SCH 3U1 THE MOLE - CHAPTER 5
advertisement

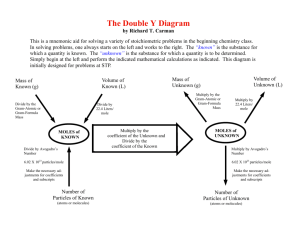
SCH 3U1 THE MOLE - CHAPTER 5 When a new compound is discovered, the formula of the compound is of great interest to scientists. When a chemical reaction is studied, scientists ask themselves how much of each substance is required for this reaction. The SI unit for amount of substance is the mole. Chemists do not count out atoms or molecules because they are too small. Measuring the mass is done instead. Thus, it is important to obtain a relationship between _______________ and ___________________________________. One helium atom has a mass of 4u (2p+and 2n0) and one nitrogen atom has a mass of 14u (7p+and 7n0). "(“u”" is the short form for "atomic mass units)" If you compare the same number of particles of each element, their ratio will always remain the same 4:14 or 2:7. It takes 6.02 x 1023 atoms (that is a lot of atoms!!! ----- 602 000 000 000 000 000 000 000) in order to have enough mass to equal 12.0000 g of the carbon-12 isotope. All other masses are compared to the carbon-12 isotope and an equal number of particles (6.02 x 1023) are recorded on the periodic table. All masses are a ratio with carbon-12. The number (N) 6.02 x 1023 is called Avogadro's number in honour of the Italian physicist, Amadeo Avogadro. One atom of an element is measured in atomic mass units (u) and is called atomic mass. One mole of a substance is equal to 6.02 x 1023 particles and is measured in grams. This mass is called the molar mass. * Using the periodic table, complete the following chart. ELEMENT H ATOMIC MASS 1.0 u MOLAR MASS 1.0 g # PARTICLES 6.02 x 1023 C __________ __________ _____________ O __________ __________ _____________ For a covalently bonded molecule such as CO2, one particle has a mass that can be obtained by adding the mass of each atom in the molecule. If that molecular mass is scaled up by Avogadro's number the molar mass is measured in grams. MOLECULE MOLECULAR MASS MOLAR MASS # MOLECULES CO2 _____________ _____________ _____________ CCl4 _____________ _____________ _____________ N2 _____________ _____________ _____________ An ionically-bonded material is made up of cations and anions. Why is sodium chloride not a molecule? ______________________________________________________________ ______________________________________________________________ In the case of ionically-bonded material, the term used is not molecular mass but formula mass. COMPOUND FORMULA MASS MOLAR MASS # OF +VE/-VE IONS NaCl ___________ ____________ ___________ CaO ___________ ____________ ___________ SnF4 ___________ ____________ ___________