Document 14233629
advertisement

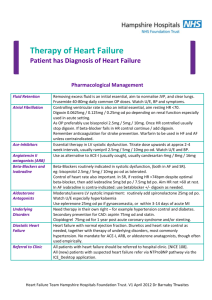
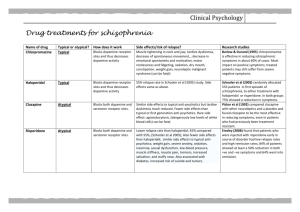
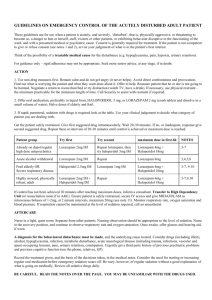
Journal of Medicine and Medical Sciences Vol. 2(3) pp. 763 –767, March 2011 Available online@ http://www.interesjournals.org/JMMS Copyright © 2011 International Research Journals Full Length Research Paper Effect of Excitatory Amino acid antagonist on haloperidol induced Parkisonian Symptom in Rat Peter I. Aziba and Ojo Ifedayo O. Department of Pharmacology and faculty of Pharmacy, Olabisi Onabanjo University, Ago Iwoye, Nigeria. Accepted 20 March, 2011 This study investigated the effect of ketamine, a selective non competitive antagonist of N-methyl-DAspartate (NMDA) on haloperidol (0.25 mg/kg) induced parkisonian symptoms in Wistar rats of both sexes, average weight 150g. The aim of the study focused on the hypothesis that blockade of NMDA receptor could have beneficial effect on haloperidol induced parkinsonian symptoms.Hypolocomotion was induced intraperitoneally (i.p) with 0.25mg/kg haloperidol, a neuroleptic. The open field test was used for assessment. Albino rats were divided into 5 groups of 6 animals each (group i, was not treated (Control), group ( ii and iii) were treated with 0.25mg/kg and 0.5mg/kg, respectively, group( iv) received 2.5mg/kg ketamine and group (v) received 0.25mg/kg of haloperidol and 2.5mg/kg of ketamine concomitantly. Results obtained showed ketamine in the concentration used significantly (P<0.01) improved locomotor activity in haloperidol induced hypolocomotion, similarly exploration, (rearing and rearing against the wall) was significantly unaltered, furthermore, the result seem to unveil the protective effect of ketamine in the concentrations used in this work. Keywords: Haloperidol, Ketamine, Locomotion, exploration Rats INTRODUCTION Excitatory neurotransmission in the brain and spinal cord is mediated mainly by the acidic amino acid, L-glutamate. Glutamate is the most abundant free amino acid in the central nervous system (CNS) and is capable of inducing an excitatory response in almost all CNS neurons. Besides its role in excitatory neurotransmission, glutamate is involved in the molecular mechanisms underlying learning processes and synaptic plasticity. Glutamate is stored in synaptic vesicles within nerve terminals, from which it is released in a Ca++-dependent manner upon depolarization. Glutamate's action is + terminated mainly by high-affinity, Na -dependent uptake into neurons and glia. Once in the glial cell, glutamate is converted to glutamine, which diffuses into nerve *Corresponding author Email: peteraziba@yahoo.com; Tel: +2348037234145. terminals. Within the terminal, glutamine is converted to glutamate (Stewart et al., 2002). Glutamate activates several types of receptors that have been grouped into two distinct classes: ionotropic and metabotropic receptors. The family of ionotropic receptors includes three subgroups: NMDA (N-methyl-D-aspartate), AMPA (α-amino-3-hydroxy-5-methylisoxazole propionic acid), and KA (kainic acid) receptors, according to their preferred agonist. Parkinson’s disease is primarily caused by degeneration of dopaminergic neurons in the substantia nigra pars compacta, which results in a dramatic decrease in dopamine content in the corpus striatum. This effect triggers a number of secondary neuronal alterations which contribute to the complex mechanisms underlying parkinsonian symptoms. Dopaminergic terminals of the nigrostriatal pathway end on mediumsize spiny GABAergic neurons which form striopallidal (“indirect”) and strionigral (“direct”) pathways (Gerfen, 764 J. Med. Med. Sci. 1992; Blandini et al., 2000). It has been postulated that a loss of dopamine in the course of Parkinson’s disease leads to functional imbalance between these two efferents (the “indirect” pathway is disinhibited and the “direct” one is inhibited) (Gerfen, 1992; Blandini et al., 2000), and finally to over activation of glutamatergic neurotransmission (Klockgether and Turski, 1989). In accordance with this view, glutamate released in excess from corticostriatal or thalamostriatal terminals activates the striopallidal GABAergic pathway, which successively leads to inhibition of pallidosubthalamic GABA-ergic neurons, activation of subthalamopallidal and subthalamonigral glutamatergic projections, and overstimulation of GABA-ergic basal ganglia outputs: the nigro- and pallidothalamic pathways (Klockgether and Turski, 1989; Gerfen, 1992; Blandini et al., 2000). This degenerative process that characterizes PD causes a functional rearrangement of basal ganglia circuitry. Thereby resulting into the impairment of the nigrostriatal pathway and increased activity of the basal ganglia output nuclei, as a result of enhanced glutamatergic drive from the subthalamic nucleus Excessive stimulation of the NMDA receptor, because of increased levels of glutamate or to decreased removal from the synaptic cleft of the transmitter, results in direct excitotoxicity which contributes to Parkinson symptoms. MATERIAL AND METHOD made of durable non-absorbent material. Open-field experiments allow the evaluation of animal’s basal activity and its evolution, in response to novelty or anxiogenic environment, and in response to pharmacological treatment, lesion or genetic modification. Locomotor activity refers to the movement from one location to another. In rodents, one of the most important components of exploration, a prominent activity of the rat’s repertoire of spontaneous activity, is locomotion. Moreover, locomotor activity and exploration are involved in many behavioural and physiological functions and are influenced by many external factors, such as environmental conditions (light, temperature, noise) Animals and Experimental procedures Albino rats of wistar strain grouped into (i-vi) of average weight150g were used in this study. The group (i) was used as a control which means no drug was administered to the rats. Haloperidol 0.25mg/kg and 0.5mg/kg were administered to the rat group (ii and iii) respectively. Ketamine 2.5mg/kg was administered to group (iv). Ketamine 2.5mg/kg and Haloperidol 0.25mg/kg were coadministered in group ( iv) experiments . The locomotor activity of these animals were examined at hourly intervals , for 5h using an open field test apparatus. The open field activity tests are used to measure locomotor activity in rodents and serves as good preliminary test to determine motor deficits induced by the Haloperidol. Drugs and Chemicals Haloperidol (Hadol®) and Ketamine Hydrochloride injection USP. were obtained from Janssen Pharmaceutical and Rotexmedica respectively. Animals Albino rats of the Wistar strain of (both sexes) were purchased from an in-breed private colony, in Ibadan, Oyo-State, Nigeria. The rats were housed in the experimental animal house, in the Department of Pharmacology, Faculty of pharmacy Olabisi Onabanjo University, Sagamu Campus, Ogun State, Nigeria, under controlled conditions with a 12 h light/12 h dark schedule and fed with commercially available rat pelleted diet and water ad libitum throughout the period of the experiment. (ii) Open Field Apparatus: The open field apparatus was constructed with plywood and measured 72 x 72cm with 36cm walls. Lines were drawn on the floor with a marker into sixteen 18x18cm squares. The arena was Maintenace of animals The open field was cleaned between each rat using 70% ethyl alcohol to avoid odour cues. Rats were handled by the base of their tails at all times. Rats were placed randomly into one of the four corners of the open field facing the centre and allowed to explore the apparatus for 5min. After 5min test, rat were returned to their cages and the open field was cleaned between tests. (iii) Measurements: The behavioural score measured in this experiment includes: • Line crossing: Frequency with which the rat crossed one of the grid lines with all four paws • Rearing: Frequency with which the rat stood on their hind limb in the maze • Rearing against a wall: Frequency with which the rat stood on their hind limb against a wall of the open field. Frequency of line crossing = Numbers of grid lines crossed within5min (5x60) s Aziba and Ifedayo 765 Table 1: The Mean Scores of the Line Crossing Frequency in Open Field Test for control and treatment groups of wistar rats. Treatment Control Haloperidol (0.25mg/kg) Haloperidol (0.5mg/kg) Ketamine (2.5mg/kg) Haloperidol (0.25mg/kg)+ ketamine (2.5mg/kg) 1 0.113±0.001 0.006±0.001* (94.7) 0.004±0.001* a (96.4) 0.068±0.001* a (39.8) 0.03±0.005** (-400)b Time of observation after administration (h) 2 3 4 0.113±0.001 0.113±000 0.113±000 0.003±0.001* 0.003±0.001* 0.006±0.001* a a a (97.3) (97.3) (94.7) 0.004±0.001* 0.006±0.001* 0.004±0.001* a a a (96.4) (94.6) (96.4) 0.060±0.005* 0.070±0.005* 0.100±0.006* a a a (46.9) (38.1) (11.5) 0.017±0.001** 0.017±0.001** 0.02±0.006** (-466.6)b (-466.6)b (-233)b 5 0.113±0.000 0.040±0.006* a (64.6) 0.037±0.001* a (67.6) 0.123±0.001* a (-8.8) 0.053±0.001** (*32.5)b Result expressed as Mean ± Standard Error, values in parenthesis represent % change; (-) = increase, (+) = decrease, (a) % change in relative to control group, (b) % change relative to haloperidol (0.25mg/kg) treated group.*p<0.001 when compare to control, **p<0.001 when compare to haloperidol (0.25mg/kg), n=6 Frequency of Rearing = Numbers of rearing within 5min (5x60) s Frequency of Rearing against the wall= Rearing against the wall within 5min Numbers of (5x60) s Statistical Analysis The value obtained from the control and treatments groups were recorded and compared statistically using the statistical package for social sciences (SPSS). RESULTS The results of the study are shown in the tables below (Tables 1-3). DISCUSSION The result reported in this study suggest a protective effect of ketamine a selective non competitive antagonist of NMDA receptor antagonist against haloperidol a neuroleptic drug, however, ketamine may not produce a selective hypoglutamate state, but more likely produce a non selective multi- system neurochemical perturbation via direct or indirect effects (Kapur and seemen, 2002) . The overall alteration as seen in the behavioural score of line crossing , rearing and rearing against the wall frequencies in the above table when haloperidol treated group was compared with the control ( group to which no drug was administered). This is an indication of the suitability of haloperidol as an agent to produce hypolocomotion, a parkinsonian-like effect in wistar rats (Ossowaka et al., 1995). The frequency of line crossing, rearing and rearing against the wall are usually used to express locomotor activity, and other gait disturbances that have been implicated with such drug as the neuroleptics, benzodiazepines, opiates, and psychostimulants ( Eweka et al and Om'Iniabohs et al., 2008). Development of behavioural measurements of locomotor activity and exploration was in part relevant in various rodent models as an initial screen for pharmacological effects predictive of therapeutic efficacy of a drug in humans. Indeed, locomotion and exploration are mediated by neurotransmitters affected by many drugs, such as neuroleptics, benzodiazepines, opiates, and psychostimulants, and consequently are changed in response to these drugs administration. Moreover, alterations of locomotor activity and exploration can have important consequences for paradigms that aim to study more specific processes, such as learning, memory reward, anxiety… Thus it is imperative to verify if a drug, lesion, strain difference or genetic manipulation influences general motor activity. Furthermore, locomotor abnormalities are associated with several human diseases such as Parkinson’s disease.The open field apparatus result showed a significant (p< 0.001) reduction in locomotor activity of haloperidol treated rats compared with the negative control groups. This decrease in locomotor activities has been reported to be 766 J. Med. Med. Sci. Table 2: Mean Scores of the Rearing Frequency in Open Field Test for control and treatment groups of wistar rats. Treatment Control Haloperidol (0.25mg/kg) Haloperidol (0.5mg/kg) Ketamine (2.5mg/kg) Haloperidol (0.25mg/kg)+ ketamine (2.5mg/kg) 1 0.013±0.001 0.000±0.000* a (100) 0.000±0.000* a (100) 0.010±0.001* a (2301) 0.000±0.000 (0)b Time of observation after administration (hrs) 2 3 0.013±0.000 0.013±0.001 0.000±0.000* 0.000±0.000* a a (100) (100) 0.000±0.000* 0.000±0.000* a a (100) (100) 0.003±0.001* 0.007±0.001* a a (76.9) (46.2) 0.000±0.000 0.000±0.000 (0)b (0)b 4 0.013±0.001 0.000±0.000* a (100) 0.000±0.000* a (100) 0.013±0.001 a (0) 0.000±0.000 (0)b 5 0.013±0.001 0.000±0.000* a (100) 0.000±0.000* a (100) 0.01±0.001* a (23.1) 0.000±0.000 (0)b Result expressed as Mean ± Standard Error, values in parenthesis represent % change; (-) = increase, (+) = decrease, (a) % change in relative to control group, (b) % change relative to haloperidol (0.25mg/kg) treated group.*p<0.001 when compare to control, **p<0.001 when compare to haloperidol (0.25mg/kg), n=6 Table 3: Mean Scores of the Frequency of Rearing Against the Wall in Open Field Test for control and treatment groups of wistar rats. Treatment Control Haloperidol (0.25mg/kg) Haloperidol (0.5mg/kg) Ketamine (2.5mg/kg) Haloperidol (0.25mg/kg)+ ketamine (2.5mg/kg) 1 0.027±0.001 0.000±0.000* (100)a 0.000±0.000* (100)a 0.017±0.001* (37.0)a 0.000±0.000 (0)b Time of observation after administration (hrs) 2 3 0.027±0.001 0.027±0.001 0.000±0.000* 0.000±0.000 (100)a (100)a 0.000±0.000* 0.000±0.000* (100)a (100)a 0.023±0.001* 0.013±0.001* (14.8)a (51.9)a 0.000±0.000 0.000±0.000 (0)b (0)b 4 0.027±0.001 0.000±0.000* (100)a 0.000±0.000* (100)a 0.017±0.001* (37.0) 0.000±0.000 (0)b 5 0.027±0.001 0.000±0.000* (100)a 0.000±0.000* (100)a 0.020±0.006 (25.9)a 0.000±0.000 (0)b Result expressed as Mean ± Standard Error, values in parenthesis represent % change; (-) = increase, (+) = decrease, (a) % change in relative to control group, (b) % change relative to haloperidol (0.25mg/kg) treated group.*p<0.001 when compare to control, **p<0.001 when compare to haloperidol (0.25mg/kg). n=6 related to blockade of dopamine transmission at nigrostratial pathway. The result showed variation in the locomotor activity and exploration among the control group, haloperidol treated group (0.25mg/kg and 0.5mg/kg), ketamine treated group (2.5mg/kg), group to which 0.25mg/kg haloperidol and 2.5mg/kg ketamine are co-administered in a time dependent manner.We have shown in this studythat deficits in locomotor activity which are associated with administration of dopamine antagonist are improved where both drugs coadministered with an NMDA antagonist ,this could the antipsychotic induced extra pyramidal symptons. Haloperidol (0.25mg/kg) treatment in the rat induced a strong hypolocomotion state within 1 h of injection, reaching a maximal plateau after 2 h and lasting for 4 h subsequent hours thereafter showed improved locomotor activity. 2.5mg/kg ketamine, a non competitive NMDA receptor blocker, when co-administered with 0.25mg/kg haloperidol in wistar rats, showed a significant (p<0.001) improvement in line crossing when compared with the haloperidol treated group as seen in table 1. However there was no significant improvement in rearing and rearing against the wall (exploration) as shown in table 2 and table 3 respectively. Aziba and Ifedayo 767 It is important to state that 2.5mg/kg Ketamine administered alone does not significantly (p<0.001) increase the locomotor activity of the wistar rats. This experiment agreed to the hypothesis that both nigrostriatal dopamine systems and corticostiatal glutamate systems participate in the regulation of striatal function (Fuxe et al., 1977, Fonnum et al., 1981) and represent major afferents to the striatum. It is therefore suggested that pharmacologic manipulation of glutamatergic neurotransmission at the level of NMDA implication of this study could involve the antipsychotic – induced extrapyramidal symptoms not necessarily Parkinson disease. REFERENCES Alabresi P, Pisani A, Mercuri NB (1996). The corticostriatal projection: From synaptic plasticity to dysfunctions of the basal ganglia. Trends Neurosci; 19:19–24. Blandini, F, Sinforiani E, Pacchetti (2000) peripheral proteasomeand caspace actinvity in Parkinson disease and Alzheimer Neurol. 69: 11037-1042 Eweka F, Om'Iniabohs (2008). The Effects Of Monosodium Glutamate On The Open Field Locomotor Activities In Adult Wistar Rats . The Internet Journal of Nutrition and Wellness. 6(2): 24-30. BLandini F, JT Greenamyre (1998) Prospect of glutamate antagonists in the therapy of Parkinson’s disease. Journal of pharmaceutical7 clinical pharmacology vol 12 issue 1 . Fuxe K, Hokfelt T, Ljungdahl A, Agnat L, Johanson O, Perez (1977). Evidence for an inhibitory gabaergic control of mesolimbic dopamine neurone possibility of improving Schizophreniaby combined treatment with neuroleptic and gabargic drugs. Med. Biol. 53(3):177-183. Gerfen CR (1992)The neostriatal mosaic multiple levels of compacxtmental organization. Trends Neuro Sci. 15:133-138. Kapur, Seeman (2002) A typical antipsychotic cortical receptor and sensitivityto endogeneous dopamine ; letter British Journal Psychiatry.180, 465-6 Klockgether , Tuski (1989) NMDA receptor; The first decade. J. Drug Asses. 3(4): 341-345 Ossowska k, konieczny J, Wardas ? (2002) The role of striatal metabotropic glutamate receptors in Parkinson disease; Amino Acids. 123(3): Amino Acid vol.14 Nub (1-3) 11-15. Ossowska K (1995) the sensitivity of striatal glutamate recptor induced by Chronic haloperidol in rats Eur J. Pharmacol. 294: 685 -691 Stewart A, Factor DO, William J, Weiner MD (2002). Parkinson’s disease Diagnosis and clinical management. JAMA 287, 1653-1661