The Periodic Table
advertisement

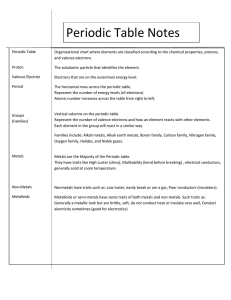
SCH 3U1 The Periodic Table Learning Goals: To determine the number of valence electrons for representative elements. To interpret Bohr-Rutherford diagrams to determine the period and group numbers. To understand the structure of the periodic table is based on atomic number and properties. Success Criteria: To draw Lewis (dot) diagram for representative elements. Organization: The periodic table is organized by groups and periods. The ________ separates the metals and ____________. See textbook (p.30) for definitions of alkali metals, alkaline earth metals, halogens, noble gases, metalloids, transition metals, representative elements, lanthanides and actinides. Bohr-Rutherford: The number of rings in the diagram is the ________ number. The group number (for the representative elements) is equal to the number of ___________________. Lewis dot symbols: Using only the element symbol, the dots will equal the number of valence electrons (don’t pair up unless you must). Eg. H , He , Mg , Ne Periodic Law: the chemical and physical properties of the elements repeat in a regular, periodic pattern when they are arranged according to their ____________________.