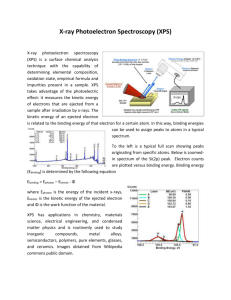
X-ray photoelectron spectroscopy - An introduction Spyros Diplas
advertisement

X-ray photoelectron spectroscopy - An
introduction
Spyros Diplas
SINTEF Materials & Chemistry, Sector of Materials and
Nanotechnology, Department of Materials Physics-Oslo
& Centre of Materials Science and Nanotechnology,
Department of Chemistry, UiO
spyros.diplas@sintef.no
spyridon.diplas@smn.uio.no
October 2015
1
Material Characterisation Methods
October 2015
2
XPS-Basic Principle
Photoelectron
Auger electron
Vacuum
Fermi
valence band
2p1/2, 2p3/2
L23
2s
L1
Internal transition
hν
(irradiative)
1s
K
Ekin = hν – EB -
Excitation
EKL2,3L2,3(Z) = EK(Z) – [EL2,3(Z) + EL2,3(Z + 1)]
October 2015
De-excitation
3
Auger electron vs x-ray emission yield
1.0
Auger Electron Emission
Probability
0.8
0.6
0.4
0.2
X-ray Photon Emission
0
5 10 15 20 25 30 35 40 Atomic Number
B Ne P Ca Mn Zn Br Zr
Elemental Symbol
October 2015
4
XPS spectrum ITO
Photoelectron peaks
x 104
80
Auger peaks
Sn 3p
In 3p
In 3s
In 3s
70
60
Sn 3d
O 1s In 3d
CPS
50
40
Sn MNN
In MNN
In/Sn 4s
30
O KLL
20
In/Sn 4p
C 1s
10
1200
1000
800
600
400
Binding Energy (eV)
200
0
October 2015
5
Peak width (ΔE)
ΔE = (ΔEn2 + ΔEp2 + ΔEa2)1/2
Gaussian broadening:
Natural width
Analyser contribution
X-ray source contribution
-Instrumental:
There is no perfectly resolving spectrometer nor a perfectly monochromatic X-ray source.
-Sample
For semiconductor surfaces in particular, variations in the defect density across the surface will lead to
variations in the band bending and, thus, the work function will vary from point to point. This variation in
surface potential produces a broadening of the XPS
peaks.
-Excitation process such as the shake-up/shake-off processes or vibrational broadening.
Lorentzian broadening.
The core-hole that the incident photon creates has a particular lifetime (τ) which is dependent on how quickly
the hole is filled by an electron from another shell. From Heisenberg’s uncertainty principle, the finite
lifetime will produce a broadening of the peak.
Γ=h/τ
Intrinsic width of the same energy level should increase with increasing atomic number
October 2015
6
Examples of XPS spectrometers
October 2015
7
Schematic of an XPS spectrometer
Hemispherical
electrodes
Electron energy
analyser
Slit
Slit
X-ray source
Electrostatic
electron lens
Al
Photon
Electron
detector
eSample
Number of emitted electrons measured
as function of their kinetic energy
October 2015
8
Instrument: Kratos Axis UltraDLD at MiNaLab
Analyser
e-
Detector
e-
Monochromator
X-ray source
X-ray source
Sample
October 2015
9
The new XPS instrument-Theta Probe
Spectroscopy
Source-defined small area XPS
15 µm to 400 µm
Snapshot spectrum acquisition
Up to 112 channels
Faster serial mapping
Faster profiling
Unique parallel ARXPS with up to 96 channels
Large samples (70 mm x 70 mm x 25 mm)
Sputter profiles
Mapping possible up to full size of sample
holder
ISS included
Target applications
•Thickness measurements
•Surface modification, plasma & chemical
•Self assembly
•Nanotechnology
•Ultra thin film technologies
•Shallow interfaces
October 2015
10
Sample requirements
Has to withstand high vacuum (≤ 10-7 Torr).
Has to withstand irradiation by X-rays
Sample surface must be clean!
Reasonably sized.
October 2015
11
XPS Depth of Analysis
The probability that a
photoelectron will escape from
the sample without losing
energy is regulated by the BeerLambert law:
Where λe is the photoelectron
inelastic mean free path
Attenuation length (λ) ≈0.9 IMFP
IMFP: The average distance an electron with a given energy travels between
successive inelastic collisions
October 2015
12
Features of the XPS spectrum
Primary structure
-
Core level photoelectron peaks (atom excitation)
Valence band spectra
CCC, CCV, CVV Auger peaks (atom de-excitation)
Secondary structure
-
X-ray satellites and ghosts
Shake up and shake off satellites
Plasmon loss features
Background (slope)
October 2015
13
Quantification
Unlike AES, SIMS, EDX, WDX there are little in the way of matrix effects to worry
about in XPS. We can use either theoretical or empirical cross sections, corrected for
transmission function of the analyser. In principle the following equation can be used:
I=JρσKλ
I is the electron intensity
J is the photon flux,
ρ is the concentration of the atom or ion in the solid,
σ s is the cross-section for photoelectron production (which depends on the element and
energy being considered),
K is a term which covers instrumental factors,
λ is the electron attenuation length.
In practice atomic sensitivity factors (F) are often used:
[A] atomic % = {(IA/FA)/Σ(I/F)}
Various compilations are available.
October 2015
14
Chemical shift
ΔE(i) = kΔq + ΔVM – ΔR
Initial state contribution
final state contribution
Δq: changes in valence charge
ΔVM : Coulomb interaction between the
photoelectron (i) and the surrounding
charged atoms.
ΔR: relaxation energy change arising from
the response of the atomic environment
(local electronic structure) to the screening
of the core hole
.
October 2015
15
Chemical shift - Growth of ITO on p c-Si
In oxide
Intensity arbitrary units
Si
In3d
Si2p
Si 2p
In 3d
Sn3d
Sn 3d
5/2
3/2
Sn oxide
5/2
3/2
In
Sn
3.0 nm
3.0 nm
SiOx
3.0
3.0
nm nm
1.5
1.5
nm nm
1.5 1.5
nmnm
1.5 nm
1.5 nm
0.50.5
nmnm
0.5 nm
oC
BHF 15 sec
BHF +
15 500
sec + 500°C
104
102
0.5 nm
0.5 nm
0.5 nm
100
98
456
454
452
450
448
446
444
442
440
Binding Energy (eV)
October 2015
500
495
490
485
480
Chemical shift
October 2015
17
Shake-up satellites in Cu 2p
2p3/2
2p1/2
Cu
CuO
Shake-up
satellites
CuSO4
970
960
950
940
930
Binding energy (eV)
October 2015
18
Plasmons
They describe the interaction (inelastic scattering) of the PE with the plasma oscillation of the outer
shell (valence band) electrons
Plasmons in their quantum mechanical description are pseudoparticles with energy E p=hω
ω = (ne2/ε0m)1/2/2π n =valence electron density,
e, m electron charge and mass
ε0=dielectric constant of vacuum
Pure elements
Mo-Si-Al
Compound
October 2015
19
Peak asymmetry
Zn
Arbitrary Units
ZnO
1055 1050 1045 1040 1035 1030 1025 1020 1015 1010
Binding Energy (eV)
ZnO
Arbitrary Units
Zn
16
14
12
10
8
6
4
Binding Energy (eV)
2
0
-2
Peak asymmetry in metals caused by small energy electron-hole excitations
near EF of metal
October 2015
20
Depth profile with ion sputtering
SnO2
Sn
Use of an ion gun to erode the sample surface and re-analyse
Enables layered structures to be investigated
Investigations of interfaces
Depth resolution improved by:
Low beam energies
Small ion beam sizes
Sample rotation
Depth
500
496
492
488
484
480
October 2015
21
Probing different depths by choosing different areas
in the XPS spectrum
Ge(0)
Ge3d
Ge(IV)
GeLMM
Ge2p3/2
Ge(0)
Ge(IV)
Ge(IV)
Ge(0)
Ge(0)
8.0
eV
Ge(IV)
8.2 eV
4.0 eV
4.0 eV
Ge3d: EK = 1453 eV
Ge2p: EK = 264eV
l = 2.8 nm
l =0.8 nm
GeLMM: EK = 1140 -1180 eV
October 2015
22
Angle Resolved XPS (ARXPS)
for non-destructive depth profileOH
I (d) = Io* exp(-d/λ)
oxide
RT
I(d) = Io*exp(-d/λcos θ)
surface
Arbitrary Units
θ
Film
AR
Substrate
bulk
536
λ=attenuation length
534
532
530
528
Binding Energy (eV)
526
524
(λ ≈0.9 IMFP)
λ=538αA/EA2 +0.41αA(αA EA)0.5
(αA3 volume of atom, EA electron energy)
October 2015
23
XPS-Check list
Depth of analysis ~ 5nm
All elements except H and He
Readily quantified (limit ca. 0.1 at%)
All materials (vacuum compatible)
Chemical/electronic state information
-Identification of chemical states
-Reflection of electronic changes to the atomic potential
Compositional depth profiling by
-ARXPS (ultra thin film <10 nm),
-change of the excitation energy
-choose of different spectral areas
-sputtering
Ultra thin film thickness measurement
Analysis area mm2 to 10 micrometres
October 2015
24
Interfacial studies of Al2O3 deposited on 4H-SiC(0001)
Avice, Diplas, Thøgersen, Christensen, Grossner, Svensson, Nilsen, Fjellvåg, Watts
Appl. Physics Letters, 2007;91, 52907, Surface & Interface Analysis,2008;40,822
Si4+
d=λSi cosθ ln(1+R/R∞)
d: SiOx film thickness
λSi:inelastic mean free path for Si,
Θ: the angle of emission,
R: the Si 2p intensity ratios ISiox/ISiC,
R∞ the Si 2p intensity ratios I∞Siox/I∞SiC where I∞ is the
intensity from an infinitely thick substrate.
R∞=(σSi,SiO2 . λSi,SiO2) / (σSi,Si . λSi,Si)
Si3+
SiC/Si2+
Al 2p plasmon contribution
Si+
Si0
Ar 1273 K 60 mins
Ar 1273 K 30 mins
Ar 1273 K 15 mins,
N2/H2 873 K 30 mins
where σSi,SiO2 and λSi,SiO2 are the number of Si atoms per
SiO2 unit volume and the inelastic mean free path
respectively
The σSi,SiO2 / σSi,Si ratio is given by
σSi,SiO2 / (σSi,Si = (DSiO2 . FSi) / DSi . FSiO2
where D is the density of the material and F the formula
weight.
N2/H2 873 K 15 mins
as-grown
For the calculations we also assumed that the Si 2p
photoelectrons from both SiC and Si oxide film will be
attenuated by the same amount as they travel through the
Al2O3 film therefore, their intensity ratio will reflect the
attenuation of the Si 2p electrons coming from the SiC
through the Si oxide film.
Al2O3
SiOx
SiC
From XPS
d= 1nm at RT, d=3nm at 1273 K
October 2015
25
XPS on ITO e-beam deposited prior and after annealing
(SINTEF SEP 09)
Sn 3d
In 3d
Air annealed 300 C
e-beam deposited
Valence band
O 1s
Air annealed 300 C
e-beam deposited
October 2015
26
Nitrogen in ZnO
NO2
N-C=O
or
N
N-C
outermost
Arbitrary Units
Arbitrary Units
AG
200 sec sputtering
outermost
900 oC
200 sec sputtering
410 408 406 404 402 400 398 396 394 392 390 388
Binding Energy (eV)
414 412 410 408 406 404 402 400 398 396 394
Binding Energy (eV)
October 2015
27
Band bending in ZnO
R. Schifano, E. V. Monakhov, B. G. Svensson, and S. Diplas, 2009, Appl. Phys. Lett. 94, 132101
Zn 2p - O 1s energy difference
Arbitrary Units
Zn 2p - O 1s energy difference (eV)
491,6
491,4
491,2
491
490,8
490,6
LR4 Zn2p-O1s
LR3 Zn2p-O1s
490,4
490,2
490
1050 1045
0
1035 1030 1025 1020 1015
Binding Energy (eV)
20
40
60
80
100
Etching time (sec)
Zn 2p-LM45M45 Auger parameter comparison
Arbitrary Units
Zn 2p-LM45M45 Auger parameter (eV)
2011
2010,5
2010
2009,5
LR4 2p-LM45M45
LR3 2p-LM45M45
2009
2008,5
0
980 982 984 986 988 990 992 994 996 998 1000
Kinetic Energy (eV)
20
40
60
80
100
Etching time (sec)
October 2015
28
CIGS solar cell
Introduction
CIGS solar cell
• Energy/environmental application
• Solar cells based on Cu(In, Ga)Se2 (CIGS)
• Thin-film stack on glass
• Mo and Zn oxide layer form electrical contacts
• p-type CIGS film (sunlight absorber) and n-type
CdS film form p-n junction
• Excellent efficiency
• Low cost compared to thicker silicon-based solar cells
3SEM
of a Cu(In,Ga)Se2 solar cell (cross-section)
and its mode of operation
• Practical problem
• Controlling film composition and interfacial chemistry
between layers (affects electrical properties)
• XPS solution
• XPS sputter depth profiling
Elemental and composition information as a function of
depth
Identify chemical gradients within layers
Investigate chemistry at layer interfaces
Acknowledgement: Thermo Electron Corporation
October 2015
29
Depth profile of CIFS film stack
CIGS solar cell
CIGS solar cell
• Depth profile of CIGS film stack
• Demonstrates standardless quantification of XPS
Mo
• Excellent quantification agreement between XPS and
Rutherford BackScattering (RBS)
• Both techniques show cross-over of In and Ga close to
1.6mm depth
• XPS tool is able to analyze product solar cell device
Se
Zn
O
Cu
CdS
C
In
Ga
Rutherford BackScatter profile of CIGS film stack
Sputter depth profile of CIGS film stack
Acknowledgement: Thermo Electron Corporation
October 2015
30
Interfaces in Solar cells
October 2015
31
Interface between p-Si/ZnO: Si HF
with and without Ar etched (SINTEF SEP 09)
No Ar etching
Ar etching
Si Zn mixed oxide
SiO2
Si
Depos at RT + Ar etching
Depos at 500 C + Ar etching
Depos at 500 C
RT depos
RT depos + 300 C annealing
October 2015
32
Elemental distribution and oxygen deficiency of magnetron sputtered ITO films
A. Thøgersen, M.Rein, E. Monakhov, J. Mayandi, S. Diplas
JOURNAL OF APPLIED PHYSICS 109, 113532 (2011)
October 2015
33
Initial stages of ITO/Si interface formation studied with XPS and DFT
O.M. Løvvik, S. Diplas, A Romanyuk, A. Ulyashin, JOURNAL OF APPLIED PHYSICS 115, 083705 (2014)
• Presence of pure In and Sn, as well as Si bonded to oxygen at the ITO/Si interface were
observed.
• The experimental observations were compared with several atomistic models of ITO/Si
Interfaces.
• These results support and provide an explanation for the creation of metallic In and Sn along
with the growth of SiOx at the ITO/Si interface.
October 2015
34
Theoretical efficiency of Cu2O/ZnO based
solar cell:[1-3]
~10-20 %
Highest experimental efficiency reported:
~ 2 % without buffer layer [2]
~ 1-4% with buffer layer [4,5]
[1] S. Jeong et al. , Electrochim. Acta (2008), 53, 2226 .
[2] A. Mittiga et al. Appl. Phys. Lett. (2006), 88, 163502 .
[3] T. Gershon , et al., Sol. Energy Mater. Sol., C. (2012), 96,48
[4] Z. Duan et al. Solar Energy Materials & Solar Cells 96
(2012)
[5]T. Minami et al., Appl. Phys. Exp.4, 062301(2011)
October 2015
35
Total transmittance (%)
100
80
Very good optical properties
Optical band gap close to the
60
Cu2O on glass
o
40
deposited at 400 C
Cu2O on quartz
20
deposited at 600 C
quartz
glass
expected value of ~2.1-2.3 eV
o
0
500
1000
1500
2000
2500
Wavelength (nm)
(a)
RA=0,4
104
mm
RC=0.15 mm
RB=0.25 mm
RB=0,25 mm
RC=0,15 mm
Current Density (mA/cm2)
Current Density J (mA/cm2)
103
(b)
102
101
0
10
-1
10
-2
10
103
102
101
100
10-1
10-2
10-3
-1,0
-0,5
0,0
Bias Voltage (V)
0,5
1,0
10-3
-1,0
-0,5
0,0
0,5
1,0
Bias Voltage (V)
Current Density-Voltage (J-V) curves for the p-n junction (reactive sputtered
films on Zn polar substrate) measured from biased voltage V=-1 to 1V, with
circular diodes (a) before chemical etching and (b) after etching.
October 2015
Mittiga et al. 2006, Applied Physics Letters 88,
163502 (2006);
Samples Investigated with XPS
Top film nominal thicknesses: 1, 3, 5, 10 nm
October 2015
37
AZO/Cu2O
October 2015
AZO/CuO
38
STEM HHADF and FFT
STEM-HAADF imaging of Cu2O/ZnO cross-section interface. (A) The
overview STEM HAADF image of the Cu2O film grown on O-polar ZnO
substrate. (B) The HRSTEM HAADF image of the boxed region indicated
in (A). The different layers in the cross-section are marked by false
coloring. (C) and (D) The FFT patterns from the top and the interfacial
layer in (B) respectively were identified as Cu2O and CuO phases.
October 2015
39