Lab Safety Notes
advertisement

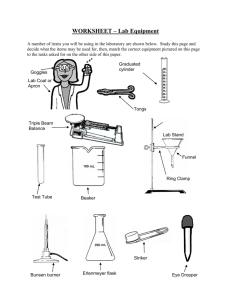
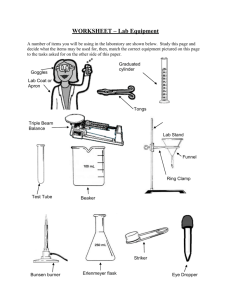
Lab Safety Notes PREPARING FOR LABORATORY WORK Never perform unauthorized experiments. Know how to use the safety shower, eye wash, fire blanket and first aid kit & bucket of sand. Do not work in the lab without your teacher in the room. DRESS FOR LABORATORY WORK Tie back long hair. Do not wear loose sleeves as they tend to get in the way. Do not wear open shoes on a lab day. Wear lab aprons during all laboratory sessions. Wear safety goggles during all laboratory sessions BUNSEN BURNER SAFETY: A Bunsen burner flame is very hot. The center of the flame will be over 1000 degrees Celsius. Treat burner flames with respect. Always use the main gas shut off valve on the desk to shut off a Bunsen burner. The valve at the base of the burner is only used to control the flame. If your hair or clothing catches on fire stop drop and roll. If someone else's hair or clothing catches on fire, wrap them with a fire blanket to smother the fire. Do not let them run. AVOID HAZARDS Use caution when handling hot glassware. When diluting acid, always add acid slowly to water. Never add water to acid. Keep caps on reagent bottles. Never switch caps. ANIMAL SAFETY Do not cause pain, discomfort , or injury to an animal Wash hands after handling animals SAFETY WITH CHEMICALS AND DISSECTING SPECIMENS: Do not touch or taste any chemical unless specifically instructed to do so. Read chemical labels more than once before using the contents - it is easy to confuse chemicals. When working with chemicals or dissections, keep your hands away from your face. The skin on your face is much more sensitive to irritation than your hands. To smell something, hold it away from your nose and wave your hand over it towards your nose. You may pass out or inhale dangerous gases is you just stick your nose over the container and breathe in. Flush any chemical spill on your skin with plenty of water. The rule of thumb is 15 minutes. When heating anything in a test tube, point the mouth of the test tube towards a wall, away from people. Do not lay the glass stopper from a reagent bottle on the lab table. Hold the handle of the stopper between two fingers while you pour from the bottle. When mixing acids and water, pour the acid into the water. Remember, AnW (root beer) Wash your hands when you are finished with the lab. SAFETY WITH GLASSWARE & BROKEN GLASS Never use chipped or broken glass. If you notice chipped or cracked glassware during a lab please report it to the instructor to be replaced. Broken glass should never be handled with your hands. Use a dustpan and broom to sweep up broken glass. Small pieces can be wiped up using a wet paper towel. Broken glass should be placed in the proper container - either a can for broken glass or a sharps container. Always have the instructor clean up a broken mercury thermometer. Mercury is a poisonous substance and should not be handled. VHHS no longer has any mercury thermometers. Broken glass that has contacted blood must be disposed of in a sharps container. The plastic cylinder guard on a graduated cylinder is to protect the cylinder from breaking if it should tip over. The cylinder guard is not made to slide up and down for measuring. A graduated cylinder should be laid down when it is empty - if it is laying down it can't be tipped over. Remove glass tubing and funnels from stoppers as soon as your lab is finished. If you do not they will become stuck in the stopper. BLOODBOURNE PATHOGEN SAFETY Wear latex gloves anytime you assist someone who is or has been bleeding or vomiting. Clean the entire area (desktop, floor, etc.) with a disinfectant when you are finished caring for the person. Remove your latex gloves by pulling them off inside out. Avoid contact with the outside of the gloves. All paper towels, tissues, latex gloves and other materials used to clean up blood and other possible infectious materials should be disposed of in the proper biohazardous waste bag. Standard Biohazardous waste bags are red with the biohazardous waste symbol on them. If one is not available, use a regular plastic garbage bag and attach a biohazardous waste sticker to the bag. Wash your hands thoroughly with a disinfectant when you are finished. CLEAN UP Consult teacher for proper disposal of chemicals. Wash hands thoroughly, following experiments. IN CASE OF ACCIDENT Report all accidents and spills immediately. Place broken glass in designated containers. Wash all chemicals from your skin immediately with plenty of running water. If chemicals get in your eyes, wash them for at least 15 minutes in an eyewash. Laboratory Procedures Notes Measuring Volume The Meniscus When water is placed in a glass or plastic container the surface takes on a curved shape. This curve is known as a meniscus. Volumetric glassware is calibrated such that reading the bottom of the meniscus, when it is viewed at eye level, will give accurate results. Viewing the meniscus at any other angle will give inaccurate results. Erlenmeyer Flasks and Beakers Erlenmeyer flasks and beakers are used for mixing, transporting, and reacting, but not for accurate measurements. The volumes stamped on the sides are approximate and accurate to within about 5%. Graduated Cylinders Graduated cylinders are useful for measuring liquid volumes to within about 1%. They are for general purpose use, but not for quantitative analysis. If greater accuracy is needed, use a pipet or volumetric flask. Some graduated cylinders are marked according to how much liquid they contain; others are marked according to how much liquid has been removed (see examples below). amount in container amount removed Measuring Mass Mass is the amount of matter in an object. There are different kinds of balances used to measure mass. Be sure you understand how your balance works. Some balances give a single reading. Others give two or more readings that you have to add together. For example, look at the triple-beam balance below. Notice that the middle beam measures the largest amounts. To read the mass of an object, find and record the masses shown on each of the beams. Then add the readings. Using the example above, 200 g + 70 g + 6.5 g = 276.5 g Measuring mass with an electronic balance: The electronic balance has many advantages over other types of balance. The most obvious is the ease with which a measurement is obtained. All that is needed is to place an object on the balance pan and the measurement can be read on the display to hundredths of a gram. A second advantage, using the Zero button on the front of the balance, is less recognized by beginning science students. Because one must never place a chemical directly on the balance pan, some container must be used. Place the container on the balance and the mass of the container will be displayed. By pressing the Zero button at this point, the balance will reset to zero and ignore the mass of the container. You may now place the substance to be weighed into the container and the balance will show only the mass of the substance. This saves calculation time and effort. However, when the container is removed from the balance, the display will go into negative numbers until the Zero button is pressed again. Most electronic balances also have a Unit button on the front. Pressing this button will change the units being measured. Since we have very few times when we need something other than metric units, you should not have to change the mode on the balance. Because of the Unit and Zero buttons, there are two things you must always do before placing objects onto an electric balance to be measured: 1. See that the display is reading 0.00 2. See that the unit sign in the upper right of the display shows g When finished with the electronic balance, press the On/Off button and hold it down until the display shows OFF. Measuring Temperature Another objective for this lesson is to use the Celsius temperature scale to measure the temperature of fluids. Liquids and gases are both fluids. The laboratory thermometers we have are calibrated in degrees Celsius rather than degrees Fahrenheit. The Celsius scale used to be called centigrade and is often still called that. It was called centigrade because the difference between the freezing temperature of water and the boiling temperature of water is divided into 100 degrees. 0oC is defined as the temperature at which water freezes and melts. 100oC is defined as the temperature at which water boils (using normal pressure). It is the scale commonly used in scientific work and in everyday use in most countries. Scientific thermometers do not have to be shaken down. Quite a few laboratory thermometers have been broken by trying to shake them down next to a table. It is best to hold a thermometer while using it instead of leaning it against the container or other object. The container it is in may or may not be able to support it. All you have to do is put the thermometer in the liquid you are measuring and watch the red alcohol level change. When it stops changing, read the red alcohol level. That is your measurement. You don't have to wait a prescribed length of time, just let it come to a rest. You may have to turn the thermometer to see the numbers and line up the mercury or alcohol with the scale. When you read thermometers, you should estimate between the lines. Try to mentally divide the sections into 10 equal parts in order to read to a tenth of a degree. Measuring Length In order to correctly measure the length of an object, make sure that one end of the object being measured is EXACTLY lined up with the zero end of your metric ruler. Then make sure that your eye is lined up with the other end of the object you are measuring and take a measurement based on EXACTLY where the object ENDS on the scale of the metric ruler. Using a laboratory burner: Many chemistry experiments require something to be heated. This is done with one of several types of laboratory burners. The lab burners use natural gas delivered through the gas outlets at student lab stations. Before attempting to light any lab burner, check to see that the jet hole between the base and the burner tube is free of obstruction. If chemicals have covered this jet, the burner will not operate properly. After attaching the hose to the gas outlet, check the tubing for cracks by gently stretching it and watching closely for small holes to appear. Make sure the gas control knob and the air control vent are closed, and then turn the handle on the main gas valve parallel to the nozzle to the gas jet. The gas valve is turned off by turning the handle 90 degrees in either direction. Open the gas control knob at the base of the laboratory burner and carefully check to see that you hear gas escaping from the mouth of the burner tube. When you are sure that gas is escaping, bring the head of the striker over the burner and squeeze the striker handle. The spark produced will ignite the gas and your burner is lit. Adjust the air control vent so that the flame has the proper color pictured here. A yellow flame is an indication of a lack of oxygen, meaning that the air vent needs to be opened. The hottest part of the burner flame is just at the top of the bright blue inner cone. Normal heating is done with an object at the top of the light blue outer cone, while strong heating is done with an object at the top of the bright blue inner cone. To heat a container gently, move the container back and forth through the outer cone. To cut off the burner flame, use the main gas valve handle (NOT the gas control knob!) to quickly turn the handle 90 degrees in either direction (forward or back). Lab Equipment Review Sheet You should know the name of each piece and its basic use. For exact volume measurements of liquids. Pipet on the left is a Volumetric pipet. It has only one graduation for delivering one exact volume. Pipet on the right is a Mohr pipet. It has graduations for delivering any number of exact volumes. Pipet There are several styles of fillers used to draw liquids into a pipet. Chemistry students use a standard pipet filler, shown at right. Pipet filler Never draw a liquid into a pipet with your mouth. Used in conjunction with a vacuum connection to a water faucet to speed up filtration. The Erlenmeyer Erlenmeyer flask flask is the most common flask in the chemistry lab. It is used to contain reaction solutions. The rounded bottom of the Florence flask makes it ideal The volumetric flask is used to Volumetric flask make solutions. It has a precise Filter flask Florence flask for boiling liquids. It also makes this flask easy to tip over when sitting on the lab table. graduation line in the neck of the flask. A solute is placed into the flask, then the solvent is used to bring the total volume up to the graduation. Beakers are the most versatile glassware in the lab and can be used for just about anything. The volume graduations on beakers should be used only for "ballpark" estimates. Used to make accurate measurements of liquid volumes. The bumper ring on larger cylinders is to prevent breakage if tipped over. Keep it near the top. Beaker This dish is used to recover dissolved solids by evaporation. Evaporating dish While it can be heated, it should not be used for "strong" heating. A watch glass can be used like an evaporating dish for very small amounts of liquid. It can also be used to cover beakers. When attached to the ring stand, this iron ring is used to support glassware above the lab table. When attached to the ring stand, this clamp is used to hold a large test tube or Florence flask above the lab table. Crucibles are used as a container when something requires "strong" Support ring Crucible and cover These tongs are used for picking up crucibles and crucible covers only. Graduated cylinder Watch glass Utility clamp Crucible tongs heating. Wire gauze Used as a support for beakers when placed across a support ring. Used to light a lab burner. Not a toy noisemaker during lab. Striker Mortar and Pestle Test tube holder Used to hold test tubes for short periods of "gentle" heating. Used to grind solids into powers. Wash bottle Filter funnel When lined with filter paper, used to filter suspended solids from a liquid. Used to transfer solids from their original container to a scale for weighing. Used for rinsing solids out of a container when filtering. Chemical spoon Used to close hoses by pinching them together. Hose clamps Desiccators are used to provide a dry environment for a Desiccator crucible or substance to cool down. This is critical in quantitative work where moisture can affect mass results. A common desiccant is anhydrous calcium chloride. Colored indicator crystals are usually included to tell the user the condition of the desiccant. A gray color in the crystals usually indicates that the desiccant is dry and will absorb moisture. A pink color usually means that the desiccant is "hydrated". When this happens, the water can be removed by heating it in an oven. Scientific Method Notes Definition: a systematic procedure for solving problems and exploring natural phenomena A Sample Method 1. Observations: the foundation of the scientific method. This observation might lead to a question regarding the event or characteristic. For example, you might drop a glass of water one day and observe it crashing to the floor near your feet. This observation might lead you to ask a question, "Why did the glass fall?" 2. Hypothesis: In attempting to answer the question, a scientist will form a hypothesis (or some would say a guess) regarding the question's answer. In our example there are many possible hypotheses, but one hypothesis might be that an invisible force (gravity) pulled the glass to the floor. Characteristics of good hypotheses: tentative explanations designed to guide experimentation a useful hypothesis must be testable must be rejected or corrected when they conflict with experiment Notice that the hypothesis postulates a relation between the independent and dependent variable, one which produces the change, the other one in which the change is produced. The hypothesis should fit well with what we already believe about the natural order of things. 3. Experimentation: Of all the steps in the scientific method, the one that truly separates science from other disciplines is the process of experimentation. In order to prove, or disprove, a hypothesis, a scientist will design an experiment to test the hypothesis. Over the centuries, many experiments have been designed to study the nature of gravity. In the late 16th century, it was generally believed that heavier objects would fall faster than lighter objects. The Italian scientist Galileo thought differently. Galileo hypothesized that two objects would fall at the same rate regardless of their mass. Legend has it that in 1590, Galileo planned out an experiment. He climbed to the top of the Leaning Tower of Pisa and dropped several large objects from the top of the Leaning Tower. What happens when you drop objects from the top of the tower? The two different objects fall at the same rate (as long as we ignore wind resistance). Data can be qualitative or quantitative. Quantitative data is obtained by making a measurement. Qualitative data is descriptive data that does not involve numbers. Accuracy indicates how close a measurement is to the accepted value. For example, we'd expect a balance to read 100.00 grams if we placed a standard 100.00 g weight on the balance. If it does not, then the balance is inaccurate. Precision indicates how close together or how repeatable the results are. A precise measuring instrument will give very nearly the same result each time it is used. Data is most useful when collected under controlled conditions (experiments). Experiments must be repeatable and reproducible. Develop a controlled experiment: Now we will take a closer look at what goes into making a "controlled" experiment. variable - The quantities on which the outcome of an experiment depends are called variables. dependent variable - this will be the single observation, or the result we will be observing . In reality one would observe as many dependent variables as possible. independent variable - this will be the single variable we elect to manipulate. All of the other variables must now be held constant so they don't influence our dependent variable. This way any change that occurs to the dependent variable can be attributed to our independent variable. "control" - this will be the part of our experiment that does not receive the independent variable. This step is critical to the validity of a controlled experiment. trials - Make sure sufficient data is gathered to form a conclusion. (the more trials, the better the data—usually!) 4. Analyze Data and Evaluate Results Galileo's experiment proved his hypothesis correct; the acceleration of a falling object is independent of the object's mass. Why is this true? A few decades after Galileo, Sir Isaac Newton would show that acceleration depends upon both force and mass. While there is greater force acting on a larger object, this force is canceled out by the object's greater mass. Thus two objects will fall (actually they are pulled) to the earth at exactly the same rate. Theories a well-tested explanation for experimental data based on a set of hypotheses tries to explain WHY or HOW something happens must be discarded or refined when they can't explain new experimental results Natural laws compactly summarize patterns in a large amount of data often apply only under special conditions tries to describe WHAT has happened Making Measurements Making measurements properly is somewhat more involved than it may seem. The proper unit has to be selected for both the property being measured and the size. The measuring instrument must be used correctly to assure accuracy of the measurement. Also, the precision of the measurement must be noted. Concept of Precision Accuracy and precision are concepts that you are already familiar with. In day to day living their importance varies considerably with the situation. If you are describing a window in terms of how it relates to its room, then you might say it is four feet by three feet in size. Not very precise but it gets the message across. On the other hand, if you were giving someone the dimensions to cut a piece of glass so that you could replace a broken window, then saying 4 ft x 3 ft would not be nearly precise enough. If it is cut 1/16th of an inch too large, it won't fit in the frame. If it is cut too small, it won't fill the frame and will either fall through or not seal properly. Note that whether we deal with general dimensions or replacing the glass, the precision necessary for an adequate description was dictated by the situation. Also note that in each case, the numbers used to describe the size were rounded off. Any measured value (except for counting) cannot be described exactly. Measured values are rounded off in a way that depends on how they are measured. Where the number is rounded off is its precision. Whether it is measured correctly is its accuracy. In science classes there are rules that determine what is expected of you when you make and record measurements. Write down measured values to the same degree of precision with which you measure them. In general, it is expected that you will make and record your measurements to a precision that involves estimating between the closest lines on the scale. Measurement Estimation All instruments intended for measurement are marked according to a scale on their surfaces. You are expected to estimate between the lines that make up the scale of an instrument to be most precise when you use any instrument. For examples, see below. Example 2 - parts a, b, c. On this scale the numbers mark 1, 2 and 3 cm. Each numbered division is divided into 10 parts, so each marked division is 0.1 cm. You are expected to read between the lines, so you need to report measurements to 0.01 cm. This is not hard but it is important. Look at line a. It extends past the 1.8 cm mark on the scale. By one estimate, it goes to 1.84 cm so that is what would be written down. You might see it the same way or estimate it at 1.83 cm or 1.85 cm. Either of those would be a reasonable measurement. Line b presents a bit of a problem. It extends to the 1.5 cm mark on the scale and no further. It could be written down as 1.5 cm, but you are supposed to estimate between the lines. Since Line b seems to line up EXACTLY with the mark you should write 1.50 cm. That gives the measurement to the nearest 0.01 cm and says that the measurement is right on the line as far as you can tell. In this measurement, the 0 that is after the 5 is a measured value and should be included in the measurement. Line c is somewhat similar to line b. Here the length of the line matches a numbered mark. so you need to put in two 0's in order to show that the measurement was measured to 0.01 unit. You should record your measurement as 2.00 cm. Also note that you shouldn’t extend any of these numbers by using any extra unmeasured digits. When dealing with precision, 1.50 cm does not mean the same thing as 1.500 cm. When writing down a measurement, put down all measured numbers including the estimate between the lines, or zero if on the line, but no more. Example 2 - Parts d and e. On this scale, the numbers mark 10, 20 and 30 cm. Again each numbered division is divided into 10 parts, so each mark is 1 cm. Since we read between the lines, measurements should be made to 0.1 cm. Thus line d is 14.3 cm rather than 14 cm or 14.30 cm. Occasionally in cases like this, people will forget that each mark is 1 cm instead of 0.1 cm and read the length as 10.43 cm. Try to avoid that mistake if you can. Line e measures at more than 20 cm and less than 21 cm and is estimated to be 20.8 cm. Be careful to note that the value is 20.8 cm and not 28 cm. General Information on Metric Measurements The sciences use a lot of measurements. Measurements have two parts: a number and a unit. The unit is just as important as the number. Writing a number without its unit is nonsense—it has no meaning. Some measurements can be measured directly, such as those below. 1. Length 2. Mass 3. Time 4. Temperature Other measurements need to be calculated, such as those below. 1. Speed 2. Acceleration 3. Force The units that we will be using are international standards called Le Systeme International d'Unites, or SI Units for short. Below is a table of the SI Base Units for some of the quantities mentioned above. Please know which unit is used to measure which quantity. NOTE: the actual SI base unit for mass is the kilogram—however, since we rarely use quantities in the science lab that are large enough to measure in kilograms, we will use a smaller, more convenient unit— grams. QUANTITY SI UNIT SI UNIT SYMBOL Length meter m Mass gram g Time second s Temperature Kelvin K Amount of Substance mole mol (this is a capital K) In science, we can measure quantities that are very large or very small. To avoid having to use very large or very small numbers we change the base units by adding prefixes to them. For the purposes of this class, we'll be using only three prefixes: kilo-, centi-, and milli-. Kilo- means 1,000 times the base unit. 1 kilo = 1,000 base unit OR 1/1,000 kilo = 1 base unit Centi- means 1/100 times the base unit. 1 centi = 1/100 base unit OR 100 centi = 1 base unit Milli- means 1/1000 times the base unit. 1 milli = 1/1,000 base unit OR 1,000 milli = 1 base unit If you notice in the lists above, there is no quantity that measures volume. We do measure a lot of volumes in science. Therefore, we need to use a derived unit. We can derive a unit for measuring volume by using length. In previous math classes you learned that the volume of a solid is calculated using the formula Volume = length x width x height If we measured a solid using meters then the derived unit would be cubic meters (m3). This doesn't help us much if we want to measure the volume of a liquid. It just so happens that one cubic centimeter (cm3) equals one milliliter (mL). We will also use the unit, liter (L), to measure the volume of liquids. Data Analysis Notes An experiment was performed in which plants were grown under the exposure of different colors of light. The hypothesis being investigated is that plants will grow best in green light. After four weeks this is what our data chart looks like. So now we have lots of data. What do we do next? We want to be able to use the data in the chart to draw a conclusion about what happened in our experiment. But it can sometimes be difficult to do this when the data are organized in a chart. Let’s take our information and graph it. A graph really helps show trends. We can use a graph to compare the heights of our plants more easily. Two types of graphs that are often used in science are line graphs and bar graphs. Line graphs are often used to show how one variable in an experiment changes over time. Bar graphs are used to display data in separate categories. For our experiment we could use a line graph to show how the heights of the plants changed over time, or we could use a bar graph to show the heights of our four plants at the end of the experiment. Both would be good ways to show the data in our chart. How do I set up a graph? There are a few rules about what goes where on a graph. In general, the independent (or manipulated) variable is plotted on the horizontal axis (x), and the dependent (or responding) variable is plotted on the vertical axis (y). The independent variable is the variable that you change during the experiment. In our experiment the independent variable was the color of light the plants received. The dependent variable is the one that changes as a result of the independent variable. The dependent variable is the one that you measure or observe during the experiment. In our experiment the dependent variable was the heights of the plants. How would the data look if we used a bar graph? Here is a bar graph of some of the data from our experiment. This graph shows the average heights of the plants after four weeks. How would our data look if we used a line graph? Let’s use a line graph to show how the plants’ heights changed during the last two weeks of the experiment. In this case time is the independent variable, and average plant height is the dependent variable. We’ll use a different line for each light color. So we’ve made graphs from our data. What are we supposed to do with the graphs? Now that the data are in a graph form, we can analyze the data and make conclusions. If you look at the graphs, you should be able to see some patterns. For instance, we can see that the plants grew to different heights, so we can conclude that the color of light that the plants received may have had an effect on their growth. In order to be certain we would have to repeat this experiment many times. We can also see that the tallest plants grew in blue light, the second tallest in red light, the third-tallest in white light, and the shortest in green light. The tomato plants grown in red and blue light grew more than our control, and the plants grown in green light grew less than our control. Wow! And we thought the green light would make the plants grow best. I guess our experiment was a waste of time, huh? Of course it wasn’t a waste of time! It’s true that our data don’t support our hypothesis, but that’s O.K. A hypothesis is an educated guess; it doesn’t have to be correct. Though we weren’t able to support our hypothesis, we discovered that the tomato plants that grew in the blue and red light were taller. This is important information that can lead to a new hypothesis to test. If the plants that received green light had grown taller than the others, could we have said that our hypothesis is true? No. We performed only a single experiment, and we used only eight plants. If we repeated our experiment, we might get slightly different results. In addition, we tested only tomato plants. If we tested other types of plants, we might find that different types of plants grow best in different colors of light. Is a hypothesis kind of like a theory? A theory is a general explanation of a set of observations about the natural world. A theory helps explain how things happen the way they do in nature. Unlike a hypothesis, a theory is supported by lots of data collected from many different experiments and observations. Theories can change over time as scientists gather more evidence. Hundreds of years ago scientists proposed a theory called spontaneous generation. This theory stated that some living things could develop from nonliving materials. For instance, it was thought that maggots could form from rotting meat. However, as scientists made more observations about the world around them, the theory of spontaneous generation was eventually replaced with the theory of biogenesis. The theory of biogenesis states that living things can come only from other living things. For example, it is now known that maggots hatch from tiny eggs that flies lay on rotting meat. SCIENCE SCIENCE Can our data tell me why the tomato plants grew taller in red and blue light than in green light? Unfortunately, our data do not answer that question. We would need to do much more research to find out how light affects plant growth. We would need to learn about the properties of red, blue, and green light. We would also need to understand how light is reflected and absorbed by plants and how plants use the light that they absorb. Maybe you could come up with another investigation!