of the gas to 500 mL. Assume constant temperature. [290 kPa] Day 4 State Charles law and explain it using the KMT. Teacher will demonstrate law using computer data. What is the Kelvin scale and why is it used ? Analyze graph page 440. What temperature is absolute zero both in Kelvin and degrees celcius ? How do you convert from celcius to kelvin and visa versa. State mathematical formula for Charles law and solve problems. Complete page 446 ( 5,6,8,9,10,11,12 ) Charles’ Experiments with Gases & Temperature Changes keep mass and pressure the same change temperature observe the effect on volume Vol (L) - if line is extrapolated to the X axis, it intercepts at ________________ - does not matter what gas is used Temp (0C) - Kelvin stated that at this temperature molecular motion would ______________ ___________ energy would be zero; the _____________ of the gas would be zero however, real gas molecules do have volume -2730C is used as the starting point for the Kelvin scale -2730C = 0 K (Kelvin), each 10C = 1 K therefore 00 C = 273 K, TK = 0C + 273 CHARLES’ LAW - the volume of a fixed mass of gas at constant pressure is ________________proportional to its __________________temperature V V = _________ x T V ---- = -----------T Thus V2 = _________________ V1 ----- = ------T1 Ex. 1 A gas occupies 457 mL at a temp. of 0o C. What volume will it occupy at 28.0o C? V1 = ______ V2 = ???? V1 T1 = ____ oC = ____ K ---- = ------T2 = ____ o C = ____ K T1 V2 = ___________ = _____________ Ex. 2 A gas occupies a volume of 733 mL at 10.0oC, at what temp. will it occupy 1225 mL ? V1 = _____ V2 = _____ T1 = _____ T2 = ???? V1 ----- = -------T1 T2 = ______= _____ K = ____ oC Explanation: T is increased, indicating an increase in the average __________ energy of the gas molecules (i.e. moving faster) Volume of the container ______________ because of the increased collisions by the gas molecules V stops increasing when the pressure of the gas is equal to the pressure of the ___________________ thus there is no increase in pressure - Complete page 446 ( 5,6,8,9,10,11,12 ) 15 16 Day 5 Sate Gay - Lussac’s law and account using KMT. Give the formula for calculations and solve problems. Do page 449 ( 13,14 and 15 ). Read section on Compressed Gases and safety concerns. Answer page 451 ( 1,2,4,5 ). Do page 468(25,26) Pressure and Temperature Law sometimes called Gay-Lussac’s Law - P1 = k T1 therefore, P1 = P2 T1 T2 P2 = k T2 this represents a direct relationship i.e. heat an aerosol can and the pressure will increase until the can ruptures. Sample Problem A sealed storage tank contains argon gas at 18ºC and a pressure of 875kPa at night. What is the new pressure of the tank and its contents when it warms up to 32ºC during the day? T1= P1= P2=? T2= P1 = P2 T1 T2 P2 = Therefore the new pressure is ______________ - as the temperature increase the particles move faster hitting the walls of the container _____________ often causing pressure to ________________ 17 Summary of the Named Gas-Laws: LAW RELATRELATIONSHIP LAW CONCONSTANTS Boyle’s P V P1V1 = P2V2 T, n Charles’ V T V1/T1 = V2/T2 P, n GayLussac’s P T P1/T1 = P2/T2 V, n Do page 449 ( 13,14 and 15 ) Answer page 451 ( 1,2,4,5 ). Do page 468(25,26) Day 6 Distinguish between STP and SATP conditions. Develop the combined gas law. Do problems page 457 (17-21). Identify the major and minor components of the atmosphere ( see page 459 ). Pg 461 #1 - STP stands for : _______________-conditions are _______________kPa and _________ oC - SATP stands for conditions are _____________________ - conditions _______________kPa and ________________ oC 18 Pressure - Temperature - Volume Relationship P T V T V T P V V P P Charles 1 P ___ V V T Gay-Lussac’s P T Boyle’s COMBINED GAS LAW P1V1 = P2V2 T1 T2 Sample Problem Q: A balloon containing hydrogen gas at 20ºC and a pressure of 100kPa has a volume of 7.50L. Calculate the volume of the balloon after it rises 10km into the upper atmosphere where the temperature is -36ºC and the outside air pressure is 28kPa. Assume that no hydrogen gas escapes and that the balloon expands, etc. A: T1= P1= V1= T2= P2 V2= ? P1V1 = P2V2 T1 T2 V2= problems page 457 (17-21) The Gas Laws 1. The gas left in a used aerosol can is at a pressure of 1 atm at 27C. If this can is thrown into a fire, what is the internal pressure of the gas when its temperature reaches 927C? given gas law formula work 19 ANSWER: 2. A sample of carbon dioxide occupies a volume of 3.50 L at 125 kPa. What pressure would the gas exert if the volume were decreased to 2.00 L? given gas law work formula ANSWER: 3. A sample of propane occupies 250.0 L at 125 kPa and 38C. Find its volume at 100.0 kPa and 95C. given gas law formula ANSWER: work 4. Oxygen gas is at a temperature of 40C when it occupies a volume of 2.3 L. To what temperature in Celsius should it be raised to occupy a volume of 6.5 dm3? given gas law work formula ANSWER: 5. Fluorine exerts a pressure of 900. torr. When the pressure is changed to 1.5 atm, its volume is 250. mL. What was the original volume? given gas law work formula ANSWER: 6. The volume of a gas is 200.0 mL at 275 K and 92.1 kPa. Find its volume at STP. given gas law work formula ANSWER: 7. A sample of N2 occupies a volume of 250 mL at 25C. What volume will it occupy at 95C? given gas law formula ANSWER: work 20 21 Major and Minor components of the atmosphere - the major components are ______________ ____% and ________________ _____% the minor components include gases such as _______ _____%, ________ ______% and __________ ______% Chemical name of the component Dalton IUPAC azotic gas nitrogen oxygenous gas oxygen aqueous vapour water carbonic acid gas carbon dioxide Pressure (mm Hg) (kPa) 593.3 79.11 157 20.9 11 1.5 0.5 0.07 Composition % 78.08 20.95 varies 0.04 Pg 461 # 1 Day 7 State Dalton’s law of Partial pressures : ______________________________________________________________ ______________________________________________________________ Ptotal = P1 + P2 + P3 ....... Understand concept of mole fraction and partial pressure. - Consider a mixture of gases 1.0 mole of oxygen, 2.0 moles of hydrogen and 3.0 moles of nitrogen. If the total pressure is 100 kPa calculate the partial pressure of each gas . Partial Pressure = mole fraction x Total pressure PO2 = Complete page 460 (22-25) PH2 = PN2 = Summary Dalton found that the total pressure of mixed gases is equal to the sum of their individual pressures (provided the gases do not react). 50 kPa 100 kPa 150 kPa Note: all of these + = volumes are the same 1 L oxygen 1 L nitrogen 1 L mixed gas This works according to the KMT because at the same temperature molecules of different gases have the same Ek. It doesn’t matter if the molecules are O2 or H2. Both collide with the container or other molecules with the same force. Vapour Pressure Defined • Vapour pressure is the pressure exerted by a vapour. E.g. the H2O(g) in a sealed container. Eventually the air above the water is filled with vapour pushing down. As temperature , more molecules fill the air, and vapour pressure . • Yet, molecules both leave and join the surface, so vapour pressure also pushes molecules up. • To measure vapour pressure we can heat a sample of liquid on top of a column of Hg and see the pressure it exerts at different °C. Vapour pressure Measuring Vapour Pressure Vapour pressure for H2O °C kPa °C kPa 10 1.23 50 12.33 20 2.34 75 38.54 Temperature 30 4.17 100 101.3 See pg. 464 for more • When the vapour pressure is equal to the atmospheric pressure (Patm), the push out is enough to overcome Patm and boiling occurs. • Thus, water will boil at a temperature below 100 °C if the atmospheric pressure is reduced. 22 COLLECTING A GAS OVER WATER called __________________ displacement of water the gas has some________________ vapour in it since it has travelled through the water and is sitting above water thus the Law of Partial Pressures must be applied since the gas is a mixture of the collected gas + water vapour MEASURING THE VOLUME OF THE GAS the water level inside and outside the container are equalized this makes the pressure of the gas the same as ___________________________pressure PRESSURE OF THE GAS PTOT = atmospheric pressure PGAS = PTOT - PH2O Read section on compressed oxygen (pg.462), 23 Use of compressed oxygen : 1. 2. - - 24 ____________________________________________________________ ____________________________________________________________ Nitrogens many uses and undersea exploration(pg.465) . 1. ________________________________________________________ 2. ________________________________________________________ 3. ________________________________________________________ Undersea exploration considering an atmospheric pressure of 100 kPa. What is the pressure at a depth of 50 m underwater ? ___________________________ To compensate for this increase in pressure body tissues and blood vessels _________________________; this may lead to ________________ _______________ which symptoms include disorientation. If the diver ascends to fast ______________ gas can come out of solution as gas bubbles in the blood vessels ; the bubbles block blood flow and lead to a painful condition known as the __________________________. Research the advances made in undersea exploration. ________________________________________________________________________ Do page 466 (2,4,5 ). Page 467 ( 9,10 ). Complete page 467 ( 2,3,4,13,16,17,19,23,24,28,32,33) 25 Day 8 quiz on chapter 11 define the law of combining gas volumes : ______________________________________________________________________________________ ______________________________________________________________________________________ - Avogadro’s hypothesis : ____________________________________________________________ - Molar volume : ___________________________________________________________________ Vm = V/n - Do page 477 (1-4). Complete thought lab molar volume of gases on page 477 Assignment SCUBA Diving and other Applications of Gas Laws 26 PART A – from the article “Gas Laws and Scuba Diving” For each question give a complete answer and state the gas law that applies to the scenario. 1. What happens if SCUBA divers hold their breath while making emergency ascents to the surface from depths of 30 m or more? 2. Why should divers not fly or take hot showers soon after deep dives? 3. Why Is contaminated compressed air more dangerous to the diver at a depth of 30 m than at the surface? 4. Why does swallowing help with the sensation felt in your ears when your friend drives down the Ancaster hill at a high speed? 5. The air pressure in your lungs is at 101.3 kPa at sea level. What is the air pressure if you were diving at a depth 25 m? Show how you arrived at your answer. 6. A diver stayed at great depths for an extended time and she started to chase a shark. How could you account for this strange behaviour? (N.B. It has nothing to do with gender- male divers would react the same way under these conditions.) PART B – other applications 1. PO2 Air pressure at the top of Mount Everest could be as low as 253 mm Hg. a) How would this affect oxygen utilization? (Calculate PO2 and compare to requirement) b) What percentage of oxygen must be in the air breathed to ensure haemoglobin saturation? (use PO2 = 80mm and the total air pressure stated for Mount Everest) 2. It is recommended that before storing an open 2 L plastic bottle of pop in the fridge, you should squeeze the bottle before replacing the lid. Explain why this should help the pop keep its “fizz” . 3. Read “Compressed Gases and Safety Concerns” page 450 - 451. a) Why is it important that safety precautions must be taken with compressed gases? b) List the CTC safety regulations outlined here. c) What is a pressure relief valve? d) List the requirements for safe storage of a compressed gas cylinder. 4. Read page 458. Explain the role of gas pressure in the eruption of a volcano and/or geyser. 5. Read page 462 and page 465. a) How can a hyperbaric oxygen chamber be used to treat sports injuries. b) Describe three industrial uses for nitrogen gas or liquid nitrogen. 27 Gas Laws & Scuba Diving Pressure We live in a sea of air. Since air molecules constantly bombard us, we always experience a pressure of about 760 mm of mercury (or one atmosphere) at the Earth’s surface. This is equivalent to 14.7 lb on each square inch of surface. If we zoom to the top of a tall building in an elevator we are no longer as deep in the sea of air as at ground level and, therefore, the pressure around us becomes lower. Ears are usually the first to respond to this change. Wiggling your jaw or swallowing sometimes corrects any discomfort or strange sensations in the ear by opening the tubes connecting the inner ear and throat, allowing the inside pressure to equalize with the outside. A reverse pressure effect is obvious during a rapid airplane descent or during a drive from a mountain pass to the valley floor below. Divers are surrounded by water molecules in constant motion that exert pressure on their bodies. When you dive to the bottom of the deep end of a swimming pool, you feel a great deal of pressure exerted by the water. Because water is much more dense than air, pressure changes are much greater for a given change in depth in water than for the same depth change in air. For example, water exerts over 100 lb of force on the surface of a one-gallon metal can pushed just one foot below the water surface. If the metal can contains air, it would not have to be pushed very far below the water surface before the can would start to collapse due to water pressure. Can divers be crushed by the pressure of water in the same manner as the can if they go too deep? After all, for every 10 meters (about 33 ft) in depth, divers experience an additional pressure of one atmosphere. PAGES Pressure-Volume Effects The changes in pressure experienced by divers are most noticeable on body cavities that contain air, such as the lungs, the middle ear, and the sinus cavities. Boyle’s law describes how these gas volumes respond to changes in pressure. For a constant amount of gas at a constant temperature, Boyle’s law states: The volume of a gas sample varies inversely with its pressure. If divers descend without scuba gear, the amount of gas contained in their body cavities is constant and the volume of these cavities decreases as the surrounding water pressure becomes greater. However, this crushing effect or squeeze is not experienced by divers using scuba gear because the regulator on their air tanks delivers air at the same pressure as the surroundings. This means that the air in divers’ lungs is at a pressure equivalent to four atmospheres at a depth of 30 meters. If divers must make emergency ascents from this depth they must remember to breathe out regularly as they return to the surface. If they don’t, the pressure of the air in their lungs will cause their lungs to expand. The extreme distortion of the lungs can cause some of the alveoli (the small sacks in the lungs) to rupture. If this happens, air can enter the bloodstream and cause a blockage that may lead to a variety of problems including loss of consciousness, brain damage, and heart attacks. The rate of lung expansion increases dramatically as the divers ascend. According to Boyle’s law the volume of a flexible gas container will approximately double when the surrounding pressure decreases to onehalf its original value. If the divers ascend while holding their breath from a depth of 30 meters (where the pressure is about four atmospheres), their lungs would have to double in volume when they are at 10 meters (where the pressure is about two atmospheres) to equalize the pressure of the water. Of course, this does not happen because the lungs are contained by the rib cage and the muscle system, and the divers are forced to breathe out. Pressure—Solubility Effects Not only does the pressure affect the volume of trapped gases, it also influences the solubility of gases in liquids. Divers must be aware of the principles described by Henry’s law, which states: The amount of gas that will dissolve in a liquid at a given temperature varies directly with the pressure above the liquid. Henry’s law is useful, therefore, in explaining why during a dive any gases entering the lungs are absorbed to a greater extent in the diver’s blood. Although this increased solubility of gases in the blood may create no problems during the dive, the diver’s body experiences an effect similar to opening a can of soda when the diver ascends rapidly to the surface. This effect can be accentuated if the diver takes a high altitude plane flight soon after a dive. In particular, nitrogen gas bubbles that form in the blood and other body fluids can produce a multitude of problems. These problems depend on the location of the gas bubbles, the size and number formed, and the way they are transported by the diver’s circulatory system. The bubbles can cause localized pain, itching of the skin, breathing difficulty, and can lead to paralysis, unconsciousness, and death.To minimize gas bubble formation (decompression sickness or “the bends”), divers carefully follow tables prepared by the U.S. Navy that 28 describe the time limits for dives at various depths greater than 10meters. The essence of the process described by the tables involves ascending to a certain point and then remaining at that depth for a time period to allow some of the dissolved nitrogen to escape. Depending on the initial depth, there may be several of these “hold points” during the ascent. If divers experience decompression sickness, the only mode of treatment is to put them in a decompression chamber, increase the pressure surrounding their bodies, and slowly decompress them back to one atmosphere of pressure.The increased solubility of nitrogen gas at higher pressures may also have a narcotic effect. Nitrogen narcosis or “rapture of the deep”generally does not occur until divers reach depths of about 30 meters. The symptoms are similar in nature to intoxication by alcohol. The divers have a feeling of happiness, overconfidence, tingling or numbness in their arms or legs, and memory impairment. This narcotic effect of nitrogen is just one of the many reasons divers should never work alone underwater. Another application of Henry’s law involves contaminants such as carbon monoxide (CO) that might be present in the compressed air used by divers. Of course, every attempt is made to ensure the purity of the air in scuba tanks, but if a contaminant is present to the extent of just 1%, its presence is more serious during a dive. For example, at a depth of 40 meters, the pressure is equivalent to about five atmospheres. Because the regulator delivers air at the same pressure as the surroundings, each breath contains five times more contaminant molecules than each breath from that same tank at the surface. This is equivalent to breathing air containing 5% of that contaminant at the surface. As the pressure increases during a dive, the solubility of oxygen in the blood also increases proportionately. This means that the effects of poisoning by a trace of carbon monoxide contaminant may go unnoticed during a dive since sufficient oxygen is available for normal cellular respiration. However, as divers surface, the solubility of oxygen decreases in their bloodstreams. Because the carbon monoxidehemoglobin combination is so stable, there may not be a corresponding decrease of carbon monoxide in the blood. If the divers do not have enough hemoglobin available to bond with oxygen cell respiration, they may lapse into unconsciousness. Temperature-Solubility Effects Gas solubility is also affected by changes in temperature. Have you ever noticed that as a cold glass of water warms to room temperature, air bubbles form, clinging to the inside of the glass surface? These bubbles are composed of air that was dissolved in the cooler water. Can you use this information to explain why it is dangerous for a diver to take a hot shower after a deep dive? A scuba diver with a good basic understanding of gas behavior will better appreciate what is happening during a dive. If you are a scuba diver, this understanding could save your life! - Give formula for Avogadro’s law and solve problems relating to moles, volume , mass and molecules. Do page 482 ( 5,7,9,11 ). Analyze table 12.1 page 482. Explain why certain gases deviate from the ideal gas concept. Explain why real gases do not behave ideally. For gases to behave more like ideal gases what pressure and temperature conditions are used. - at STP 1.00 mole of an ideal gas occupies a volume of _______________ = Vm pg 482 ( 5,7,9,11 ) - real gases behave more like ideal gases the ____________ the temperature and __________________ the pressure ; this leads to ________________ causing an ___________________ in the distance between particles thus _______________ attractive forces. Why is there some deviation ? ______________________ and ____________________. going back to the thought lab pg 477; which gas deviates the most from being an ideal gas ______________________ ; _____________________ molecules have more attractive forces causing molar volume to be ____________ than expected. 29 Day 9 Develop the ideal gas law equation, manipulate the equation to determine the ideal gas law constant. Do problems page 487 ( 12, 15 ) Complete page 488 (1-5). Pg 500 ( 20,23 ). Pg 500 (1,2). Independent research THE IDEAL GAS LAW An ideal gas is a hypothetical gas that obeys the gas laws perfectly under all conditions i.e. won’t condense into a liquid when cooled, graphs are all perfect straight lines, etc. Ideal Gas Law Equation : How does the volume of a gas vary with respect to : Pressure : V _________ Temp.: V _________ # of moles ( n): V__________ Thus : V _______________ V = __________________ Let constant be R ----> ideal gas law constant PV = _____________ ----> ideal gas law constant To determine R consider STP conditions : P = ______________ n = ______________ V = ______________ T = _________ R = __________________ = _________________ kPa x L ---------------- mol x K Ex.1 What pressure is exerted by 0.622 mol of gas in a 9.22 L vessel at 16.0o C ? P = ???? n = ________ V = ________ T = _________ R = _________ PV = _______________ P = __________________ = ___________ = ___ Ex.2 How many moles of gas occupy a 486 mL flask at 11o C and 66.7 kPa. n = ???? V = __________ = _________ L T = __________ P = __________ R = __________ 487 ( 12, 15 ) PV = ____________ n = ___________ = __________ n = ____________ 30 31 Monitoring Air Quality During the summer many people find that poor air quality makes outdoor activities unhealthy and unpleasant. Air quality is due to a variety of pollutants that are found in the air Primary pollutants include carbon dioxide, carbon monoxide, sulphur dioxide, methane, nitrogen oxides, particulate matter (PM) , and the vapours from volatile organic compounds (VOC). Secondary air pollutants, which include ground-level ozone and smog, are not emitted but are formed during chemical reactions between primary pollutants and sunlight or components of air. In Ontario, the Air Quality Index (AQI) is used to indicate the air quality. Measurements are taken hourly of the six most common air pollutants. These six pollutants all have adverse effects on humans and on the environment when they are found at high levels. They are listed in Table 1 below. The concentration of each pollutant is converted into a number on a scale of 0 to 100 using the table below. (e.g A station reading of 25 ppb SP (suspended particles) falls in the middle of the 20–29 ppb range. Therefore, an AQI value of 40 would be assigned to this pollutant, representing the corresponding midpoint in the AQI range.) The pollutant with the highest number of the six becomes the AQI for that station with the pollutant being named. For example, it would be reported as “Barrie: AQI 20, reason: ozone.” The Ontario Ministry of the Environment website has information on each of these pollutants. http://www.airqualityontario.com/science/pollutants/ Table 1 Relationship between Ground-Level Pollutant Concentrations and AQI Levels SO2 0–15 100– 199 16–31 200– 299 32–49 300– 799 50–99 0–24 0–15 25–49 16–31 50–74 32–49 75–199 50–99 100– 199 16–31 200– 299 32–49 300– 699 50–99 0–9 0–15 10–19 16–31 20–29 32–49 30–79 50–99 Concentration (ppb) 0–49 50–99 Index level 0–15 16–31 100– 149 32–49 150– 499 50–99 10–19 16–31 20–29 32–49 30–89 50–99 Concentration (ppb) 0–99 Index level >800 >100 O3 Concentration (ppb) Index level >200 >100 NO2 Concentration (ppb) 0–99 Index level 0–15 >700 >100 TRS (total reduced sulfur) Concentration (ppb) Index level >80 >100 CO >500 >100 SP (suspended particles) Concentration (ppb) Index level 0–9 0–15 >90 >100 Table 2: Primary Air Pollutants and Their Effects Component Properties carbon dioxide, CO2(g) • has no odour or colour carbon monoxide, CO(g) • has no odour or colour Effects • • • • • • is necessary for photosynthesis by plants increasing levels linked to global warming can increase the acidity of water reduces body’s ability to use oxygen lowlevel, short-term exposure decreases athletic performance and aggravates cardiac symptoms can cause premature death 32 sulfur dioxide, SO2(g) • • • • • • • • • has a pungent odour is fairly soluble in water has an irritating odour is a reddishbrown colour reacts with VOCs to form ground-level ozone is a clear, colourless gas has no odour fuels Bunsen burners is produced by bacterial decomposition of organic waste • • • • • • • reacts in water vapour to produce acids aggravates asthma at high levels inhibits plant growth (at 0.5 ppm) causes human respiratory distress (3 to 5 ppm) reacts in water vapour to produce acids can cause human respiratory distress can cause death by asphyxiation particulate matter (PM) • • includes PM10 (particles that are < 10 µm in diameter) and PM2.5 (particles that are < 2.5 µ in diameter) contributes to smog • • • • • volatile organic compounds (VOCs) • • • • evaporate quickly at ordinary temperatures are present in the atmosphere at very low levels are found in higher concentrations indoors than outdoors react with nitrogen oxides to form ground-level ozone • • • • PM2.5 particles get into the respiratory system causes breathing and respiratory problems causes irritation, inflammation, and damage to the lungs can irritate the eyes, nose, and throat can cause premature death cause burning eyes may be irritants are harmful to humans with heart disease or respiratory conditions may be carcinogenic (for example, formaldehyde and benzene) nitrogen dioxide, NO2(g) methane, CH4(g) Gases and Atmospheric Chemistry : Relating Science to Technology, Society, and the Environment 1. Air Quality Index a) Construct a table listing the six substances found in the AQI table. For each of these describe the source, properties and major effects on humans and/or the environment. b) Table 2 below lists data for a hypothetical location over a five day period. Construct a new chart containing this information. Include a column for each day to record the AQI value. Convert each reading to an index value using Table 1 on the previous page. Add two extra rows to report the AQI value and the “reason” as described in the introduction. (Do this for 2 days only) Table 2: Sample AQI Data Substance sulfur dioxide ozone nitrogen dioxide TRS carbon monoxide SP Monday readings (ppm) 153 53 Tuesday readings (ppm) 612 85 Wednesday readings (ppm) 288 32 Thursday readings (ppm) 351 60 Friday readings (ppm) 428 76 233 771 460 588 697 17 56 33 24 46 63 428 216 412 366 12 65 32 40 62 2. Ozone Depletion and The Greenhouse Effect Two of the major environmental problems that affect Canada as well as the rest of Earth are depletion of the ozone layer and global warming due to the greenhouse effect. 33 A. Ozone Depletion a) Explain the importance of the ozone layer and consequences of its depletion. b) List the major substances that are responsible for ozone depletion. What are these substances used for? c) Describe the process in which CFC’s deplete the ozone. d) “The Montreal Protocol on Substances that Deplete the Ozone Layer”. Give information on this agreement, to include intent of the agreement, date, global participation, Canada’s participation and achievements. e) What can the individual Canadian do to help protect the ozone layer? B. The Greenhouse Effect a) Explain how the greenhouse effect occurs. b) List the gases responsible for this phenomenon. Include the major sources of each of these gases. c) Describe some of the major consequences of the greenhouse effect. d) Explain what the Kyoto Protocol is and how this agreement strives to improve air quality. The Government of Canada maintains a website with information on this agreement (http://www.climatechange.gc.ca/cop/cop6_hague/english/overview_e.html) e) Describe Canada’s commitments with respect to this agreement. f) Describe the “One-Tonne Challenge” and how the Canadian government expects this to help the country meet the commitments made to the Kyoto Protocol. g) How effective has the Ontario Drive Clean program been in reducing greenhouse gas emissions in the province? h) What are some industrial and geographical factors that might make air quality in some communities very different from that in others? i) What are some municipal governments doing to improve local air quality ? j) How can public transit initiatives help improve air quality ? Day 10 - 34 manipulate the ideal gas law equation to determine the molar mass and density of a gas. Do page 500 (20,21,22 ----> don’t forget to convert your pressure values to kPa. Also page 500 (2) Do page 522 ( 16,20 ) PV = mRT -----M M = ______________________ PV = mRT -----M P = mRT -----VM P = dRT ----M d = _____________________ pg 488 ( 1,2,3,5 ) pg 500 (20,21,22) page 500 (2) Do page 522 ( 16,20 ) SCH3U1: Ideal Gas Equation 35 1. If 2.5 mole of a gas occupies 20.5 L at 26C, what is the pressure of the gas? (303 kPa) 2. If a sample of a gas occupies 450 mL at 105 kPa and 25C, how many moles of gas are present in the sample? ( 0.02 mole ) 3. What is the temperature in Celsius of 1.10 g of CO2 that occupies 600 mL at 99 kPa? ( 13C) 4. If 1.74 g of a volatile liquid is vapourized and occupies 224 mL at STP, what is the molar mass of the compound? ( 174 g/mol ) 5. If 1.57 g of N2 occupy 350 mL at 27C , what is the pressure of the gas? ( 399 kPa) 6. If 0.150 mole of gas are collected at 105 kPa and 22C, what is the volume of the gas? (3.5 L) 7. What is the density of N2 gas at STP? (1.25 g/L ) 8. How many grams of CO2 are contained in a sample that occupies 1.09 L at 127C and 40.5 kPa? ( 0.58 g ) 9. If the density of a gas is 1.74 g/L at a pressure of 97.0 kPa and a temperature of 26.5C, what is the molar mass of the gas? ( 44.6 g/mol ) 10. Calculate the volume of 0.5 moles of methane at 33 kPa and -35C. ( 30 L ) Ideal Gas Law Worksheet 1. Using the information from STP or SATP conditions determine the value of the ideal gas constant. 2. A sample of 1.00 moles of oxygen at 50.0 o Cand 98.6 kPa occupies what volume? 3. A sample of 4.25 moles of hydrogen at 20.0 o C occupies a volume of 25.0 L. Under what pressure is this sample? 4. If a steel cylinder with a volume of 1.50 L contains 10.0 moles of oxygen, under what pressure is the oxygen if the temperature is 27.0o C? 5. When the pressure in a certain gas cylinder with a volume of 4.50 L reaches 500 atm, the cylinder is likely to explode. If this cylinder contains 40.0 moles of argon at 25.0o C, is it on the verge of exploding? Calculate the pressure in atmospheres. 6. At 22.0o C and a pressure of 100.6 kPa, a gas was found to have a density of 1.14 g/L. Calculate its molecular mass. 7. A gas was found to have a density of 1.76 mg/mL at 24.0o C and a pressure of 98.8 kPa. What is its molecular mass? 36 8. How many millilitres of nitrogen, N2, would have to be collected at 99.19 kPa and 28o Cto have a sample containing 0.015 moles of N2? 9. The density of a certain gas at 27.0o Cand 98.66 kPa is 2.53 g/L. Calculate its molecular mass. 10. What volume is occupied by 0.25 grams of O2 measured at 25.0C and 100.66 kPa? 11. What is the molecular mass of a gas if 2.82 grams of the gas occupies 3.16 litres at STP? 12. A balloon is to be filled with 30.0 kg of helium gas. What volume can be filled to a pressure of 1.15 atm if the temperature is 20.0o C? 13. In a gas thermometer, the pressure needed to fix the volume of 0.20 g of helium at 0.50 L is 113.30 kPa. What is the temperature? 14. A gaseous compound has the empirical formula CHCl. At 100o C, its density at 99.97 kPa is 3.12 x 10-3 g cm-3. What is the molecular formula of this compound? 15. The pressure exerted on a diver by the water increases by about 100 kPa for every 10 m of depth. A scuba diver uses air at the rate of 8 L/min at a depth of 10 m where the pressure is 200 kPa (100 kPa due to the atmosphere and 100 kPa due to the water pressure) and the temperature 8o C. If the diver's 10 L air tank is filled to a pressure of 2.1 X 104 kPa at a dockside temperature of 32o C, how long can the diver remain safely submerged? 16. You want to send chlorine gas, Cl2, safely from Vancouver to Kingston. Chlorine gas is very poisonous and corrosive. You have a 5000 L truck cylinder that will withstand a pressure of 100 atm. The cylinder will be kept at 2o C throughout the trip. How many moles of chlorine gas can you safely ship? 37 SCH 3U1: Worksheet ( Mix and Match ) 1. The pressure exerted by a sample of gas is 2.0 atmospheres while its volume is 100 ml. Write the equation expressing the relationship between pressure and the volume of the sample when the temperature is held constant. 2. A quantity of gas under a pressure of 200 mm Hg has a volume of 600 ml. The pressure is increased to 400 mm Hg. (The temperature is held constant) a. b. 3 What will happen to the volume of the gas? ___________________ What would the actual volume of the gas be under these conditions? A quantity of gas under a pressure of 600 mm Hg has a volume of 200 ml. The pressure is increased to 200 mm Hg. (The temperature is held constant) a. b. What will happen to the volume of the gas? ___________________ What would the actual volume of the gas be under these conditions? 4. If a sample of gas is heated at constant pressure, what will happen to the volume of the sample? __________________________ 5. The temperature and volume of a gas is recorded. The volume is recorded for several different temperatures while pressure is held constant. 6. a. What would the graph of this relationship look like? b. What would the equation of this relationship look like? A quantity of gas has a volume of 100 ml at a temperature of 200 K. The temperature of the gas is raised to 400 K, while the pressure remains constant. a. b. 7. What will happen to the volume of the gas? ___________________ What will the actual volume of the gas be? A quantity of gas has a volume of 200 ml at a temperature of -3 oC. The is raised to 27 oC, while the pressure remains constant. a. b. temperature of the gas What will happen to the volume of the gas? ________________ What will the actual volume of the gas be? 8. At a temperature of -33 oC, a quantity of a confined gas exerts a pressure of 400 mm Hg, If volume remains constant, at what temperature will the pressure reach 1000 mm Hg? 9. A quantity of gas has a volume of 850 ml when its temperature is 27 oC and its pressure is 730 mm Hg. The conditions are changed to STP conditions. a. b. c. d. Based upon temperature alone, what should happen to the volume? _____ Based upon pressure alone, what should happen to the volume?________ What is the actual value of the new volume? If the density of the gas is given as 1.50 g/L, what is the mass of the gas? 38 10. The volume of a quantity of gas at STP is 500 L. What volume will this gas occupy at 37 oC and 105 kPa? If the density of the gas is 1.20 g/L, what is the mass of the gas? 11. If a given mass of gas occupies a volume of 175 ml at a pressure of 95.0 kPa and a temperature of 27 oC. What will the Celsius temperature be when its volume is 200 ml and its temperature is 50 oC? 12. Suppose you have one litre of oxygen gas at a pressure of one atmosphere, one litre of nitrogen gas at a pressure of two atmospheres, and one litre of hydrogen gas at a pressure of three atmospheres. All three samples of gas are art room temperature. Then you transfer the oxygen and nitrogen gas into the container of hydrogen gas. a. b. c. The pressure exerted by the oxygen gas will be? _______________ The pressure exerted by the final mixture will be? ______________ Which law is this demonstrating? ____________________ 13. Using the K.M.T., explain why gases diffuse at different rates. 14. A person had a barometer on the wall of her living room. One cold morning, she read the barometer and found that the air pressure was considerably below normal. She turned up her thermostat to start the heating system and warm the house. An hour later, she read the barometer again. Since the air in the room was much warmer than before, and its volume was the same, she expected the pressure to be greater. Actually, the pressure was even lower. How do you explain this apparent contradiction of the gas laws? 15. A piston in an automobile engine compresses a mixture of air and gasoline vapour from 700 cm 3 to 100 cm3 during its compression stroke. If the gas mixture just before the compression stroke had a pressure of 75 kPa and a temperature of 120 oC, calculate the pressure in mm Hg in the cylinder after the compression stroke. The heat produced by the compression raises the temperature of the gas to 200 oC. Day 11 Do volume - volume stoichiometry problems. Do problems page 503 (26-28). Do mass - mass problems. Do page 506 ( 29,30,31,34 ) GAS LAW STOICHIOMETRY Volume to Volume - remember Gay-Lussac’s Law of Combining Volumes - When gases react, the volumes of the reactants and the products, measured at equal temperatures and pressures, are always in _____________ number ratios - the ratio of volumes in the reaction will be the same as the ratio of moles -it does not matter what the value is for P & V, but they must be the same for all gases concerned N2 (g) + 1 volume 1. 3 H2 (g) -----> 3 volumes 2NH3 (g) 2 volumes If 12.0L of nitrogen gas reacts with excess hydrogen gas at the same temperature and pressure, what volume of ammonia gas is produced? Let x be the volume of ammonia gas. N2 ----NH3 = -------- = -------------x X = _______________________ 2. Burning butane in a lighter 2C4H10(g) + 13O2(g) -----8CO2(g) + 10H2O(g) amounts: volumes: - 2mol 2L 13mol 13L 8mol 8L 10mol 10L if we have 120mL of butane, how much O2 is needed? page 503 (26-28). 39 40 Mass to Volume - Ex. 1 if mass is given and volume of a second compound is required, then treat as a mass-to-mole problem find the moles of the second compound and then apply the ideal gas law to find volume (V = nRT ) P Ammonia (NH3) gas can be synthesized from nitrogen gas + hydrogen gas. What volume of ammonia at 450 kPa and 80°C can be obtained from the complete reaction of 7.5 kg hydrogen? N2(g) + 3H2(g) 2NH3(g) n H2 = n NH3 --------- = n H2 V NH3 = Do page 506 ( 29,30,31,34 ) 29. n ______ = ________________ mole ratio : ______________ = ________________ = ________________ V = _______________________ = _________________________________ 30. 31. 34. Day 12 do more of the above problems but include daltons law of partial pressure i.e. problems where the gas is collected over water. Do page 511 (35,36,37,39). Read over lab page 512 . Do page 514 (1-5) 35. n MgO = mole ratio : n O2 = V O2 = 36. V H2 = mole ratio : mass Zn = 37. 39. 41 Dalton’s Law of Partial Pressure: Collecting a Gas Over Water 1. A 2.0 L container is used to collect oxygen over water at an atmospheric pressure of 100 kPa and a temperature of 30C. What volume would the oxygen occupy dry at STP? ( 1.7 L ) 2. 500 mL of hydrogen is collected over water at 120 kPa and 20C. How many moles of hydrogen gas would be collected? (0.024 mol ) 3. 200 mL of carbon dioxide gas is collected over water at SATP, what is the mass of the carbon dioxide gas? ( 0.34 g ) 4. A gas with a density of 1.7 g/L is collected over water at 101.3 kPa and 22C. What is the molar mass of this gas? ( 42 g/mol ) Do page 514 (1-5) Day 13 do lab production of hydrogen gas page 512. Formal lab write up required. 42 SCH 3U1 Chemistry Molar Volume of a Gas Names ________________________ INTRODUCTION: The name given to the volume that one mole of any gas occupies at standard temperature and pressure is the molar volume. In this experiment, you will react a known mass of magnesium with an excess amount of hydrochloric acid to produce hydrogen gas. The hydrogen gas produced will be collected by the displacement of water. You will then need to correct this volume to standard condition using the gas laws. This corrected data will be used to calculate the molar volume of hydrogen gas. NOTE: Since you will be working with small quantities of materials, it is essential that you work with great care. PROCEDURE: 1. Fill a 400-mL beaker about 3/4 full with tap water. 2. Obtain a 1.0-1.5 cm piece of magnesium wire. Record its length in your data table. Also record the mass of 1.0-m of magnesium ribbon provided by your instructor. 3. Obtain a piece of copper wire about 10-cm long. Make a cage for your magnesium by folding the magnesium over the wire and then rolling the wire around the magnesium. Fit the wire cage into a one hole stopper. The cage should be at least 3-cm above the small end of the stopper. 4. Obtain 3.0-mL of 2.0 M HCl in a 10.0-mL graduated cylinder. 5. Using a dropper bottle, carefully fill the graduated cylinder with distilled water by letting the water run down the side of the graduated cylinder. Be sure that no air remains in the cylinder. 6. Insert the stopper-magnesium assembly into the graduated cylinder. 7. Place your finger over the hole in the stopper and invert the graduated cylinder. Lower the stoppered end of the graduated cylinder into the beaker of water. Record any evidence of a chemical rxn. 8. Allow the graduated cylinder to stand for 5 minutes after the magnesium has completely reacted. Then, tap the sides gently to dislodge any gas bubbles that may have become attached to the sides of the graduated cylinder. 9. Move the graduated cylinder, keeping the open submerged, up and down until the water level inside the cylinder is the same as the water level in the beaker. This is done to equalize the pressure inside the cylinder. Record the volume of the gas in the cylinder. 10. Record the temperature of the water bath and the atmospheric pressure. 11. Repeat this experiment doing three more trials. Observations Trial 1 Trial 2 Trial 3 Mass of Mg temperature Patm Volume of H2 CALCULATIONS: 1. Calculate the mass of your magnesium strip by using the known mass per one meter of ribbon. You may assume the magnesium ribbon was of uniform thickness and width. 2. Calculate the moles of magnesium reacted. 3. Based upon the balanced equation for the reaction, determine the moles of hydrogen gas produced. 43 4. Since you collected the gas over water, it is considered to be wet. You need to find the pressure exerted by dry hydrogen gas. The total pressure of a gas mixture is equal to the sum of the partial pressure of each gas. Using the table for the partial pressure of water vapor at different temperatures, find the pressure exerted by the water vapor in the graduated cylinder. The equation for the partial pressure of hydrogen is: Phydrogen = Patmosphere – Pwater = _____________________________________ = ___________ 5. Convert the temperature of the water bath to Kelvin. ____________________ 6. Find the volume of hydrogen gas at STP, using the gas laws. 7. Calculate the molar volume of hydrogen at STP. 8. Calculate the avg. value for the molar volume of hydrogen at STP. CONCLUSIONS & QUESTIONS 1. The accepted value for the molar volume of any gas at STP is 22.4 liters. How does your avg. value for the molar volume of hydrogen gas at STP compare with the accepted value? What is the percent error of your results? 2. What are some possible sources of error in this experiment? 3. What happens to the other products of this rxn? 4. What type of chemical rxn is observed in this experiment? 5. Write a summary of the lab. 44 SCH 3U1: Worksheet ( Mix and Match ) 1. A manometer contains nitrogen gas in one end and the other end is open to the atmosphere. The liquid mercury level in the manometer is 100.00 mm higher in the nitrogen gas end. The atmospheric pressure was recorded to be 102.00 kPa at 15.00oC. What pressure does the nitrogen gas exert against the liquid mercury? ______________ 2. A manometer contains hydrogen gas in one end and the other end is closed to the atmosphere and contains a vacuum. The liquid mercury level in the manometer pushes 150.00 mm higher in the vacuum end. What pressure does the hydrogen gas exert against the liquid mercury? ____________________ 3. A sample of gas at 7.00oC and 304 kPa contains 1.30 x 1022 molecules of gas. What volume does the gas occupies? 4. A glass bulb with a volume of 2.750 L contains 0.2200 grams of helium gas at a pressure of 150.00 mm Hg. What is the temperature of the gas in Celsius degrees? 5. A gas at 1621 kPa occupies 11.00 L at 35.00oC. What will be the new volume, if the temperature is reduced by half and the pressure is doubled. 6. At standard pressure and 27.00oC a gas occupies a volume of 42.00 L. What is the number of gas molecules present? 7. What mass of ethane gas C2H6 at 60.8 kPa and -25.00oC contains the same number of moles as 9.200 g of nitrogen dioxide gas at STP conditions. 8. The average breath of a 17 year old male, when NOT exercising is 300.0 mL at 20.00oC and 100.0 kPa. The respiratory rate is 20 breaths per minute. What volume of air does the average 17 year old male breathe in a minute under STP conditions. 9. The density of oxygen gas is found to be 1.31 g/mL at 25.00oC and 760.0 mm Hg. If 1.00 moles of oxygen gas is present under these conditions, what volume does the oxygen gas occupy? 10. Methane gas is burnt in an unlimited amount of oxygen gas. If 5.00 L of methane gas is allowed to react under the following conditions 750.0 mm Hg and 10.00oC, answer the following questions? a. Write out the properly balanced chemical equation for the above reaction. b. Determine the number of moles of methane gas under these conditions. c. Determine the number of moles of water produced under these conditions. d. Determine the volume of water produced if the conditions where changed to STP conditions. 45 46 11. The heating of calcium hydrogen carbonate releases a gas that can put out a flaming splint. If 100.0 grams of the compound is heated, at 80.00oC and 102.5 kPa, answer the following questions: 12. a. Write out the properly balanced chemical equation for the above reaction. b. Determine the number of moles of compound used under these conditions. c. Determine the number of moles of gas produced under these conditions. d. Determine the volume of gas produced under these conditions? If 50.00 grams of iron metal is added to 100.00 mL of 3.00 mol/L of hydrochloric acid solution at 15.00oC and 750.0 mm Hg, answer the following questions. a. Write out the properly balanced chemical equation for the above reaction. b. Determine the number of moles of each reactant used. c. Determine the limiting reactant. d. Determine the number of moles of gas produced under these conditions. e. Determine the volume of gas produced under these conditions. Day 14 read section 12.4 focusing on improving air quality. Describe the Montreal protocol and why is it so significant. Do page 521 (3,4,5,10,14,15,21,22,26,27,28,30,32 ) - goal was to reduce the use of ________________ ; signed on ________________. Name chemicals that were to be phased out _______________________________ Day 15 Review : page 526 (1-9,11,13,15,16-20,23,25,26,27,28,30,32-34) Day 16 Test Review : page 526 (1-11,13,15-20,23-28,30,32,33,34,39,46) 47 Unit 4 review 1. On the following blank manometer, draw the mercury levels such that the atmospheric pressure is greater than the pressure of the gas in the sphere. 2. Solve for the missing variable in the following chart: P1 P2 V1 V2 101 kPa 125 kPa 7.52 L ? 3. A bubble of methane gas, CH4, is released from a deep bog. The temperature at the bottom of the bog is 12ºC with a pressure of 375 kPa. If the bubble has a volume of 475 mL at the bottom, what will the new volume be, just underneath the surface of the bog water level, if the outside temperature is 35ºC and the pressure is 99.5 kPa? 4. Butane from a lighter undergoes combustion in the following manner: 2C4H10 (g) + 13O2 (g) → 8CO2 (g) + 10 H2O (g) What volume of butane was burned to produce 325 mL of CO2 (g)? 5. Determine the volume of a balloon at STP assuming that it occupies a volume of 7.75 L at a temperature of 0ºC and a pressure of 125 kPa. 6. What pressure is required to reduce a balloon's volume to 2.25 L from a volume of 9.75 L at a pressure of 1.00 atm? Assume that the temperature remains constant throughout the process. 7. A gas occupies a volume of 825 mL at a temperature of 25ºC and a pressure of 0.985 atm. What will the new pressure be, if the volume is decreased to 555 mL while the temperature remains constant? 8. Determine the volume occupied by 3.45 g of carbon dioxide gas at STP. 48 9. Determine the number of moles of neon gas contained in a 5.25-L gas tank at 105 kPa and a temperature of 299 K. If the gas is nitrogen instead of neon, will the answer be the same? Explain your reasoning. 10. If 45.6 g of CO2 is contained in a 2.75-L container at 25ºC, what will the pressure be in this container expressed in units of kPa and atm? 11. A balloon is brought to the top of Mt. Logan where it occupies a volume of 775 mL at a temperature of –28ºC and a pressure of 92.5 kPa. What is the pressure at the bottom of the mountain if the same balloon has a volume of 825 mL at a temperature of 15ºC? 12. A gas is collected at a temperature of 35ºC and a pressure of 95.3 kPa. It occupies a volume of 825 mL. What will the volume of this gas be, in litres, at SATP? 13. A fire extinguisher filled with carbon dioxide has a mass of 3500 g. After releasing all of the CO 2, the mass of the extinguisher is 2735 g. What was the pressure reading on the gauge before any CO2 was released, if the volume of the extinguisher is 4.25 L and it is stored at a temperature of 25ºC? 14. A low-temperature bath is made by adding dry ice to acetone to reach a temperature of –78ºC at 1 atm of pressure. What would the volume of a balloon placed in this low temperature bath be if it has a volume of 2.25 L at 25ºC and 1 atm? 15. Magnesium was added to hydrochloric acid, HCl, and produced 5.25 L of H2 gas at a temperature of 325 K and a pressure of 100 kPa. What mass of Mg was used in this single displacement reaction? 49 16. Acetylene is burned in the presence of oxygen gas during the welding process according to the following balanced chemical equation: 2C2H2 (g) + 5O2 (g) → 2H2O (g) + 4CO2 (g) If you have 40 L of acetylene at STP, what volume of oxygen gas will you need at STP to completely burn the acetylene? 17. What mass of O2 gas can be collected by water displacement from a 250-mL flask at a temperature of 25ºC and a pressure of 105.325 kPa? (The vapour pressure of H2O at these conditions is 3.169 kPa.) 50