SCH 4U1 MOLAR SOLUBILITY AND THE COMMON ION EFFECT
advertisement

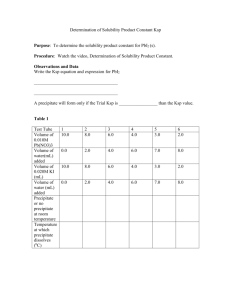
SCH 4U1 MOLAR SOLUBILITY AND THE COMMON ION EFFECT THE MOLAR CONCENTRATION CAN ALSO BE FOUND IF WE KNOW THE VALUE OF THE Ksp (APPENDIX C – p. 802). EX. 1 LEAD (II) IODIDE HAS A Ksp VALUE OF 7.9 x 10-9. WHAT IS THE MOLAR SOLUBILITY IN WATER? Ksp = ________ PbI2(s) === Pb2+(aq) + INIT. CONC (M) CHANGE (M) EQU'M CONC (M) 0.0 __ 2 I1-(aq) 0.0 _______________ Ksp = __________ Ksp = _______ = __________ 4x3 = ____________ x3 = __________ x = _________ THE MOLAR SOLUBILITY OF PbI2 IS _______ mol/L THE COMMON ION EFFECT EX. 2. WHAT IS THE MOLAR SOLUBILITY OF PbI2 IN A 0.10 M NaI SOLUTION? FROM EX. 1 WE KNOW, Ksp = [Pb2+][I1-]2 = 7.9 x 10-9 THE SOLUTION IN WHICH THE PbI2 WAS PLACED CONTAINS NO Pb2+ IONS SO ITS INITIAL CONCENTRATION IS ZERO. HOWEVER, THE ORIGINAL CONCENTRATION OF THE I1- ION HAS A CONCENTRATION OF 0.10 M. SO, PbI2(s) === Pb2+(aq) + INIT. CONC (M) CHANGE (M) EQU'M CONC (M) 0.0 2 I1-(aq) 0.10 _______________ FOR THE EQUILIBRIUM CONCENTRATION OF I1- THE EXPRESSION "0.10 + 2x" CAN BE APPROXIMATED AS 0.10 M SINCE THE VALUE OF "x" IS SO SMALL IN COMPARISON. Ksp = [Pb2+][I1-]2 Ksp = ______ = _______ x = _________ x = [Pb2+] = ___________ IF WE COMPARE THE SOLUBILITIES OF LEAD IN PURE WATER AND IN AN IODIDE SOLUTION WE CAN SEE THAT THE SOLUBILITY IS REDUCED. THIS IS PREDICTED BY LE CHATELIER'S PRINCIPLE. IF THE CONCENTRATION OF IODIDE ION IS INCREASED, THE EQUILIBRIUM SHIFTS TO THE LEFT AND MORE SOLID IS FORMED.