Review: Thermodynamics and Cell Respiration
advertisement

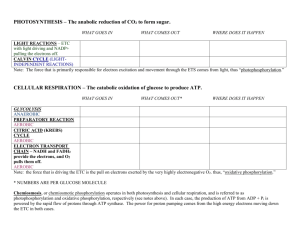
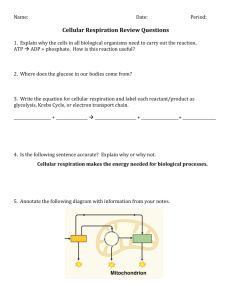
Review: Thermodynamics and Cell Respiration 1. Define thermodynamics and explain its relevance to cellular respiration. 2. Explain, with an example, what a coupled reaction is and its relationship to biochemical reactions. 3. What is Gibbs’ Free Energy? 4. Distinguish between endergonic and exergonic reactions. 5. Define activation energy and briefly describe why it is a part of all biochemical reactions. 6. What is an enzyme and give an example. 7. Explain the Induced Fit Hypothesis of enzyme action. 8. How do enzymes reduce the activation energy of a reaction? Use a graph to help explain your answer. 9. Write the (reversible) equation for ATP synthesis. 10. Describe the effects of PH, temperature and enzyme concentration on enzyme action. 11. Distinguish between substrate level phosphorylation and oxidative phosphorylation. In aerobic respiration, how many ATP are formed by substrate level phosphorylation and how many are formed by oxidative phosphorylation? 12. Identity the 3 stages of aerobic cellular respiration and their precise location within the cell. 13. Summarize glycolysis in terms of initial reactant, kind and number of ATP, NADH. 14. What is the role of NAD+ in cellular respiration? 15. Distinguish between oxidized and reduced NAD+. 16. How is glycolysis related to anaerobic respiration? 17. What is the role of the Kreb’s cycle in aerobic respiration? 18. What happens to the 6 carbon glucose molecule in aerobic respiration? Alcoholic fermentation? Lactic acid fermentation? 19. Given that oxygen is not required by the Kreb’s cycle, why does the cycle shut down when oxygen is not available? 20. Briefly describe the electron transport chain in terms of structure and function. Make specific reference to the organization of the membrane and the number of ATP made. 21. Define chemiosmosis. 22. How many proton pumps function for each electron pair released into the ETC by NADH and FADH2? 23. What are the 2 possible end products for pyruvate if oxygen is not present? Give an example of an organism in which each could occur.