GOUEGB in partial fulfilliaent of th© requirements for the <}©gree of
advertisement

AH ACCBISM'M) QtlMtIOH BffiflOD FOR fIE
ESHMTIOB! OF TIE SfOMGE W%
OS? PEOZBM SEJfOODS
A THESIS
submitted to
OHECCT SJAtS GOUEGB
in partial fulfilliaent of
th© requirements for the
<}©gree of
mSBSfl OF SGIEMffi
June 1961
Asaocis&e Professor ofT^Sod and Balr^ toehaolo^r
%n Charge of &Jor
Eaad of Hepsa^mnt of FQ^^M Sair§r f ©elmoXogy
Ohaimaa of" School (k-mm&tQ Cfflgaittee
Dean of Qraduat© School
Date thesis is presented
Jung ifl, IQ6Q
Typed by Dorothy Jeaa Falaatder
tmm OF eoifsKts
PASS
mMRwmm *•. .. . .•.. . . * . . .■•..■,..,..,...■....-, »..«...
x
mw*& txmkvm . . . .■. .•...-..•*< .■..• *• *• ,■. .■.,..♦.■.
s
Amiosddatlon of Fata ..................
Eadical Chain theory •• . *■■ » .• .■ •• .....■*'..' .- .
Eeaotioas of frm BidiealiSJ
Seeon&aafy Firoduet© of Autoxidation ..........
Faotors Affeeting Autoxidation ............
Effect ©f feajp©ratiyif© on th©. Quality of Froae®
Seafoods .
.■ . . .
Storage Variables
.........
Effect of Fluctuating tempsratuJ'e ...........
Effect of Antioxidants and Synetfglst© on th®
Quality of Frozen Seafoods
.
types of Antioxidants . • » *
Effectiveness of Antioxidants
.
Action of Antioxidants
Packaging ........................
Acceleration Reactions
•
Evaluation of Quality
Cheroical .
EXPERlMBOTAt WHODS
Acceleration Heaetion
Procurement of Sample .................
Preparation of Sample .................
Purification of Surfactant
.......
Method of JSixing Surfactant With Seafoods .......
Evaluation of Osidative Rancidity ...........
Treatments
.................
Effect of Purification
Effect of temperature
.......
Effect of Fluctuating feiaperature
Effect of Antioxidants .......
*
Effect of Packaging on Autoxidation of Frozen Seafood
as Detere&ned by Cteaic&l and Sensory Evaluations. ...
Procurement of Sample
Preparation of Simple
Treatment of Saaples .........
Packaging ...
Storage .................
5
§
f
9
9
11
11
12
1^
14
15
16
1?
19
19
20
21
21
21
21
21
22
22
23
23
23
24
25
26
26
27
2?
27
28
DABIE OF CQRTEHTS - continued
Evaluation ......
Ohsmical
Sensory .
PA®
*..»..
.............
...........
28
28
28
RESULfS MB D1SGUSSI0M .'...'»
,
Aec©l©rattd Oxidation Method .............
Bf^eet of Purification of Surfactant on th®
Aee©lerat®& Autosidation of Frozen Fish ......
31
31
%
Xnflmme of temperature on the 'Bm®lopmtit
of Qj&dativ® Rancidity
Effect of Fluctuating f©apsratures on ths
AutoKidstioa of Seafoods .............
Influence of Antioxidants oa th© Aeeelorated
Autojddation of Fros©n Saliaon «,»♦...
Cheisiical m& Sensory Evaluatioa of the
. r
Packaging freateent, ............
smmm AW mmwsim
B1BUG0E&P1Z .,*...
f
................. .,
33
33
jB
kO
50
52
uss OF nmm
FIOTOE
1
FA®
the Effect of Purification of Surfactant
on the Acoel&rated Autosidatlon ©f
Frozen Roekfish
,
32
fh@ Bffeet of Storag© fenqperature ©n
Aceeleratei Autoxidation of Frosen Fish
3^
the Influence of femperature Fluctuations
on the Accelerated Autoxldation ef Frozen
Roekfish
•......*
36
k
the Effect of an Antioxidant on th©
Accelerated Autoxidatlon ©f Froaen Salmon ... *
39
5
Effect of Packaging Containers ©n Tendemesis
of Frozen iioekfish Fillets as Indicated by
Sensoxy Evaluation
»
ky
Effect of Packaging Containers ©n Degree of
Ojddatlv© Rancidity in Frozen Roekfish Fillets
as Indicated by Sensory Evaluation
45
Effect of Packaging Containers on Overall
Desirability of Frozen Eoekfish Fillet© as
Indicated by Sensory Evaluation *...♦....
k6
Iffeot of Packaging Containers on Degree of
Oxidativ© Rancidity in Fyosen Roekfish
Fillets as Determined by Chemical Syaluation .
k&
2
3
6
7
8
9
.
A Comparison of Sensoiy and Choiaical Evaluations
for Qxidatlv® Eancidity Resulting From
Various Packaging freatments of Frosen Roekfish
Fillets
..
' k®
X2SS OF fASXiSS
w
1
2
3
4
5
Effect of I're-.Stor^ge t(Mper&ture fluetuations
on; the E&ta of Autoxid&tion of SoEmercially
Packaged Roeicfiah Fillets ............
3?
feaderness Scores of Frozen Rocltfish Fillets
sm Affected fegr Packaging freatment and
Storage
* • . . *
41
Rmciditj Scores of froaen. Hockfish Fillets
as Affected by -Packaging freatment and
Storage .....................
41
Desirability Scores of Frosen Eoekfiah Fillets
as Affected by Packaging treataent and
Storage
42
Chemcal Deteminatien of (kidative Eancidity
in Frozen Rockfish Fillets as Affected by
. Paekagiug Treataaent
'........
42
fh© author wighes t© exprsss his sincere thanlts to the saany
i
members of the Food and Dairy tsohnology Stajff ^ho contributed very
generously of their tia© and effort to laake the coapletion of thie
work pogaifel©.
I am particularly grateful to Profeasor Eussell 0. Sismhuber and
Professor f. C,. fu; xdthout their constant ©noouragepent, patience and
invaluable suggestions, this study would have been far more difficult.
Grateful appreciation is als© extended to Br. R. C« Cain aad Br.
A. F. AngleBder for taking the tiae to review this tianuseriptf their
constmctiv© criticisEis greatly iapr@ved the quality of the finished
■ssork.
I would also like to take this opportunity to extend tsy veiy
sincere thaaks to Professor fhomas Onadorff who was never too busy to
atop and help me tdth s^r problems.
I am very grateful for the jaany
hours h© spent in photographing and printing tuy graphs.
Special thanks go to isy colleagues Mr. David L. Crawford for
hie 'eozspanionghip and aany helpful suggestions stfid to Bra. slohn (Mary)
landers for graciously assuming many of ay duties thereby enabling ae ■
to devote more time to this wrk.
% very deepest appreciation is extended to w ^© ®®& faadly;
their patience and understanding throughout say undergraduate years was
the deciding factor for the continuation of ray education and ©ompletioa
of this study.
DBSEC&fXOl
This thesis is dedicated to the mem&rj of ra^ fathor,
Mr. Oral Edwin Palmateei*
AH ACCE1SMTED OXIDATIOH METHOD FOR TOE
BsnmTiojs OF tm Bfomm um
OF FROZSR SMFOODS
IOTROOTCTIOEI
"Stm Mghly couqpetativa nature of todays food industry coupled
with the discriminating tastes of the consumer etaphasize that only
those products maintaining strict adherence to high quality standards
will rscaiv© favorable acceptance.
It is to this end that much
research in food science and technology is dedicated.
Deteriorative changes in quality resulting fro© bacterial,
chamical, physical and enzymatic action may begin at the time of
harvest or start at some point during handling, processing or storage.
this change of quality may seem insignificant at the time, but unless
corrective measures are taken the ultimate result is spoilage and
economic loss.
Many treatments are enployed to prolong the period of
product a,eceptability by inhibiting these deteriorative changes.
These treatments may include blanching, smoking, salting, freessing,
packaging, dehydration and irradiation to name but a few.
fh© variable
composition of food products often raquires the application of several
treataents to achieve an extended shelf life.
It must be remeaibered,
however, that those treatsaents only offer a postponement of eventual
unacceptability.
Although great advances have been aade in the frozen seafood
industry since the first fish was CQWiercial3y froaen in I865, rancidity
is still one of the major problems.
In its broadest sense, rancidity
may be defined as the development of disagreeable odors or flavors in
2
fats and oils and the foods containing thorn,
lea (38, p. 200) lists
five causes for rancidity of fats and oils:
(l) absorption of odors,
(2) action of tissue lipase, (3) action of aicro-organisras, (k) atmospheric oxidation, and (5) "reversion'' and "fishiness".
the most
important form of rancidity oceuring in frozen seafoods is that
produced by the action of the oxygen of the air on fats and oils.
Atmospheric oxidation (autoxidation) occurs spontaneously when ai^r
material containing unsaturated fat is exposed to air, though the rate
of change varies enormously with the type of fat and the conditions of
storage (3®, P« 206).
this reaction, also knom as ©xidative
rancidity, is charaeterieed by the formation of hydroperoxides.
the
decoaposition of these hydroperoxides yield carfconyls which are the
compounds responsible for the off-flavors and odors that taste panels
classify as "painty", "fishy", or rancid.
Chemical, as distinct from biological, spoilage of foods is
mainly due to two types of reactions:
oxidative rancidity of fats and
oils and the so-called "brotaning® reaction,
fher© are, hmsever, many
causes of oteaieal deterioration uhich lead to degrees of deterioration
tMch scarcely qualify as spoilage, since the result is a small loss
in palatability, appearance, and nutritive value with comparatively
little effect on t&olesoaeness and edibility (3, p. .199).
^hese mild
spoilage factors may be noted as the discoloration of meat by the
oxidation of ^yoglobin to metsayoglohin, the loss of vitamins during
storage or processing due to oxidation or theraal decomposition, and
storage induced flavor changes (3, p. 199).
Preliminary stages of
3
©sddattve fancidity ma& -also be considered aador this claaslficatiou*
Huch iEiiprov©a@nt kas fesen notod In. tte qualitj @f £w>&m seafood
product© but delstoriou© changes in tte product still ocotar* fheso
ehange© maj result £rm prolonged freeaor storag®, fluctuating
t©ap©ratur© conditions and the tte® of o3§rge»*p»Qabl© paoltaging
aat^rials* the d@gi^© ©f ©siiiativo rmciditj resulting frosa tkm®
vaaAs&fod is vos^r difficult to ovaluat© «Lth the prosent a^thod©- avftUft*
bl©j partially because ©f the iliffleulty of achieving rsprosentativo
tmits® ©isiaplisigs, and th© lengtlhy g.toi?®go paidodg required for @©3^1©t©
quality &s8G!@eE2©mt3, Becmee of them smpling md eterag© profeloras,
th© valuation ©f antioxMaati and ^yaergisits. is ©jstr»©V diffioult.
Seafood products ar© m$y suaoopiiblo to ©xidativ® rancidity
foseais© of ttoir feigh iaia©4turat®d fat content. This liability ie n#i
redsaeed to aay groat ©xt@Bt by tbs • present aet&ods of fiandling, .
proesaaing and st©ra©a.
Smetsl eh©Me&l aad sensorsr aothodi:
MVQ
bsea 0iagg©st<ad -for tfe©
©valuatioa of oaidativ© raxioiditj in froson seafood produets but non©
hair© proven ©atir©2^ ssaiiefaotor^* It was not until thin 2*tiiio*
barbiturie aoid (fM) nettod tff & and Siimhubtr C?l» p* i0%*i08> va»
deveiopedi that reprodueeabl© results on intact samples wro aehi©?©d
for fisheries prod&etd*
this thesis it eonc©m@d tAth the dsvelopmont ©f an aeeelorated
autoxidation proeodur© for the ©stia&tlon ©f th© storage life in
froeen seafood produot©. ?he effects ©f packaging aaterial©, toiporature,, and antioxidants on the rat© of autoj&d&tion nwr® gtiidiod as
wll as the ^Kistane© of a possible c©s,s8sl^tioa bottssen eteKleal wtd
Qma&vy ©ve^miton^, this ppopesed, ace@l©pat®ia preesdw^ 0hD«id
peysdt tfe® rapici ©valiaatloa ©f a pree^asiag vsalablo In a Bjatt®?' of
The quality of fiah and fish productSi like other foods* depends
upon such faotors as nutritive value, palatability, flavor, odor, and
general appsaraisc©.
It is at its. best, as far as th© fisherman and
consumer are eoneerned, at the tia© the fish is removad from th© ^at©r«
Little ean be don© to iajprove tha original quality of th© product, but
aueh ea« be done to retain it.
Before w can intelligently approach
the probleias involved ^Ath quality retention, wa must first understand
the factors which cause tfrsss deleterious changes.
I* #MfiafMWA&K M. gate
• 0sddati©B by ataospterie otiggm, descrlbsd m satosidation, ig. •
of great is^rtas©© in both, the d©velopasnt of rancidity to edible
fats'aad in tha drying- of paint oils. Oxidative reactions' in edible
fats ar$ imd©sir&ble :and ooasiddirable investigation hm been dis^otod
totirard ^©.tarding ©r '©lisainaiing Xhm* th» rwwrae 4® tru@ in ptint
oils where ©sddstive raactiena a^ neetsaaryto aehieve- ptepet tordanM
ing of the finlshad surfac© (29, p* 115s 3^» P* 79)* Barly studies
rswaCIM the eo^loaity of th© reaction*
• In 'a wvy ccmprshtsusiv® roviow of early theories pertainisag to
the m®GfopM&m of autossidation, I®a (3^, p. 86*f2) points out that th©
autoxidatioa of an organic substanc© -with a carbon to carbon double
ti
bond was recogniged fey Schonbein in the Qm& of turpantta© as early
as 1858..
B® also, indicates that modem theories as to the ©eohanisa
of reactions of this t^fp© date f roa the work of Bach and
of Englex6
and Mild in l8$y»
A.
itadieal Chain Theory,
?he radioal chain theory is the currently accepted theozy of
autoxidation (29, p. 115-11^; 35, p. hB^k®; 37, p. 13Q3i *2« p. 126;
^8, p. 238-240).
According to this theory ©Kidatioa takes plac© not
at the doable bond itself, but at th@ reactive aethylone group1
adjacent to a dotibl© bond tdth production ©f a hydr0p®roxid@ vshldk
still retains the original unaaturation.
Mi Iho ssn® time a
resonance systea set up by the froe radical leads to the production
of conjugated isosaers.
Since geoaetrie isOTaeriaatlon of a considerable
proportion of the double bonds from the ois to the tfia^a configuration
also occurs, the product of this first stag® of autoxidation i© a
eoraplex mixture of hydroperoxides isith a high content of conjugated
a**^ trans unsaturatlon (37, p. 1303).
the fundamental concepts
leading to the acceptance of the radical chain theory have been
substantiated by the developzaent of new or isaproved techniques in
low*temperature orystalliaations, chromatography, counter-current
distribution, polarography, and spectrophotometry in the ultraviolet and infrared ranges (kZ, p. 126).
Fanaer (2k, p. 86-93) stated that the foraation of free radicals
fros fat molecules is dependent on hydrogen lability; the latter
determined largely by the type of unsaturation pattern and also by
the presence or absence of substitution on the ethylenic carbon atoms,
fo illustrate this, Farmer (24, p. 86-93) groups the four types of
7
ti&£atiar&t§d systems on th© feasia of hydrogQint laMlitjr a®' follows*
K
I
I
Group XII
I
E
I
%oup JX
I
C * C * € *• C
B
I
Group IV
0 * '0 # C * C
Group I shows the highest degree of hydrogeHi lability since the alphaaothylem group is flanked by adjacent double bonds*
iroup IX shows
less lability because th© influence of th© two double bonds is divided
between th© two aethyloa© groups.
Group III shows still less hydrogen
lability, and Group XV* b©ing a conjugated olefln has lost most of
its hydrogen lability (42, p. 127).
According to Eoeh (35* p* 49)» th® mmrgp tor fre© radical
production stems ftorn th© fact that the doubl® boad is §apable of
capturing outside sources of ©ner^, such as light and heat, eaeriy
which puts the ©lectrons in a higher ©nergy of excited stat®.
KJhg®
©Eotjgh efi©rgy has been absorbed &o that th© elections reaoh a critical
excitation lewl, ■tho"ese©0a mmgg- i@ dissipated by tte ©leistrort'
breaking ataay from the- r«sgt of th© moleeul© and taldn$ a protoa tiitfe
it.
.Both th© fatty acid moleeule yieaainisg and th® released hydrogen
stm are r©f®rr#i. to as free radicals siac© they possts$ us^pais*@d. ■
eleetroas.
Free radicals are extremely unstable substances and will tend
a
to seek another electron to complete the stable electron structure.
Sm& of th© methods by tahich the fatty ester free radical can obtain
an electron are shown below*
•CHgCHsGHCHGHg-
+ B.
.tC^gGBeCH^HCBjg*'
*Gi*GHCH..
•-C^CHsCBCHGHg-
-fc-J
0.
-CB^H
.CHgCU^CHCEgClg-
6
0
i
etc.
free radical has a high affinity for oxygen %Mch is believed
to add as molecular oxygen to produce a peroxide.
fhi@ peroxide may
react with a free hydrogen atoa to form a hydroperoxid© and thereby
terminate the reaction, or with another molecule of oleie ester which
would start a chain reaction that could only b® terein&ted by reaction
with another free radical or Tsrith an antioxidant.
the oxidation of polyunsaturated fatty esters, such as llneleate,
follows the same steps except that the hydrogen xMch escapes is
believed to com© from the active Methylene group (35. ?• ^9*51).
9
£• ^Q-eo.aggJpy Prodacts pS, Aui
She h^droporoaides which for® the prtosry atatoxidation product
axo lafoil© eoiapounds readily undergoing further reaction, accounting
for the decrease in peroxide values and iodine numbers during the
latter stages of autoxidation (29* p* 119)*
Sera© of the secondaxy
reactiona of hydroperoxides are as follot^sr
^JSL
5M*GH«BCE.
»eH*CilteCH.-
hydroperoside
* ^20
<t)
O2
(u) a.
-CH-.CH=GH»
D.
OS 0
+
,CE.CH^
OH 001!
-CH2-GH=eH=
*CBUCE.
OH CO.
Factora Af feetAna Autoxidation.
Xt should be noted that factors affecting autoxidation may be
either eat&lytie o** inhibitory in nature.
Many factors are known to
10
accelerate' ©r eatelya© autoxldative reaciions*
the prdxidant eff$ct
of isetalS', either in metallic form or in solution, on the rate of
autorMatioa has been shorn by m&iagr authors (15, p. ZkX+Zkbi %, p.. l^
161; 38, p. 214? 41 # p.; 119} and 66, p* 32«»:33).
Otter investigators
(8, p. 27»31{: 56, p. S4*26) shots' that there is a direct correlation
between the heiaatln*c©mpeund content of the variou© speciea of fieh
and the -oatalyiic: effect m the linoleatss oxidation.
GQ&
(15» P» 241*
244) states that the prisaary factors Aich aceelerat© oxidation in
fats and oils are metals and light.
That light has a definite
proxidant effect is w&ll substantiated <36, p. 139-152; 38» P* 213i
68, p. 36-<»37).
^kis oppllea not only to photosensitization by
pigments but also to photoexcited electron transfer due to the presence
of trace metals.
Air, moisture, and temperature may be considered as
contributory factor© (15, p. 241*244).
feiaperature, as a proxidant
in thermal oxidation reactions is noted in the work of Bishov, Heniok
and Koch (7, p. 174»182) and also in connection tilth the well known
Active Oxygen Method (4?, p. 39'»»398).
Ifo® effect of fluctuating
temperatures on the autoxidation reaction has been reported by
nuraerous workers (44, p. 200-205? 46, p.
19*27J
68, p. 37'-3Q} 69,
p. 311-31B).
Some of the factors having an inhibitory effect on autoxidation
include cheaical antioxidante, natural antioxMants, low tsiaperatures,
smoke, spices, ehelating agent®, and packaging materials (18, p. 123124; 38* P* 216-222; 48, p. 251-268; 65, p. 321-322; 66, p. 33*34;
68, p. 34-42).
The effect of these factors will be reviewed in
greater detail later.
n
iiualit^ of fwm Seafoods^
Mnefei has besn ^Jrliten m t&© ffeeaiag metbiod of preserving
fishorr pp©djaets«- 40 t«mg (70, p. A47) polats out, rafli0 stttbj©ei
lias b©©a c@^©r®i f*em sabstaatlallj ©v©sy aspsiet of intareat to the
t$<Ama3Logi9t, so ^ai If kaowledgd' in itsolf were th© k@^ td Idg^
cpalitj,
©B©
«jttl<j ©xpaet that nothing but ©Keslleat tm&m tisb
maM b&- aark©t®d. in Aaesfiea toaaj?,* Sine© this- is not tfce ease,
it is j|#@Ht©d toportaat t© F©vi©t'f
©OBI©
of ttie aspaeta @£ %mp®mkvtf@
aad its ©ffeet on ^ie qtuH^r of irosson ooafeoda* ff«ssl©r ©M Sv©r©
(65, p» 7^7)» i» th©i? voty mmprnkmsiv® ^tv^ ©n the fs'^aing
p^s#wati©n of. foods* point out that MCfo of th» ©ariy <s^p©riiitntaX
work on gvoming ma oar3?i©^ omt ©a fish, Qssiek fre^aing. of fisSi
was a coosiOB ee^areial ppaetie© for a decade prior to its adoption
£©*• th© freezing of other foods.
A. H^gaj^Og^iyii^ With .atapafffo
Wben fi©h Aieh haw been froaen and h®ld in «©M storage for
an ©xtestded period of tiia© ar® tha«d aad ®«»in©d, a nmiSasr of
change©, stay b© fmmd to hm® taksn plao© ^sioh asj difforentiat®
thosa from *m£r©z©n fr©@h fish, fh© fi©sh of th® tlmwd fish «ill
OSTisist of teso phases^ th© solid flesh plug a flaid know a@ ^drip9
■Eihioh wm not r©absorfoo& bsr th© flesh after thawing,. Also, the
textur© of th© thawed fish say foe soft and the aurfaoe m^ ham
homm ^q©iceat$d by ths loss of laolstiMf®, Mi®r oooldng* tho £is% faay
b® £mM to hsv© aeqid«^.(l m &£i*£tm®r or tli© fleoh ®^ be tcmgh aM
fi^pduii These ^liaisges ivm the (Sriglaal ^eaflltio© of tin© xmitozm
£i&h m?© SMatiaes ©atsaod by the fip^ssins pwe^eg* Aetual^, almost
all of' ©uqh Qhaag^gs art oaaoM, »di % th© ij^oaing of tte fish, bat
mth©r tjy tha si&seqWKit eold storag© ©f 'Wte- trmm %ir®&m% (57» p» 3).
On© of the ©ayli^t ehasngeg taldrsg pla^© la fish ©eewa shorty
after they a** trnghi* . ffai© cduungo is teotaa -as j&ggg M^^IM ^^ i9
eharaeterlati® of pdi'feeUy £rssb fish* jUseg ®s|i^, Is eaiaB^i W
the) eoagfcl&t&oH of tlio ©sH pv-@%&im and. i© »*©e®@fd^@d ^y a genoral
b©«|gr flgl^it^ er ©tiffisis, fhls eonfaitioa eois^ about saoxiotf md
lasts a ©^©Her tin© at higho^ tsstpofatw©© thoji. at lamt %mp®mtema
(&** P» 3^9)*, Aithonagh lot? towpssfatar© storagct h©lpe to prmQfit^r
mtard enB^aatle autdlysis md'buoterial iavasiOB (6^., p. 33^335) t
cai?© w©t %© takesi to ps^voat ©tkar ehaiag©@ taMssg pt&m in the f^oaoo
prodtto^ tosoaHse of the storage oonditions (57. p* i7«^7| ^^» P* 335$
#5.. p.. 31?*52§)* fh'SS© ohsmgss :fflajst be oleaK1^ ra©©g»ls©d and
aroiisd as far as poeaiKUu A® iadleatsd bj Stansby^ Potti^ger imd
l:%ai20M C5?» p. 2» 17*2?)* th© ohattg@s a^r fo© rdoognlsod as desiecatioa,
off#flavor do^iomonfc, *dripw, and eolor ohasig©#* %©y susggest that
^araful eoatrol of tesatdity, air elrculatioaj teap^riitur®,, and p&efeaging
®a^ b® v©!?^ h&ipfttl In ainiaisljig thqs© ehaagag.
Ift ©asj^r artlolss dealing tdth th@ ©torag© of froaoa foods»
donsitorabl© smphasts has bom placed .on th© need iot hoping footS©
at a ©©iastsmt storage tsBjpsraturG at all times. It is dbvious that
tills eonetitioa is alaost irapossibl© to staintain, Soias flwetaaticja
alw^s o&au&a during the om«®t£ eyol® of th© refrigerator eQ^T@&mtB
as tj^ll as t-ffam the fjpeeaoj? dao^a are. ©p©a©«S .or .«fe<gn larg© quaatitiss
of ■earsjar fish ar© ttepesitM in th© oold storag© roos©. Pottissg^r
(b6, p, 19) lists rajsaemia iteas that ism ems® flwiuatidas in
teiapsratiaroa. ?tea© meg inelmd© pmor failure, eqaipasent fcreafcstom,
an€ transportatioa.of th© frosen prodmet from th© predsiesr to th®
oemstsa&r.. At ©no tise it ma thoiaght that fluotuationa in toapsrature
mm hanaffel t© th® quality of the froaen fish by oaasing larg® lm
cryatalg to fossa ^ithi® th® tisgma t-Moh sight result in alteration
in textare., -Pottinges!? C^* p. 19-2?) hm ohoim that ihie theory dms
not hold* Aecor^ing to Stansby gt ^, (57, p. 21.), fluetuating'
teinperatttros say aecel©rate. oxidative ©fearsgei aad bring abomt an
inereas© in th© frost foftaatlon sitfeia pa^kag^s tjhero air poekets ar^
present.
Hikkila and Uako <^r p* 200*205)} Seagraa (52., 106*109) arwl
Staasb^ ^ a|. (5?# p. 23*24) suggest that th© alterations in the
tejttur© of fishery products are due, at least in part, to protein
deaaturatioa eaueed by flaotuatieas .in teaperature.
14
Xt has long feeeia kaom that th@ roaotloa betwen asi autojddia^e
stibstaac© arad. oj^gon ©aa ba dsl^ed for Xo&g period© of tie© % tM®
pr@3©n6© 0f v®sy Mall eoneentratioa© of additives v^riousl^ toiiaed
iBhibitors or aatioiddaat©.
Jasoba (32, fol. JXtt p. 193?) points out that tfo© foUo^dag
criteria for a che&ieal additiir©, as set fey H. S. Banaard in 1911
still hoMs;
1. It must not under any reasonable condition injure
tto health of the eonsuaer.
2. It aust not allow the tttilization of unfit tm
mterial.
3a Its m® ismst not oak® possible the (mplojmnt of
oarelesa and isperf^et Biethods of manufaotwre.
^. It aust h© non-irritant.
5. It ©ttst'b© @ffiei@nt in its aetlem.
6« It. £a»st not retard th© action of digestiv® ®m$m®*
7. It saust hav© no t®©iea©y t© deeecpos© within th©
feo# into s^bstanoes tshloh bm® a gF©at©r tosioity
than that of th® pr&aervatiir© itself.
8. Xt shotald l@na itself to siapl© Mthod® ©f
^termination m& thm ©ij^lifr tl» control pr<M,m,
On© of th® biggest probleH® in th© preservation of fat® and
15
fatty foods is rancidity caused by oxidation of unsaturated fats,
producing aldehydes and ketones that have strong and undesirable
odors sad tastes.
Antioxidants applicable for stabilisation of
unsatur&ted fats are of two general types (59. p. 73*'7^)'
(1.) $he
phonolies such as bmtylated hydroxyanisoiQ^ (BH&), nordihydroguaiaratic
acid (HPdfA), propyl gallate (PO), and the tocopherols which inhibit
fat oxidation by reacting idth free radical intermediates of the
oxidizing fats; and (2) the synsrgists, or aetal deactivators -which
have no direct effect upon fat oxidation but supplesaent the priaary
phenolio antioxidants, usually by forming stable chelates with trace
metal contamnanks.
Citric acid and ascorbic acid may be classified
as good synergists.
B. iXfectjveness ^f Antioxids^ts.
The effectiveness of the above compounds, either alone or as
synergistic mixtures, has been demonstrated by many authors for a
did© variety of fats and fat*containing foods:
seafoods (5, 25^'260;
20, p. 371-383* 51, p. 1-6; 61, p. 137-15^; 62, p. 237-247); cereals
(58, P. 585-587)* turkey (y*, p. 308*311$1 irradiated cooked pork
(60, p. 635-639); and fats (19, p. ^57-460; 2?, p. 386.390).
According to Letf and Tappel (39, p. 285-289), a synergiatic saixtur©
of W3Qkt BHA and ascorbic acid tuas very effective in retarding
rancidity in fat containing products catalyzed by hematin coiapounds.
It is strange that with all this evidence as to the effectiveness
of antioxidants and synergists, little or no application of their
use is noted in present-day coamercial fisheries products.
It may
b^ m SimBW* Bl £k» (57, p* 32.) point omt, that oXUmngh. awtioxidaBt©
a^e effeetiv® in sretsrding anitoxidatioft ol" oils* when the oils are
present, in th® tissue, as in tha ease of frosen fish,, the effeetivemB® of the aatioxidant is greats diasiaished b©ea»se of the aoisttu?©
present,
fh© action of antioxidants accox'ding to Moek (35, p.. 52), lends
support to the free radical ehaln theory.
&u aatiosidant has an
. affinity for free radicals and will give tip a hsrdrogea atosa to the
free radical more readily than will a fatty aeid molecule,
the
action of the free radical is stopped ^hen it accepts the hydrogen
atoai and consequently the chain reaction is terainated.
fhe antioxidant
becones a free radical tatoen it gives up the hydrogea atom but it fonas
a 8@£2i«-qain©n@ struetttre xMeh is- smeh sore stable than a free radical
©f th® fatty aeid type,,
th© reason for this stability iss that th©
©leetron is capable of a jaovement through the ring struettire as
illustrated for the structures urittea for the s®ai*quinoBe.
OCH,
OCH,
-H-
OCH,
CXH,
^
>■
OH
-C(CH3)3
-CCCH3)3
-C(CH3)3
I
o
II
o
fhia i© tmom m resonance and iaparts stability to the girueture.
This resonating stracture is ia^jortant since it is not strong .enough
to propagate a chain reaction by taking a hydrogen atoa from an alpha-
uX
17
sastlhylafie gfonp of a .fatty aidd* Hotsew^r, if tho. s@Eii*qtti!aoR© showii
eueoumter & f2^0 fegrdrogm atoa th© ©rigiBal aatioxiiant mmQA h&
msUmd (35* p* 52).
W. FaekMJsaa
Fa^kagiag is an teportant factor in ih© retentioa of quality of
froaesj foods teeaws© ^ffi@i@ni paekagss, aaoag ©tfe©r thiag®, preTOsit
drying out (dQaioeatioa), eontact tsitk air (oxidation),, sad loss of
th© natersl, fr©sh flavor*
Ball (2, p* 703*704) siaggoets that th© appraisal of & container
fe© based on two 8©ri©a of faetorsJ
(l) basic faotors m®h a© pries
t» paeisager, sasiitajcy qiaaliti©©, r©sista«e© to is^act. .injurgr*
©ffieaoj of interior s«rfae©, and afoserac© of haadHng problasas; aM
(2) conswsBor aoeeptaifse© faotors sttch as sis©».
QB&Q
of opeaing, rsaeal
featwr©©,. pouriag qualities,, light protection., tre«spar©ney, taE^psrpipooimm-, sM ©as© of disposal* A aaathod of ©v6lwation is suggested
mim th© abov© faetors» aceoapaDded by a very detailed o^lanatios
of ©aeh faetor aj^l how it fits into th® overall pioter© (2, p. 704*732).
Brom© (9» p* 1992~20i7) givas a v©ry coBipr©h©Bsiv© coverage
of the fttnetion© of packaging and the saateriaia xwed in their aama*
faotur®. He points out that paokaging of food, for the most part,
ie striotly utilitarian;, md that the prin&ry purpose of th® food
package is to preserv© the flavor and teep th® produet in good eon<3itiO!a
until it reaches th© eonsumer. fh© ftmetions «feich paeksging perfor®
18
are protection, convenience, eeonongr, and appeal*
materials are used for packaging containers?
Many different
paper, plastics, fibers»
metals, coatings and laminations consisting of a variety of substances.
Bach type of food product augt be evaluated as to the particular type
of packaging best suited to give it maximusQ protection*
Various protective coatings are used to protect fishery products
during frossen storage.
Probably the flfrst ''package0 that caste into
general use was an ice glaze*
$tat until the introduction of quick
frozen foods in prepared form was any serious attempt made to improve
upon protective covering for frozen fishery products (57, P* ^l).
Stansby, §t a|* (57/ P* kl-^) list many types of proteetive coverings
used for frozen seafood products and explain the functions of each
type*
These may include:
(l) glazes, which include ice glasee,
pectinate films, gelatine-base coatings, and thenaoplaatie waxes;
(2) films and wraps such as cellophane, polyethylene, aluminum foil,
waxed paper, and plastie*paper larainante; (3) packages and cartons
that are generally made out of paperboardt and (4) metal eontainers
such as the hermetically sealed tin can and the semi-rigid aluminum
pan package.
Ag one can r©adily see^ • the paek&g© plays an itsportant role in ■.
the preservation of frozen seafoods*
Undesirable -changes in quality
due to the action of air, light, and moisture can be eliminated or
at least retarded by'reasonable care in the .selection of the proper
packaging container for th© product to be frozen*
19
I4any acceleration reactions have been proposed and evaluated
as a raeans of evaluating various treatments, additives, and relative
stabilities of fats and oils <?, p* 174«182j 16, p. 47*72, 98»10l5
S3, p. 1O$*109; ^3, p. 191-19^1 ^5. p. 161*1^)»
Qearhart, Stuckey
and Austin (28, p. 427*430) compared the oonventionsil Active ©xygen
Method, the Schaal Oven Method, and a norlificd ASTI-I Oxygen Bomb iiethod.
for detewaining th© stability ©f fats and oils, and of fatty or oily
foods.
They found that the ASfM borate data eorrelated x*@ll with both
the other methods and has the advantage of speed, precision, aM
siaplielty of operation.
Filer, Hattil mid tongenecker (25# P* 19^*
201), using a modified Swift Stability test, studied the rates of
change of peroxide and iodine values, eaponificatlon equivalents,
and the amounts of "linoleie and llnolenio acids" during the
accelerated ran&ldificatlon ®f hydrogenated vegetable oil shortenings
and refined cottonseed ell,
these acceleration reactions may be depen-
dent upon heat, light, or a supporting media such as sand or cellulose
to achieve the desired results*
VI.
EvaluaUon ££ .QualLto
The majority of the present day methods for determining quality
generally fall into one or more of four categories»
chemical, microbiological, and physical.
sensory,
Of these, the efeemieal
tests for various indices seem to predominate.
20
A. ghf^ajfrU
Ag am stated above, choaioal d©t©rmriations as indioators of
quality isi food products m<m to pr©d«inat©, Various iMdie©^ ar©
tised,. thesis iiielado iriaoth^lQiainQ and trlraetl^lafflin© osid© (10,
p*. 3IOiU30%{ 21» p.
23#25J
22, p. 2^2-29^)$ SttoeinLe acid (31, p. 842.
8%?)} md volttila aoidi aad bases (30, p* ?&%'T?&i 53* P* 227*231}
63, p* 614-.617)* ^bod dorrslatioi is ^hoiim tetwea the <s©at©3it of
volatile redssoteg aubstaaQ©© md orgiMolaptio jtidg^Msit© ©f freshness
in a "mri&ty of flish md fish prodtutts (23, p.. 67?#6BQ)t th® thlo*
barTbittirie aeld (TBh) test has beea applied to a variety of foods as
& aaasure 0f quality ai affoetod by oxidatiire raaeldity. fh©s@
Indluddf butter^ai (6, p» 138**1^5)| oat ooreal©, oatiioal oookies
sad aoda oraekors (12, p. 185*106)» fish (26, p. l«35j 71, p. 10V108)j
cooled oysters (50, p. 7^*32)i and fats (5^, p. 6Q3-6Q6). A quanti*
tatlve method for the determination of n&lonaldehyde (the red pigment
developed by the fBk test) is presented by Slrmhttber and Tu (55, p« 912); th© tem, fM. number or Jailligraas of malonaldehyde per 1000 gram
of sample, is suggested.
21
iiiWMm mHOPS.
Pr^sh roe&fish fillets (a^baatods^. ja.) tore ptarohased locally
as msedMt hircbaso© mr®. Ba4© ©a th© saaa© «Jaar th© fish ^©re dslitr0r©d
thereby assuriing relatively frosk saapl^s for ©aeh tr®ats©nt.
%prosipat©ly t«0' powad© of tha fr^ah fillat «@r© passed through
a liand*op@ratetJ maat grinder tMoh had fe^en aodlfi©d to prevent iron
oontaiainatiofi' by <*ro« plating tl\©,iat©riaal parts and installing a
Fomiea fa^se plato* Mier tesiug. grouadt, tl»e sasapl© urn© well aijsedi.
quaHer®d# s^d ths alternate quarters ^dieearded, ^is was repeated
tfere© tiaraa- after vMsh a 10 * 20 grssa sas^lo mm r©s©v@d,
& di&tosnaeeotts silica, Colit© Analytieal Filter-Aid, ami ^sed as
the support siedia for this aec@l®ration prooedur®, the surfactant,
a product of the Celite division of th© flohno-Manville Company, -ms
8©l©et©d beeaus© it is eomercially availabl© ©nd uniform in quality.
Preliainary inv@stigationt indicated that this product does not
interfere in any my with th© T&h »©thod employed for cheadeal
d©t®minatiion"'of oxldative rancidity. Ifenufaeturor's specifioationa
of this and giailar diatomacoous ©arth products point out thoir large
surface ar@a per unit weight (i.©., frow four to si* aquar© aetars per
22
gram).
Chemical analysis for the presence of trace naetals which would
influence the oxidation reaction revaaled appreciable amounts of iron
present.
Purification by removal of tho iron was aecoaaplished by
reacting the surfactant with aqua regla for 48 hours, after which the
solution was transferred to a large percolator and washed continuously
with distilled water, using suction, until a negative chloride test
was obtained with silver nitrate.
Excess water was removed by suction
and drying then complfcted at 212^.
*>•
Method of E&xing Surfactant With Seafoods.
A raindcEa sasaple (10.00 g.) of the ssafood to be tested was
obtained as previous^ described and raixed with purified Celit©
Analytieal Filter-Aid (30.00 g.) by thoroughly grinding th© sample
in a number six mortar with a small amount of surfactant,
the
remaiader of the purified surfactant was added after a hoaoganous
mixture was achieved and blended into th© original to yield a fin&l
proportioB of 1?3 (fiah to surfactant).
Previous investigation by
th© aathdr indicated that a hooogisnous Bdxtuii^ was thieved after
sdadng for 20 minutes in 35 '$ r®mt
Standardisation of th© saisdng
procedure as to time and temperature is important to achieve
uniforraity between treatments,
the aixtur© was nest passed through a
20 mesh nylon screen, to eliminate any caking that a&ght occur, and
placed in suitable containers for storage at the designated temperatures.
B.
Evaluation of Oxidatlve Rancidity.
The 2-thiobarbituric acid (TM) method of Yu and Slnnhuber
(71, p. 10^-108), was used for the chemical evaluation of oxidatlve
23
rancidity.
F.
fh© Allowing titeateGnts w&re desigae^ to show the effect o£
Certain variables ©a the accelerated autoxidatioa rsaction o£ seafood©,
th© r@awlts of th© fBfi resetion for tha detos^daatlcwai of «idatiTO
rmoidlty, ®%$>r®m&& as fBA iawab©i% ®or© plott©d against tia© in da^s.
' 1. Effect of P^rifieatl^afr fh@ folloitdfis method m© wsed to
detemla© the aoed for purification of surfaotant and. th© ©ffeet of
metal (@spr©ss©d as p©rc©at ferric oiside) on th® aecelorated
atttoxM&tioa t©st. A raiidoia ample of fr©©ht ground j^ockfish fillet
ma divided into thm© portions aad indi'eldual^ saixed with the
three selected diatomaceous silica products' is a isortar using th©
prescribsd procGduro* Th© products used mv&t Calite %9t Celite
Ana^rtical Flltor-Aids and th® pwifled Galite AmOytieal Filt©r^Aid.
The latter' material was purifisd, as previously deseribed by treataant
^Ath acid.. ^h@ iron content Coxpr&gged as percent forri© oxido) of
Celite 319 and ©alii© Analytical Filter«Aid, as stated in the
Biffiriufacturer,'s 0p©cifications, wm 1.0,.'* 1,5 percent and 0.2 pereont
respeetivelsr.
fhe smplQ® wr© placed in on© ouaes, serm*mp glass
JM'S,
after
b@ing aiffcod through a 20 ffl©sh nylon screen, and ©tored at CnP* for
subsequent TBA deterainations.
2, Effect of Teraperaturer A random a&api© of fr©.sht ground rook*
fish fillet was prepared and fidbced ^with purified surfactant as
2*
prsviouslj describad.
After sifting, fisto^surfactant saaplea ware
placed in on® ounce, screw-cap,glass jars and stored at 0°, 10° and
20°?.
Sach jar contained approxiaatelsr 1*5 grass of ©aapl© and served
as an individual sample for chemical determination of ©xldative
rancidity by the ffik method.
3.
Sffect of Flttctmating; temperature«
The following experiasnt
tms used to determine the effect of fluctuatisng teaperatures on the
extent and rat© of oxidative rancidity in fresh frozen rockfish using
the accelerated procedure described.
Thirty-six, one pound cartons
of comercially packaged, fresh-frozen rockfish fillets were purchased
from a comereial packer.
Initial ?BA determinations were made and a
siioilar eassple was presented to a trained taste panel for sensory
evaluation,
sample.
the results, in each case, indicated a relatively fresh
The remaining cartons were evenly divided into four lots
(Ox, Ix, 2x, 3x).
tot 0x (control) wa© held at O**?.
Lots Ix, 2x and
3x were placed in a 35°?» cool room and held for 12 hours* during
which time, the internal temperature of all the fillets unifonuly
increased to a maxiaasa of 29%» as recorded by a electronic recording
potentiometer, using a copper-eoastantin thereioeouple.
To assure
equilibration of temperature, the saiaplee were rapidly cooled to 0*^.
and held at that temperature for 2^ hours between each fluctuation
period.
I«ot Ix ■&&& held at 0°^., lots 2x and 3x wr© returned to
the cool room and subjected to another 12 hour fluctuation in
temperature.
After equilibration at 0°?., lot 3x was given a third
and final treatment.
After this third fluctuation period, randotaly
25
selected fillets fPOSJ two- eartons por lot mrQ grotmd md Edsed raith
tfee ptu*ifi<sd surfaetaat iss a ratio of 1*3 ®s previously dssorlbed*
After sifting to establish unifoitei partiele 8!a©> eack lot ^^s ©tored
ia m ©iglit ©wio®, scrw*e©p, glasg jar and Si©3Ld at (yf. storage for
subsequent choaical determination of this accelerated autoxidation
procedure. For purposes of ooaparisoa, the raaaiaiag esa?toas ©f
fillets txm -Qmh lot t^rere etored* m gmdh, lit O^F* &iad evaluated by
fM method at the sajae storage intervale as the ground, aocolorated
f@ 4s0ure that tmifona tesperatBres esdsted ia'th® samples
during th® fluetuatioa oyol© and the eqailibratioii period, the
sasaples ware stored Indi'tfiduallir oft shelves, with free oirculation
about each- paekage,. asid tdlthout benefit ©f proteotion free msa
packaging ia a carton*
4*. Effect of Antioxidantst
Fresh silver ealaon steals
(OncorhyinchUQ. kJautch),. «ero prepared in the saae raajaner ae th© ground
rockfioh fillet^,, after carefully removing th© skin and bones*, the
saatpl© vm& divided, 100 .grasie thoroughly iixed xdth 70 sdlligraaQ of
propylene glycol to serve as the control, the other half carefully
taixed raith 70 ailligraais of a nordihydrogu&iaretie acid (IWA)
asitioxidant solution containing 10 percent MIXM, 20 percent butylated
hydro^ranisol© (BHa) and six percent citric acid; the resulting
a&sture yielding a synergiatic concentration of 0.007 percent KDQft,,
0*014 percent Wh and 0^00^2 percent citric acid on a final wight
basis.
26
k lik ratio of fiah to siirfaotant tsas uedd la this treatment*
Mixing w&s don© in a mortar^ as desorifeed previously* under standaird*
laed eonditions ©f tiae and te^erature*
The smplm were stored in on© ounee, scretJ-cap, glass $&*$ and
held at OF* for cheraical d@temination.
m M££>>im& fei: Chr^^i and sgj^iso: gmj-j&^te^
to insure the use of the freshest possibl© aaiaples, 225 pounds
of fresh rookfish fillets were acquired from a coffiaerelai packing
house in Astoria, Oregon, within ttsr© hours from the tio© they had
been removed from the fishing boat.
The samples were less than four
days old and had all been caught Just off the Washington coast at
approximately k60 30' HL (along Loran Idne 3700).
The fish had been
well iced and tsere in excellent condition at the time of purchase*
The sample consisted of a random assortment of the following species:
pinftiflM:
red rockfish
Seba§tgd|
)
)
black rockfish
)
The fillets had all received a sodium bengoats-benzolo acid dip to
retard bacterial action.
27
fh© total sasstpl© «as divldeii thrt© ^aysi-
125 founds of fillets
wer® held for organoleptie ©valuation, 30 pounds of fillets trier® held
for cheraic&i ©valuation, and th© remaining 5® |)©uads ware grouad by
p&ssing th© fillsts through a Fitspatrick Comminutiag Kachin®, Model S.
fitted tdth & mmher three sieve.
C.
lyfff^fB^ ^f, gaai5>le3t
1.
Packaging:
wesre used:
fhree representative types of packaging containers
Cl) a coiamereial carton with overwrap; (2) a foeat-seala&L©
plastic pouchj and (3), a wCn enameled tin can.
?ho ccraaereial caafton,
produced by fiberboardj Portland, Oregon tm® a 0.015 inch solid,.
bleached eulfite. with a cold wax finish; the overwrap •eras a plain, SS
bleached salfite, glasseal product of Western Saxsid© Division of
Grown Zellerbaek, Portland, Oregon,
fh© plastic pouch, 108? Durafilsi,
was produced by th© Uobectoun Company, a division of the Dow Oh^aical
Oeaapany.
this film, a tri-laainant of ^lar/Saran/polyethylene, is
v^sy. strong,, easily hand sealed and ©stable for us© la boiling water
baths.
A 211 x fyOO, "C® enameled tin can manufactured by Continental
Can Company was used as the third type of container.
Sine© none of the packages vrere vacuum packed, care was exercised
while filling the packages to eliminate any air pockets and to keep
the head ©pace at a. minifiram.
A dtmp ©pong© was used during the
sealing operation of the plastic pouches to force out as much air as
possible..
fh® grinding and packaging operation was done at the Oregon State
28
CoUdg© Seafood laboratory- ©ad tho ©atlro operation was eQii^3L@t®4
idthiia 16 honjrs froa. the time the sssaples wsr© acquired* the packaged
senaplos mt® held at oV* prior to shipsant under dry ieo eonditioKS
to Orogcm State Co|l©g®».
2. Storage J fha
SCM^ISS
wars teld at 0® t 2°$. Observations for
oheaical and soneery evalmatiosas wer© sad® on randoa ©asaplea fr©& each
packaging trea^iessi at ia©ntfo2y iatervals @v©r a niae mnth storag©
period.
1* 6h£Bioal« fh© fm oothod of ITta aad SiianSi«b©r <?1, p. 104*108)
was asad to deteiftaln® tka extent ■ of oxidativ© rancidity d©v©l©ping la'
the samples prssoatod to th& taste pm&l m troll as the saEiples
designated for eheaical ©valuation, to ascertain the possible
existsnee of a correlation bettsso's chemieal and senso^ ©valiiations,
the resalta of the eheiaieal doteraination -mr® eoapared tdth the
degr®© ©f raneiditgr in the saraples m described by th© tasters.
6olorim®trie evaluation of th© results uas aecoaplishod using a
Beefeaan .epeetrophotmieterj aodol M. th® resialts ^r© expressed as
Wk muab^r (ng. of malonaldehgfd© p©r 1000 g. of sample) by raaltiplying
the 5 r_
at 535 mj* by th© constant k6 (55. p. 9-12).
JL Cm,
2.
Sensory s
fho sensory evaluation© wore made % a train&d,
staff panel of Ik tasters ®ho laer© present throughout th® test,
fhe
samples to b© evaluated were reaoved froa 0%. storage 2b hours prior
to the tiae'.of serving and thawed at refrigerator temperature*
the
29
fillets wr© cut into approximatoly 1*1/2 inch squares and prepared
tor de©p*fat frgring by first eoating them with an egg batter followd
by a coating, of cracker crambo.
?h© samples were deep-fat ffried in.
Vreasa, a h^drogenated vegetable oil shortening mamifaetwed by Sx4ft
and Coapany.
Experiuientation indicated that a well cooked sample
resulted after ccoking for three sdnutes at 350%.
Bach packaging
treatment me statistically evaluated, as indicated below, on the
basis of iw replications.
these replications war© presented to the
panel on the first Tuesday and Thursday of each month throughout the
storage period*
fh@ ssaiples were served in randomly coded paper cups
and tasting was carried out In individual booths designed for that
purpose.
So seasoning was added to any sample.
A facsimile of the evaluation sheet used for taste panel judge*
nents is shown on page 30,
A seven-point line ballot xras used for
scoring rancidity and tenderness tdth "l" being extrestely tough and
©xtremely rancid, and "T9 being very tender and no rancidity.
Samples were also scored on a seven-point hedonic preference scale
tdth al* as very undesirable and "7" as very desirable.
the judgeaents of the tasters were st&tistiealSy evaluated by
analysis of variance (40, p. 309*324) and the means plotted against
storage time.
It was understood that a taste panel of fourteen judges
would not give a preference score indicative of a consumer population,
but emphasis was placed on using the earn© judges throughout the test
and therefore the large student panel could not be utilized.
3©
faesiaile of th& S^alufiitiea Shost Used for fast© Panel Jadfomoss^s.
Oat©:
%sm
SB At 0 09
HMfSDVSt
<tanir,CT-.ii-<!
Veasy
lorn
Desirable
Hoderate^r
fender
w
Very
Slight
Slightly
fender
_ Slight
Slighter
„,,„ Moderately
Tough
Hoderat@ly
-l_
fough
"■
UlMmpi
fl®d©s"at0ly
Uesirable
_ Slightly
Desirable
featral
Slightly
Undesirable
EtEtarenQ^
ModeratoJy
Undesirable
Extreme
Undesirable
31
BB0»LfS Mm DISCUSSIOil
I.
Apcelerated Oxidation Hethod
A. Efffecft @£ fti
T!ao oatalytic effect of iron amd iron containing ooiapowads on
oxidative reactions has te^n ©howi by a, nwb©r of investigators (8,
p. 27-31; 3^, p. ISkA&t V6, p. 2^0«2^2).
fh© relstionsfeip between
the removal of iron by purification and th® degre© of eat&ly&ed
awtoxidation is sho^n in figisr© 1.
As indicated by TM a©thod (71,
p. l6&*l©B)t purification of th© aurfaetant by reitoval of W8k§&
catalysts (expressed as ferric oxide) frsatly daoroases th© rat© of
autoxidation of th© froesea seafood semplo.
&.& stated previously,
Celit© %$ and Celit© Analytical filt©r**Aid contain 1.0 * 1.5 percent
and 0.2 percent ferric oxide respectively.
The effect of this
reduction in percent ferric oxide was drmmtlcally illustrated in
Figure 1 where the IM nuuiber for C©lit© 319 at th© end of 10 days
atorag©. was apprce&H&tely 20 tlxses greater than that of th© Celit®
Analytical Filter-Aid;
fhat th© rat© of autoxidation can be decreased
even more was shown by plotting th© Wk nuab©rs resulting £roa
cheBieal analysis of the acid treated Celit© Analytical Filter^ld
and eoiBparing thea sith the untreated surfactant.
It is believed that this procedure could b© used t© st-ady the
catalytic oxidation aetion of raetals, the pro^oxidant effect of
32
FIGURE I.
THE EFFECT OF PURIFICATION OF SURFACTANT ON THE ACCELERATED
AUTOXIDATION
OF FROZEN
ROCKFISH.
C.
CELITE 319
A. CELITE ANALYTICAL FILTER-AID
P.
PURIFIED ANALYTICAL FLTER-AID
—A
25
nf5 "
"0
_4>
.0
10
0
0
20
STORAGE
30
TIME - DAYS, 0* F.
40
33
iumoglobln, ®M the fmetloa o£ sB©tal daactivators or syaergists la
fh© ^s© of low t©Mp®ratiiir© storage Sias h®m rocoafiaBdsd by
teohaol^gists for th® ®aint©aa«o© of good quality iss frosen ©©afoods.
flaoy aphasia©*! the teportaaee of storiing fisfesry prodtest© at §%* or
below md deaoaatratQd thai th© .a®0©a©ity for lot? t^ap^ratwr^ storisg©
iner©as€33 as th© lessgth of th© storage period ma inoroaseei (67> p. 9^).
fb& ©ffe©t of storag®. tea^afatur© oa th© rat© of amtojddatiOB of a,
fieh^gmrfaetaat «ixtar© aa shorn in Figisjr© 2 iadieai©© that the rat©
of autoKldatloB is a|»^rosiinat@2^ doiabled for what mvld b@ aa 3,8*^.
rise in t^apiaratiar®. this tim- also noted .by .lea (36, p* 138) ^^so
further states that th© effect of teaparaiurQ on the ©aidatlve p@aeti©a
is of th© ©ri^r ms»ally found for eh«ie@l proeessesi
It would sseffl a|>par©at, that by this aee©larat©d ppoe®iur©>. it
may b© posslbl© to avaliiiat© th© irarioBs storage varissbles in a aiatier
of dajrs. ?r$irio»s ©spariene© has show that wsaks or ©v®» aoath©
would b© neeessaty to • aehiev© a siiailar stats of ©xidatioa tdth «holQ.
or @T©a ground fillets; the advantages of evaluating aaiiosidaate or
ethar additiTOs by this method ar@ equally apparent •
0. Bffqc^ of Flu^u^tjn^ f^per^uyep,
9n th^.A^o^da.tiQn ^,^^09^.
Fottiuger (46, p. 19-27) eaphaiBized the need for keeping froaea
seafood© at & eomatant t^perature. He found appreciisbl© differeftces,
3k
FIGURE 2. THE EFFECT OF STORAGE TEMPERATURE ON
ACCELERATED AUTOXIDATION OF FROZEN FISH.
5
STORAGE
10
TIME-DAYS
35
due to storage temperatures, in the storage life of the samples*
The
samples held at 0^. or below had two to three tisaes the storage life
of the samples held at 15°^.
He also pointed out that rancidity and
discoloration occurred much sooner and progressed more rapidly with
samples that were loosely trapped and where care had not been
exercised in eliminating the air pockets.
In referring to the samples
held under fluctuating storage temperature conditions, Pottinger
(k6, p. 19) stated that based on palatability scores, volatile acid
slumbers, "drip0 deterrainatiens and visual exaraination, there was no
adverse effect on the quality of frozen fish in the temperature
range covered {0° to * 10°^* and 0° to 150Z).
He concluded that the
average temperature enoountered during th© fluctuation periods would
seen t© be th© deciding factor on the quality of the froisen fillets.
Th© effect of fluctuations in storage temperature on the rate of
autoxidation of frozen seafood was shotan in Figure 3»
The degree of
oxidative rancidity increases rapidly with an increase in the
number of fluctuation periods.
This rapid increase in the rate of
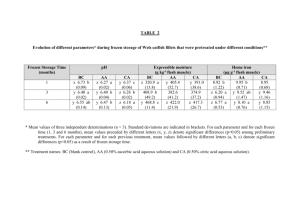
autoxidation is in direct contrast \ritb results shown in Table 1
«rhere very little change is noted in TM numbers of ceamercial
packaged rockfish fillets Srcm the same lot© evaluated over the same
period of time.
The sanples described (Figure 3) tfere ground and
mixed with purified surfactant prior to storage at 0oP. whereas the
samples referred to in Table 1 were stored as oomeroially packaged
fillets.
36
FIGURE 3.
THE INFLUENCE OF TEMPERATURE FLUCTUATIONS
ON THE ACCELERATED AUTOXIDATION OF
FROZEN ROCKFISH.
3x.
3 FLUCTUATION PERIODS
2x.
2 FLUCTUATION PERIODS
lx.
I FLUCTUATION PERIOD
Ox.
5
STORAGE
CONTROL
10
TIME- DAYS, 0° F.
15
37
fable 1
ISFFECf OF PRE^STOMQ^ TEMPBMfURB FIAJCfUASI0I0
01 tm Wm Of AUfOXlMTlOI -OF CQMMSXMX
PACKAOED ROCXfXSS ftUMS ■
Kuaber of
femperature
Fluctuations
Msm
20
0
1
2
1*1%
3
1.24
1.13
1.20
1.38
1.21
1.20
1.43
1.56
\
(Identifiable to lots Ojt, 1*, Zx,> 3x).
The data shown in Figure 3 and fable 1 indicate that the
accelerated autoiEldatlon method could be used for rapid evaluation of
th© various processing and storage variables asaooiated with froaen
seafoods.
Ag noted in Figure 3» very definite inereasee in the rates
of autoxidation ^re shown for the samples mxed with surfactant^
whereas little or no change was shown for the untreated samples even
after 20 days of storage..
Application of the accelerated method to
frozen seafoods idould therefore perait evaluation of the product
before shipment to the retail market.
This experiment also suggests
the possible application of this accelerated autoxidation procedure
to the evaluation of processing variables in frozen vegetables and
precooked frozen foods as well.
B»
Influence of Antioxidant^ on the Aggjal&mt&d,
Autoacidation of Frozen Salmon.
The need for antioxidants to protect frozen seafoods and thereby
increase their storage stability has been the subject of considerable
research and development (k, p* 250-256; 5» p. 25^260} 51, p. 1*6;
61, p. 137«15^i ^2, p. 237-24'?).
3Cn spite of the advantages sho«n b?
the application of antioxidants to both fish and other foods, they
have not received a very enthusiastic reception by the fisheries
industry*
This lack of acceptance, in spite of the great need, jaay
be due in part t©' the difficulty of application and the costs
involved.
Sauapl© variations and lengthy storage requireraenta often
make a olear»cut evaluation of an antioxidant treatastent a forsmidable
task.
Figure k dramatically illustrates the effect of a synergistie
mixture of phenolie antioxidants added to an accelerated oxidation
reaction involving ground silver saltton steaks.
the ©ffefctiyeneQ© of the ©Oft solution in prolonging the induction
period is clearly seen.
It should also b© noted that less than 30
days is needed to evaluate the stabilizing effect of the antioxidant
instead of the many oonths usually required*
The extremely high HBk
number obtained for the rancid salmon tissue is apparently due to the
high fat content (8.7 percent) and the highly unsaturated fats vMch
are present.
39
FIGURE 4.
THE EFFECT OF AN ANTIOXIOANT
OF FROZEN SALMON.
ON
THE ACCELERATED AUTOXIOATION
800
600
tt.
Ui
m
s
3
400
<
CD
C. CONTROL
200
A.
-tfj
STORAGE
re
TIME - DAYS, O'F.
ANTIOXIOANT
kQ
II.
Ghejaieal and Sensory Ey^luftjon
M ^S P^ffka^i^ .Yye^en^
Analysis of variance (40, p. 309*32^) oalculations were raade on
the sensory evaluations of "Tenderness0, "Rancidity,, and "Overall
Desirability" and significance determined at the five percent level,.
Since this Investigation was initiated in August, no sensory evaluations
were made until after classes began and the panel was trained.
The
first scheduled taste panel session was set up to evaluate samples that
had been stored at 0°?* for four months.
Thereafter, taste panel
sessions wer© held on the first Tuesday and Thursday of each month,
with the exception of January, until the tests were ccHapleted with the
evaluation of the nine month storage sample.
Results of analysis of variance calculations for the evaluation
of ,,Rancidity,, and "Overall Desirability" indicated a significant
difference, at the five percent level, between the treatment means.
The method of least significant difference (40, p. 233-238) was used
to determine which treatments were significant.
It was shown that
the carton was significantly different from the pouch and can during
©very storage period evaluated except the first one.
This was not th®
case with "Tenderness" evaluations where no significant difference was
noted between treatments throughout the duration of the storage study.
To elucidate the above statistical analyses, the means of the
observations for each package treatment pertaining to a particular
flavor evaluation (Tables 2 ■• k) were plotted against storage time.
The erratic results (Figure 5) shown for tenderness evaluations, as
41
fable 2
TEHDBRIgSS SCORES OF fWZM
MtiWtSM TtUM$ AS AFFBCTE0 BY
Pics^cESQ fBE^rasif &m BT<mm
(U&m Flavor Sco^ei)
Vtvafamk
Months
Storage
5
$
7
8
Goaimes'eiai
Carton
Plastte
Pouch
«ln
C^n
5.19
^.5?
5.16
^.58
5.28
4..93
^.96
5.20
5.35
5.^2
5.3?
5.^6
5.03
k.m
5.41
5.23
4.93
Table 3
MICTOIfl SGOHBS OF FE02K3
EOOSKSH Fxusfs AS Armm
(Mean Flavos? Scor©)
Troateeni
Honths
Storey©
at 0oF.
4
I
?
S
9
Coam^rcial
Carton
Plastic
Powefe
tin
Qm
^.85
4,36
4.62
4.98
4.5?
4.48
5.26
5.18
5.16
5.96
5.62
5.32
5.53
5.34
5.6?
5.48
5.66
5.03
42
fablo k
JESlMBllin SCORES OF FEOmi
ROCIFISH FlUMB AS AFFECTED BY
PACKAQXBO THEktmSt AE3D SfOMQB
(Mean Flavor1 Scores)
freatmont
Months
Storage
at OPP.
4
5
6
7
8
9
Commereial
Carton
Plaetie
Pou«3h
fin
Can
4.58
4.30
4.33
4.51
4.74
4.91
4.98
5.14
5.16
5.49
5.30
5.50
3.68
$M
hM
5.46
5.05
4.32
fable 5
CHEMICAL DBTERfflmtlOM OF OXIMfOT
RMCIDm II FR0» EOCKFISH FIUSTS AS
AFFBCKD BI PACKAQIW fREAfIffiOT
Treatment
Months
Storage
at OF.
Commercial
Carton
Plastic
Pouch
fin
Can
4
5
6
7
8
9
2.21
3.08
3.86
4.76
3.26
4.00
1.33
2.42
2.29
2.70
2.12
2.07
1.24
1.77
1.98
1.58
1.85
1.78
^3
FIGURE 5. EFFECT OF PACKAGING
OF
FROZEN
CONTAINERS ON
TENDERNESS
ROCKFISH FILLETS AS INDICATED BY
SENSORY EVALUATION.
CARTON
POUCH
CAN
i
6
STORAGE
-L
7
_L
8
TIME (MONTHS), 0oF.
affected by packaging eoiat&in©?©, clearly indicates, that th© t&stera
could not consistently differentiate h@>tmm tte treatments, this
substantiates the results indicated, by m&S&sia of ^arianc®
calculations* Figure $ show© the ©ffect of packaging containers ©n
the d©v©l@paent of rancidity as evaluated by a trained taste panel.
That th© tasters consistently noted a difference between the treatiwts
Is quit© apparent*. It should also be noted that the filleis packaged
in ooHiai&reial carton wre rated aueh Immv than those paefeaged in
plastic pouches and tin cans. This would tend to indicate that
seafood® packaged in eoaaereial cartons do not have the sane protection
from atoospherie oxygen as those packaged in the peueh aM can* fhis
result tias also shoxm by the -saethod ©f least significant differeKce.
this sarae preference for fillets packaged in plastic pouches and tin
cons was shown in Figure 7 where th© tasters wsre asked to evaluate
the samples as to "Overall B®giraMlityn* Xt ms interesting to note,
in reviewing Figures. 5* 6» and 7, ^hai overall desirability is
primarily dependent upon th© evaluation ©f rsmcidiiy*
It should also
be noted that although changes in flavor scores isere detected during
the overall storage period, littl© or no difference vm ahem bettieea
the score for the fourth and ninth Eionih evaluation* this cannot fee
adequately explained except to- note that sensory evaluations are
influenced by outside factors that cannot b© controlled*
fo ascertain if a possible relationship existed bettieen sensory
and ehesaical evaluations, T&k determinations were Kade on randoffl
samples of each treatment served to the panel* the results (fable 5)
^5
FIGURE 6. EFFECT OF PACKAGING CONTAINERS ON DEGREE
OXIDATIVE RANCIDITY IN
AS INDICATED BY
FROZEN ROCKFISH
SENSORY
OF
FILLETS
EVALUATION.
CARTON
POUCH
CAN
<
2 5
—e
_L
6
STORAGE
_L
7
8
TIME (MONTHS), 0*F.
k6
FIGURE 7. EFFECT OF
PACKAGING CONTAINERS ON
DESIRABILITY
AS
OF
INDICATED BY
FROZEN
ROCKFISH
SENSORY
OVERALL
FILLETS
EVALUATION.
CARTON
POUCH
CAN
z
<
2 5
•»*'""
-.-'"0^
^
^
/
_L
6
-L
7
8
STORAGE TIME (MONTHS), 0° F
k-7
■mv® plotted against storag© tiiaa (figui'e 8), aad (slearly substantiate
th© rosialte indicated by the sonsoyy evaluations for r&neiditj.
H©w©ver, th© ch©aicial analysis indicated that the degree of rancidity
increased x-dth storage time and that this increase was greatest in
those fillets packaged in QOiaereial cartons.
Although both aethods of evaluation (Figure 9) point out that
the eoiamereial carton offers the least proteetion against atttoxidatidn
of frozen rockflsh fillets, a positive relationship eould not be shown
due to instifficieht data*
48
FIGURE
8. EFFECT OF PACKAGING
CONTAINERS
OXIDATIVE RANCIDITY IN FROZEN
AS
DETERMINED
BY
ON
ROCKFISH
CHEMICAL
/
/
\
DEGREE OF
FILLETS
EVALUATION.
\
4h
\\
v
/
\ //
/
2
3
Z
<
IS
CARTON
POUCH
CAN
8
STORAGE
TIME (MONTHS), 0oF.
^9
FIGURE 9.
A
COMPARISON
EVALUATIONS
RESULTING
TREATMENTS
..
OF
FOR
OXIDATIVE
FROM
OF
SENSORY
VARIOUS
FROZEN
or
AND
CHEMICAL
RANCIDITY
PACKAGING
ROCKFISH FILLETS.
• -o
_
3
CARTON
POUCH
CAN
X
6
_L
7
8
STORAGE TIME (MONTHS), OT
9
<
UJ
50
siiMMftflT km mmtmim
An accelerated autoxidation procedure for frozen seafoods and
fats and oils was presented.
This method depends on the use of a
purified diateaaceous silica,. Celite Anal^rtical Fiiter*Aid, as a
support for the aaterial to be tested.
The increase in surface area
that results, persaits the autoxidation reaction to proceed at an
aceelerated rate, even at sub*fr©eaing teraperaturee.
the effect of temperature on th© rate of autoxidation was
deterained a© well as the effect of fluctuating temperatures oft the
storage of frozen seafoods.
The application of this procedure to
evaluate antloxidants was demonstrated.
Eepresentative types of packaging containers wer€> used to
demonstrate the effect of packaging on the rate of oxidative rancidity
in frozen seafoods.
A possible relationship betmen chemical and
sensory evaluations was also shown.
The use of this accelerated autoxidation procedure for th©
©valuation, of processing variables pertaining to certain frozen
vegetables and precooked frozen foods was suggested.
Conclusions..
1. 'The necessity of an inert matrix as a support media for th©
proposed accelerated autoxidation procedore was shown by the cataj^tic
©ffeet of irbn (expressed as ferric oxide) on the frozen seafood
samples.
2.
An increase in storage temperature, as well &$ fluctuating
51
temperatures, is shown to greatly increase th© rate of osidative
rancidity in frozen seafoods,
the need for storing seafoods at a
constant temperature of 0%. or below was deiaonBtrated.
3,
The storage life of frozen silver salmon, as evaluated by the
USA method, was noticeably extended by the incorporation of an HB&&
antioxidant solution tdth th© ground fish flesh*
k*
the proposed accelerated autoxidation procedure pemitted the
very rapid evaluation of quality in frozen seafoods as affected by
temporature, catalysts and antioxidants.
5.
Analysis of variance calculations of taste panel evaluations
for tenderness, rancidity, and overall desirability indicated that a
significant difference existed between the treatment means for
rancidity said overall desirability but not for tenderness.
6.
The method of least significant difference indicated that
both th© plastic pouch and the tin can wre significantly different
from the commercial carton as concerns rancidity and overall desirability.
There was no significant difference between the pouch and the can.
7.
Xt was shown that sensory evaluation of frozen rockfish fillets
as to overall desirability is primarily dependent on the degree of
rancidity present in the ssjnple.
8.
the greater susceptability to autoxidatioa of frozen fish
packaged in oowaereial cartons as compared to %lar/Saran/Poly
lasdnated pouohs and uCn enameled tin cans was showa by both sensory
and chesdoal ©valuations,
fhis would tend to indicate that a possible
relationship existed between sensory and chemical evaluations for
oxidative rancidity in packaged, frozen seafoods.
52
X, ArtMr D. I&ttl©, lac. (ed). Flavor research and flavox*
accaptanc©. Urn York, Reinhold, 1958. 391 p.2w Ball, €. Olin. Food paekaging. Ins Fr9d.C.; Blanek's iaadbodk
of feed aad agrietaliure, He^ fork, Eeinhold, 1955* p* 691-732.
3.
Bat&nStatth, Si C; and T. I. Morris* (©d's). Food sciences 4
spapaslw on quality aad preservation of foods. Caabridga, 1952.
319 p.
k*
Banks, A, Bev^lopaeBt of raacidity in cold-siorag© herrings
ffoe inflmefie© of $&m aatioxlda^to; Journal of the Soienc© of
Food susd AgriCttltur^ 3:250-256. 1952;
5;
8au©rnf®tod, J. C;, B. 0. Saith and Q. F. Sieaers. Cowaereial
proeessing of froson fish t&th ascorbic acid: Food feehnology
5:25^26®. 1951^
6.
Bigg®, B* A. said J*. Ri Bryant. She thlobarblturio acid teat
for butterfat osidatloa. Oaaadian Journal of technology JXildB*
1^5. 1953.
7.
Bishov, S. J., A. S. lenick and S. B. loch; Oxidation of fat in
iaodel §yst©si@ related to dehydrated foods. Food Besearoh 2517^-182. I960.
8.
Brotmj, W. J).■ g£ ^g,. Osidative deterloratioa in fish and fishery
products. 11* Progress on studies concerning ©echanis® of
oxidation ©f oil In fish tissue. Go»M©rci&l Fisheries. Heview feehnoldgy Smpplesent 19 (5a): 27*31, 1957.
9.
Browne, Christopher ^. Facka^ingi In* Morris B. Jacobus fh©
eheaistry and technology of food and food prodwots* 2d edi
Vol. SIX. lew fork, Intersclenc®, 1951* p. 1992*2017.
10.
Bystedt, J*, %** S^nae and B. M» Am. Betersnination of trimothylaffiin© oxids in fish muscle. Journal of the Science of Food
and Agriculture 10:301*30^. 1959;
11.
Cain, E. F, gt ^ Acceptatoility of fresh and precooked radiated
seats. Food Kesearoh 23»603-6l0. 1958.
12.
Cald^sell, E. F. and B. Sregg. Application of the thiobarbiturio
add test to cereal and baked products. Food technology 95185-186.
1955.
53
13*
Calvin, Lyle D. and Lois A. Sathor* A comparison of student
preference panel® with a household consuiaer panel* Food
Technoiogy lyM^tyZ. 1959.
14.
Clausen, D. F. e|, §1.
Packaging 29:149-156.
15.
Coe, M. S. Photochimieal studies of rancidity? Factors uhich
increase the'rate of oxidation of fats and oils with special
reference to the role of light. Oil and Soap 18J2&U2W, 19^1.
16.
Coimittee on Food K©s©arch. Deterioration of fats and oils.
QHC 17*7. «lun© 19-21, 19^5. Washington, D. 0., Arn^y Sorvic®
Forces, Office of the Quartertaster General, 1945* 153 p*
17.
Covor, Sylvia. Scoring for three cosaponents of tenderness to
characterise differences aaong heefsto'ak. Food Eeaearch Zkt$6k~
573. 1959.
18.
Desrosier, W» ¥. Th© technology of food preservation*
Conn., Avi Publishing, 1959. ^1B p.
19*
Bugan, l>, E. %& ®1>, Butylated hydrosyaBaisol© as an antiexidant
for fats and food inade with fat. Food technolo^r 4?^57*.^0. 1950.
20.
Ounn, C. 0. Ghefflical agents give quality Isprovesients in
fisheries. Food feoktology 1<371*383• 19^7.
21.
Sussault, H. P. frimetl^laaine test for ©valuating th© quality
of rosefish fillets. In: Fisheries Research Board of Canada,
Atlantic Coast Stations, Halifaa, K. S. Progress Soport, Mo* $7*
Movecaber, 1957. p. 23-25.
22.
%er, W. «f. Beport on trisaethylamine In fish. Joujmal of the
Assooiatioa of Official Agricultural Chemists 42»292-294* 1959.
23.
Farber, L. and P,. A. lerke. A review of th© value of volatile
reducing substances for the chemical assessment of the freshness
of fish and fish products. Food Technology 12»677*680» 1958.
24.
Farmer, B. H. Certain fundamental concepts relating to nonpolar
saechanisms in olefinio systems. Journal of the Society of Cheiaical
Industrgr 66:86-93. 1947.
25.
Filer, 1». <J. Jr.., K. I»» Mattil md H. B. tongenecker. Changes
occuring in fat autoxidation. Oil and Soap 22tl9&#2Ql. 1945.
26.
Fridriksdottir, Sigrun. The application of th© tMobarbituri©
acid test to the determination of oxidative rancidity in frozen
herring-. Special, problem. Ithaca, Hew Xork, Cornell fnivorsity,
1953. 35 numb, leaves.
Quantitative odor measureiaent*
Soveafeor 1955.
ISodem
W^stport,
2?»
Oearhart-, W» M, and B, I, Stueksy-, A coaparisoa of eo»i©reial23r
used phsnolle aatioxidants ia edible smtesd fats. J©iamal pf
tla© hmritm Oil Ohaaists1. Society 32:38^*399* 1955*
3$, 6©arhart, W. K,^ B, I. Stuekej and <J* <J* Austin* C«paris0a ©f
sotliods for tosting th© stability of fats md oils* aM of f®od$
coBtaiaing thsffl, Joimial of the te^ricaM Oil Otaist©1 Soei©ty
3%^2?-430» 1957?
29. Sungtone, % P. An introdraetioa to the chaBistxy of fats mid
fatty acids. %m fork, »il©yr 195®* 161 p*
30.
Hllllg, F, j^ ^j Clieaical indi^^s of d©eos^@oitioa ia ^od;.
sFoiaraal of th© Assoei&tiOQ of Official AgfieuAtm*®! Oheaist©
*X*763-776* 1958:.
31.
Hillig, F.t ¥i I. Pattarsoa and Hargarst &cl©an-. Siaocinio acid
m aa indoK of deceraposition in tunav ^o^rnal of the Association
of Official Agricaltural Cteaiats y$tWMk1, 1950.
32.
Jacobs, M. B. <ed,) The chesaistrgr and technology of food and
food products. 2d ed. Mew iork, laterseieac®;, 1951* 3 troie,
33.
King, A. B., H. L. Roachsn and W. B. Xrediiu ^a accelerated
stability test using the peroxide value as aa index. Oil and
Soap l®a05-109. 1933.
34.
Slose, A. A., i. P. Mocehi and H. L. Hanson. Bse of antioxidaats
in the frozen storage of turkey* food technology 6:308-311.
1952--.
35.
8beht R* 3. ieaehanissi of fat oxidation,
30 (2)!feS*53, 68»69. i95&.
36.
I«a» C* I» Boneidltar in edible fats*
Publishing, 1939. 230 B».
37.
iaa, 0*. B» Recent developaents in the study of exidaii^
deterioration of lipids* Cheadstry m& Industry, Bee* 3* 1953.
p. 13©3^309*
38.
loa, C. H. Bancidity in
In: B. C. Bato^S&aith and
sj^o^iu®' ©a quality and
University Frees, 1952.
!&je Bakor's Pigest
Saw t&rk, Ch^aieal
edible fats aud fat containing foods*
f. ft» Morids's, Food Scieae©. A
preservation ©f foods. Carabridge,
p. 200-228.
39. &w, ^* T* and A. t. tappel* Antioxidants and synergist inhibition
©f heaatin-eatalyBed oxidativ© fat rancidity. Food f©chnolo^
105285^209* 1956.
55
40*
Li, Jeroae C. R. Introduction to statistical inference.
Arbor, ^ckigan, Bdwards, 195?. 533 £>*
kli
LuMborg, Walter ©♦ lipida. In? Morris B. <Jae©bfs fh® cheaists!^
and technology of food and food prodaets* Vol. I* lew York,
Int^rseienee, 1951* P* 104-13S*
42 *
Korris, $• Q* Rocerat studies on th® KQohanisia of fat oxidation
in its' relation to rancidity* Jouraal of Agricultural and food
Cheraistry 2J126-.132* 1954*
43*
Het»3r# J. F» and Li E#, Dugani Ji,« ^ ^w method for a©as«idng
th© davolopaent of rancidity« Food T©ctool©^ 75l91'*i94* 1953*
44*
liMcila, Olavi E„ and Boino B. Idn&o* Denaturation of ingrosin
during defrosting of frozen fish. Pood Eeaearefe 19J2O0*205*
195^*
45.
Olcott, H# S. and B, Binget. A wlgliiag aethod for moasuring the
induction period of marine and otfoor oils*' Journal of th® American
Oil Ghofflist©1 Society 35:161*162. 195B.
46.
Fottijiger, 3. E. Iffect of fluctuating tes^eratures on Quality
of froaoa fish fillets. CeamorGial Fisherias Reviow 13 (2):19-2?*
1951*
47i
Report of th® fat Stability Subcoffimittee of the Fat Analysis
Gonmittee * 1956: Active oaiygen aethod for determing fat
stability. Journal of th© Aaorican 011 Chemists * Society 34? 394398* 1957*
48.
Biamen&chneider, Bi W« Oxidativ© raacidlty and antioxidonts.
In: Fred 6* Slamk's Handbook of food and agriculture* lew lork,
Beinhold, 1955* p. 237-2?3.
k9»
Bowanf A* M. Fresh fish: fh® assessment of the freshnese of fish
by odor. Annual leport. Fishing Industry Besearoh Institute,
April-»Bee©aber 1956, Capetom, South Africa. (Abstracted in
Goiaiaereiai Fisheries Abstracts 12 (3) tl?. 1953)
50*
Schwarta* M* Q. .and Betty Kt. Watts. Application of the
thiobarbituric acid test as a quantitative measure of deterioration
in cooked oysters. Food Besoarch 22*76*32. 1957*"
51*
Scltearts, M. G* and Betty M. Matte* Ascorbic acid as an smtioxidant
for froaea oysters and effect of eepper-ehelating ability of oyster
tissue on ascorbic acid oxidation. CoHraer^ial Fisheries Beview
21 (3)51-6* 1959*
Arm
%
5%. Soagraa, Marvp It. Ghtsaieal ebang®®- ia fish actcs^rosia dariijg
tt@&ts3L6g m& storag©* fooftM&e&e-Qk 21:105-109* 1956. '
$% 8lmt?m, J. S. and A* S# 0. Shfwtitarg* Vol^til© bases as ^alit^r
indi€©Si JoTjmal ©f th©' Seieaee of food and Agrioulttisr© @82S?231. 1957*
$ki Sidmll, Q* '0. a^ ^+ ffes we!e of thi©feaAiturie a,eid as a
sasamir® of fat ojddati^B* Journal of the Amepicaa Oil Ghanidte.1
Soei^ty 31:603«$0^, l^5ifr.
55* Simifeubar, E* Oa and T* C. fu. 2-Ehi®tmrMiur!e mM method for
th© ia©a$ur©ii©at ©f ranciditj ia fistorf produets* XX.* th®
quantitative deteraination of aalonaldeh^da. Food technology
12*9*12* 1958*
56, Stasisby, M* S. Oxidativ® d®t®ri©ration in Sish and fishery
prodmets. O^aaereial fisteries Beview 19 (5&)J2^26. Jfey 195?.
57* Stansty, M. B*, S* B, P@tting©r and ®* f. Migrauchi* Sefrigeration
©f fish* X2X. Faetora to b© considered in th© freezing and eeld
storage of fisfc©r$r produets. Washington* 0* C,, IJ* S. Fish and
Uildlif© Serrtoa* 1956. 65 nmab* Imws* (fistexy leaflet ^29)
58* Sttiokey, Ben H* Inereasing th© shelf Sife of e©r©al8 viith ph©noli©
antiosidants* Food f©ehn®l0©r 9* 585-58?. 1955.
59. ?&pp®X» A. L. Expanding ia©© of antiosidants* Food Sngineeriag
26 (6)t7>.75, 13% 13^. 195^*
60 farladgiSj B. ©., Margaret T. Tounathan and Betty M« Watts.
Anti®«id!®ts in irradiated looked pork. Food feeha©!©©? 135635639. 1959*
61* Tarr, H. I. A* Control ©f rancidity in fish flesh. I* GheHiieal
aatiojcidants. Journal of th© Fish®yi©g E©soareh Board of Canada
?tl3?-.l^. 19^7*
62. Saw, H. L. A* Control of r^icidity ia fish flosh. XX. Physical
and eheaieal methods. Journal of th© Fisheries Hesearch Board
of Gasaada 7J237*24?. 19^8*
63. foMyaiaa, f., A. A. DaCosi and J* A.. Stem. A rapid vaeuu®
distillation proesdur© for th© deterainatioa of volatile acids
and volatile bas©s in fish fi©$h. Food feclmol®^ 10:61^*617.
1956.
6^* fressler, D. K. and J* MeW. Leaon. Marine products of e©JM©re©,
2d ed. lew fork, Reinhold, 1951. 762 p.
57
65.'
dressier, B.- I. and C. F. Evera,; ?he freestog ps«aser^sition of
foods. 3d ©d* Westport,: Conn.j A^i Fublishing»: 1957* Vol. II.
66*
Wri» t. The.mechanism.of th© ©siilatioB of liaolole acid with
partio«lar r©f©r«iBo© to aetal eatalysis* la; H. M.; Sinelair*©
Basentlal fatty aeids. W©w fork, Academic»' 1958. 'p. 30-35.
67;
II.., S, Bureau of Coamorcial fisheries* lUsu storage tcstperaturep
help maintain quality; Coram©reial ^ieherios B©iri©w %% i$)i$h*
1959;
68, Watts* Bstty Ji Qj&dativ© rancidity aM discoloration In saaats*
Mimnets) in Food l©s©are!i 5!l»52* 195^.
69;
Winter* J. D; j& ll* ^© effaet of fluctuating teapcratwes oia
the quality of stored frozen foods* Food Techjiolo^ 6:311*313•
1952.
70.
Young, 0, C. Quality of fresh and froscn fish and facilities for
freezing, storing.and transporting fiahery products., food
Teetmolo©' 4;^7*^50. 1950,
71;
^u, ?. G. sad ft. 0, Sinnhuaiber. 2*5hi©barbiturie acid method for
th© saeasuroaent of rancidity in fishery products. Food Technolojsy
11:10^-108. 1957.