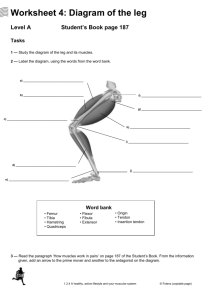
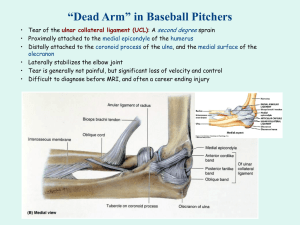
Current concepts in the rehabilitation of an ANNOTATION
advertisement

ANNOTATION Current concepts in the rehabilitation of an acute rupture of the tendo Achillis R. S. Kearney, M. L. Costa From University of Warwick, Coventry, United Kingdom R. S. Kearney, MSc, BSc Physiotherapy, Research Physiotherapist University of Warwick, Warwick Orthopaedics, Division of Health Sciences, Warwick Medical School, Clinical Sciences Research Laboratories, University Hospital, Clifford Bridge Road, Coventry CV2 2DX, UK. M. L. Costa, PhD, FRCS(Tr & Orth), Professor of Trauma and Orthopaedic Surgery University of Warwick, Warwick Clinical Trials Unit, Division of Health Sciences, Warwick Medical School, Coventry CV4 7AL, UK. Correspondence should be sent to Mrs R. S. Kearney; e-mail: r.s.kearney@warwick.ac.uk ©2012 British Editorial Society of Bone and Joint Surgery doi:10.1302/0301-620X.94B1. 28008 $2.00 J Bone Joint Surg Br 2012;94-B:28–31. 28 Rupture of the tendo Achillis is a common injury with a rising incidence. Traditionally the key question following this injury has been whether or not to operate. However a contemporary Cochrane review highlighted that the method of rehabilitation may also have an important contribution to the outcome. Since this review, various early weight-bearing rehabilitation protocols have been described. Currently evidence points to the use of early functional rehabilitation, regardless of operative or non-operative management. However, there is no consensus on which exact functional rehabilitation protocol should be used. Future research should be directed towards improving our understanding of how the different rehabilitative components interact in the tendo Achillis as it heals. Rupture of the tendo Achillis is a common injury and the incidence is rising.1 There is a bimodal distribution according to age with the first peak predominantly involving men aged 30 to 40 years and the second women aged 60 to 80 years.2 The first peak in incidence is often associated with participation in sport, such as football and racquet sports, whereas the second peak often occurs during normal daily activities such as climbing stairs.2 However, both often occur below the threshold of ‘macro-failure’ that might be expected in a healthy tendon. Macro-failure is a term associated with the stress–strain curve that characterises the mechanical response of tendons, and occurs beyond approximately 8% strain.3 It is widely accepted that spontaneous ruptures are the consequence of a pre-existing tendinopathy, which itself is attributed to failure in the protective/regenerative function that responds to repeated microscopic injury.4,5 Tendinopathy may develop in individuals with no obvious predisposition, but it has been linked to a number of external factors including use of topical corticosteroids,6 fluoroquinolone antibiotics7 and mechanical abnormalities of the foot.8 Intrinsic factors have also been associated with a predisposition including autoimmune and neurological conditions.9 The most common mechanism of injury is rapid eccentric contraction of the triceps surae, which may occur during sports with activities such as sprint starts, jumping and lunging or after an unexpected dorsiflexion of the ankle, which might occur when a patient stumbles. The patient typically presents with a sudden pain in the area of the tendo Achillis, often reporting a feeling of having been kicked in the back of the leg.10 Physical findings include a positive calf squeeze test,11 decreased ankle plantarflexion strength, presence of a palpable gap and increased ankle dorsiflexion with gentle manipulation.12 Following a diagnosis of a mid-substance rupture of the tendo Achillis the clinician is faced with a range of management options. This article will summarise the latest developments in this area. Management options Traditionally the key question in relation to a rupture of the tendo Achillis was simply whether to operate or not. In 1981 Nistor13 designed and published the first randomised control trial (RCT) to address this clinical question, involving 105 patients, all of whom underwent between six and nine weeks of immobilisation in a cast, irrespective of whether they were in the operative group. This study was followed by a series of trials which were pooled in a meta-analysis by the Cochrane review group in 2004.14 The results suggested a re-rupture rate of 3.5% in the operatively treated group and 12.6% in the non-operatively treated group. However, this was alongside a pooled rate of ‘other’ complications of 34.1% in the operatively treated group and 2.7% in the non-operatively treated group. Therefore, operative repair seemed to reduce the risk of re-rupture but came with an increased risk of other complications, most of which were associated with infection and wound healing. THE JOURNAL OF BONE AND JOINT SURGERY CURRENT CONCEPTS IN THE REHABILITATION OF AN ACUTE RUPTURE OF THE TENDO ACHILLIS In an attempt to reduce the number of wound complications associated with operative repair, clinicians have investigated percutaneous repair, as opposed to the traditional open longitudinal approach. The percutaneous approach involves small stab incisions along both borders of the tendon and then passing a suture through the tendon using these incisions, as first described by Ma and Griffith.15 The first RCT to examine the benefit of this approach was published by Schroeder, Lehmann and Steinbrueck16 in 1997, followed by two further RCTs in 2001 and 2008.17,18 These have all reported favourably towards the percutaneous approach, regarding complication rates. However, it was also noted that there is a higher rate of sural nerve injury with percutaneous approaches. Consequently, a range of surgical techniques, which sit between the open longitudinal and fully percutaneous techniques, have been developed but there is currently no standard procedure amongst surgeons. The 2004 Cochrane review also evaluated two RCTs published in 199219 and 2002,20 which compared cast immobilisation with functional bracing within patients managed non-operatively. These suggested that there was a reduced rerupture rate of 2.4% in the functional bracing group, compared with 12.2% in the casting group. This raised the possibility that functional rehabilitation might reduce the rate of re-rupture to that observed in operatively managed patients, in which case the strength of the indication for surgical intervention was reduced. However, the authors of the review also noted the considerable variation in functional bracing protocols documented within the literature. Functional rehabilitation The term functional rehabilitation has been applied in various studies using a variety of orthotics and rehabilitation regimes. However, the key components are weight-bearing mobilisation, usually within an orthotic device with early, but limited, range of movement being permitted. With regard to the orthosis, there are two basic designs: rigid rocker bottom style,21 or the more flexible carbon-fibre dorsal orthoses.22 The flexible orthosis generally facilitates a greater range of movement than the more rigid designs. The choice of orthosis is just one consideration. There is also the issue of what degree of plantarflexion should be maintained within the orthosis. Some studies have advocated that the ankle should be positioned in neutral21 (plantar-grade) but with restricted dorsiflexion, while others have used three heel-wedge inserts such that the ankle joint is initially maintained at near full plantarflexion.23 The latter protocol reproduces the more traditional use of sequential plaster casts with gradually reducing plantarflexion. Finally, there is the consideration of when weightbearing should be permitted (day one or within first two weeks), for how long the orthosis should be worn and whether or not to permit active range of movement exercises throughout the period when the orthosis is worn.21,22,24-26 There is a trend towards immediate full VOL. 94-B, No. 1, JANUARY 2012 29 weight-bearing and earlier removal of the orthosis with an average of eight weeks reported in one study,26 but it would be wrong to suggest that there is a consensus in this area. This range of protocols exemplifies the complexity of this early rehabilitative intervention. When developing an intervention that has several interacting components the clinician can reasonably refer to the growing number of case series, documenting what has been tried and tested, and implement a protocol into their practice accordingly. An alternative approach, recommended by the Medical Research Council,27 is to develop an understanding of the underlying causal mechanisms why these different components may contribute to improved outcomes following a rupture of the tendo Achillis. The phases of tendon healing have been divided into inflammation (first week), proliferation (weeks two to eight) and remodelling (up to 12 months).28 Throughout these phases the tendon’s tensile strength gradually increases, but remains inferior to the uninjured tissue. The biomechanical inferiority of the newly formed scar tissue is due to its increased stiffness and subsequently decreased visco-elastic properties.29 Animal models have shown that early loaded movement improves the biomechanical properties of the scar tissue, decreases excessive adhesion formation and subsequently enhances the gliding function of the tendon.30,31 Furthermore weight-bearing during the early phases of healing stimulates fibroblast activity and type III collagen synthesis.31 Consequently, there is both preliminary clinical evidence and underlying basic science research to support early loading and early movement. The RCT using functional rehabilitation involved early, but not immediate weight-bearing.19 Subsequently, there were RCTs to compare cast immobilisation with ‘immediate’ full weight-bearing functional bracing, firstly within operatively managed patients and secondly within nonoperatively managed patients.22,32 These studies suggested that immediate weight-bearing is safe, in terms of both risk of re-rupture and the risk of tendon lengthening, for both groups, and offer practical advantages for patients and some evidence of improved functional outcomes. Operative versus non-operative treatment with functional rehabilitation The next logical research question was therefore to compare operative with non-operatively managed patients using accelerated, functional rehabilitation methods for both groups. The first RCT for this step was carried out by Metz et al25 in 2008 and then more recently by Willits et al33 in 2010. Both studies used early functional bracing (within two weeks of injury) and both found no statistically significant differences in outcome between operatively and nonoperatively managed patients. The primary outcome measures were complications other than re-rupture in one study25 and the rate of re-rupture in the other.33 Both RCTs had clear inclusion and exclusion criteria and accountability for participant flow throughout the studies. They provide strong 30 R. S. KEARNEY, M. L. COSTA evidence to suggest that there is no difference in the risk of rerupture whether the rupture is treated with operative repair or managed non-operatively, provided functional rehabilitation is undertaken in the early phases of rehabilitation. However there are some unresolved issues. For example, Metz et al25 managed the two groups using somewhat different rehabilitation protocols. In contrast, Willits et al33 used an identical rehabilitation protocol for both groups, but the patient sample was recruited from two sports medical centres, bringing into question the external validity of the results. Furthermore it is important to acknowledge that both studies used complications as their primary measure of treatment success. The most appropriate measure of the success of treatment, however, remains controversial. Outcome measures In order to ascertain superiority of one intervention over another, the appropriate choice of primary outcome measure is imperative. Traditionally, within the musculoskeletal field, published studies have focused on the technical outcomes of procedures, but more recently there has been a move towards the assessment of effectiveness from the patients’ perspective.34 A wide range of patient reported functional measures have been developed over the last two decades, such as the Victorian Institute of Sports Assessment questionnaire for patellar and Achilles tendinopathy,35,36 and in other areas assessments for both the upper and lower limb.37-39 However, until recently there was no validated, patient-reported outcome tool specifically for patients with a rupture of the tendo Achillis. The Thermann score40 and Leppilahti score41 have been used in some studies but neither tool has undergone in-depth validation, being predominantly based on expert opinion.42 In 2007, a research group addressed this gap publishing a new patient reported outcome measure with supporting validation data.42 This outcome measure was the Achilles tendon Total Rupture Score (ATRS). The ATRS contains ten items, for which patients are asked to respond using an 11-grade Likert scale.43 This scale ranges from a score of zero, which is equivalent to a patient having major limitations or symptoms, to a score of ten, which is equivalent to a patient having no limitations or symptoms. The original validation paper is based on a select population of patients aged 20 to 70 years, within a Swedish sample.42 Conclusions and future research In 2010, the first clinical guidelines on the treatment of acute rupture of the tendo Achillis were published by the American Academy of Orthopaedic Surgeons and endorsed by the American Foot and Ankle Society.44 The guidelines provided moderate recommendations in two areas: firstly the use of early (two weeks or before) post-operative weight-bearing and secondly the use of a protective/ orthotic device to allow mobilisation. However, this report recognised that there were a range of protocols and ‘protective devices’ and there is a current lack of clinical consensus regarding which devices should be used, the degree of ankle plantarflexion they should facilitate and how long they should be worn. Over the past few years, the potential benefits of early functional rehabilitation have increasingly been documented within randomised controlled trials, meta-analyses and guideline development groups. Yet, despite this growing body of evidence, there are still reports of prolonged cast immobilisation following this injury. This is particularly puzzling in light of the increased complications associated with prolonged immobilisation, including venous thromboembolism, muscle atrophy from disuse and higher rates of re-rupture.22,33,45,46 The literature to date has focused on three core areas. Firstly, the question of whether to operate or not.47,48 Secondly, if operative repair is favoured, which technique is the best, and finally whether to immobilise the patient in a plaster cast or to use accelerated rehabilitation with functional bracing. However, new and emerging themes are developing as a result of advances in basic science and animalbased research. One such area is improving the ‘biology’ that governs tendon healing. Published animal studies have investigated the use of bone morphogenetic proteins, autologous conditioned serum, fibroblast growth factor, vascular endothelial growth factor, platelet derived growth factor and insulin-like growth factor-1.49-52 The findings are increasingly being translated into clinical research. One example specific to the tendo Achillis is an ongoing RCT comparing the use of autologous platelet-rich plasma versus standard treatment for patients with an acute rupture (trial registration: ISRCTN93608625), but there are likely to be many more studies as our understanding of tendon healing improves. In conclusion, although current evidence points to the use of early functional rehabilitation, regardless of operative or non-operative management, there is no consensus on which exact functional rehabilitation protocols should be used. Unanswered questions include: Which type of orthosis should be used? Is movement important or is early loading enough? What degree of plantarflexion provides the best balance between tendon lengthening/re-rupture and disuse atrophy in the calf muscles? The next few years of research will no doubt improve our understanding of these ‘mechanical’ factors but perhaps the most exciting developments will come with a better knowledge of how the mechanical and biological environment interacts in the tendo Achillis as it heals. No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article. References 1. Leppilahti J, Puranen J, Orava S. Incidence of Achilles tendon rupture. Acta Orthop Scand 1996;67:277–279. 2. Maffulli N, Waterston SW, Squair J, Reaper J, Douglas AS. Changing incidence of Achilles tendon rupture in Scotland: a 15-year study. Clin J Sport Med 1999;9:157– 160. THE JOURNAL OF BONE AND JOINT SURGERY CURRENT CONCEPTS IN THE REHABILITATION OF AN ACUTE RUPTURE OF THE TENDO ACHILLIS 3. Wang JH, Iosifidis MI, Fu FH. Biomechanical basis for tendinopathy. Clin Orthop 2006;443:320–332. 4. Riley G. Tendinopathy: from basic science to treatment. Nat Clin Pract Rheumatol 2008;4:82–89. 5. Tallon C, Maffulli N, Ewen SW. Ruptured Achilles tendons are significantly more degenerated than tendinopathic tendons. Med Sci Sports Exerc 2001;33:1983–1990. 6. Newnham DM, Douglas JG, Legge JS, Friend JA. Achilles tendon rupture: an underrated complication of corticosteroid treatment. Thorax 1991;46:853–854. 7. Royer RJ, Pierfitte C, Netter P. Features of tendon disorders with fluoroquinolones. Therapie 1994;49:75–76. 8. Clement DB, Taunton JE, Smart GW. Achilles tendinitis and peritendinitis: etiology and treatment. Am J Sports Med 1984;12:179–184. 9. Maffulli N, Kader D. Tendinopathy of tendo Achillis. J Bone Joint Surg [Br] 2002;84B:1–8. 10. Maffulli N. Rupture of the Achilles tendon. J Bone Joint Surg [Am] 1999;81-A:1019– 1036. 11. Simmonds FA. The diagnosis of the ruptured Achilles tendon. Practitioner 1957;179:56–58. 12. Maffulli N. The clinical diagnosis of subcutaneous tear of the Achilles tendon: a prospective study in 174 patients. Am J Sports Med 1998;26:266–270. 13. Nistor L. Surgical and non-surgical treatment of Achilles Tendon rupture: a prospective randomized study. J Bone Joint Surg [Am] 1981;63-A:394–399. 14. Khan RJ, Fick D, Brammar TJ, Crawford J, Parker MJ. Interventions for treating acute Achilles tendon ruptures. Cochrane Database Syst Rev 2004;CD003674. . 15. Ma GW, Griffith TG. Percutaneous repair of acute closed ruptured achilles tendon: a new technique. Clin Orthop 1977;128:247–255. 16. Schroeder D, Lehmann M, Steinbrueck K. Treatment of acute achilles tendon ruptures: open vs. percutaneous repair vs. conservative treatment: a prospective randomized study. Orthop Trans 1997;21:1228. 17. Lim J, Dalal R, Waseem M. Percutaneous vs. open repair of the ruptured Achilles tendon: a prospective randomized controlled study. Foot and Ankle Int 2001;22:559– 568. 18. Gigante A, Moschini A, Verdenelli A, et al. Open versus percutaneous repair in the treatment of acute Achilles tendon rupture: a randomized prospective study. Knee Surg Sports Traumatol Arthrosc 2008;16:204–209. 19. Saleh M, Marshall PD, Senior R, MacFarlane A. The Sheffield splint for controlled early mobilisation after rupture of the calcaneal tendon: a prospective, randomised comparison with plaster treatment. J Bone Joint Surg [Br] 1992;74-B:206– 209. 20. Petersen OF, Nielsen MB, Jensen KH, Solgaard S. Randomized comparison of CAM walker and light-weight plaster cast in the treatment of first-time Achilles tendon rupture. Ugeskr Laeger 2002;164:3852–3855 (in Danish). 21. Speck M, Klaue K. Early full weightbearing and functional treatment after surgical repair of acute achilles tendon rupture. Am J Sports Med 1998;26:789–793. 22. Costa ML, MacMillan K, Halliday D, et al. Randomised controlled trials of immediate weight-bearing mobilisation for rupture of the tendo Achillis. J Bone Joint Surg [Br] 2006;88-B:69–77. 23. Bhattacharyya M, Gerber B. Mini-invasive surgical repair of the Achilles tendon: does it reduce post-operative morbidity? Int Orthop 2009;33:151–156. 24. Jacob KM, Paterson R. Surgical repair followed by functional rehabilitation for acute and chronic achilles tendon injuries: excellent functional results, patient satisfaction and no reruptures. ANZ J Surg 2007;77:287–291. 25. Metz R, Verleisdonk EJ, van der Heijden GJ, et al. Acute Achilles tendon rupture: minimally invasive surgery versus nonoperative treatment with immediate full weightbearing: a randomized controlled trial. Am J Sports Med 2008;36:1688–1694. 26. Kearney RS, McGuinness K, Achten J, Costa ML. Physiotherapy: a systematic review of early rehabilitation methods following a rupture of the achilles tendon, 2011. http://www.totallyphysio.com/uploads/6/6/2/4/6624770/ piis0031940611004160.pdf (date last accessed 4 August 2011). 27. No authors listed. Medical Research Council: Complex interventions guidance, 2008. http://www.mrc.ac.uk/Utilities/Documentrecord/index.htm?d=MRC004871 (date last accessed 4 August 2011). VOL. 94-B, No. 1, JANUARY 2012 31 28. Maffulli N, Moller HD, Evans CH. Tendon healing: can it be optimised? Br J Sports Med 2002;36:315–316. 29. Evans NA, Stanish WD. The basic science of tendon injuries. Curr Orthop 2000;14:403–412. 30. Lin TW, Cardenas L, Soslowsky LJ. Biomechanics of tendon injury and repair. J Biomech 2004;37:865–877. 31. Enwemeka CS. Functional loading augments the initial tensile strength and energy absorption capacity of regenerating rabbit Achilles tendons. Am J Phys Med Rehabil 1992;71:31–38. 32. Maffulli N, Tallon C, Wong J, Lim KP, Bleakney R. Early weightbearing and ankle mobilization after open repair of acute midsubstance tears of the achilles tendon. Am J Sports Med 2003;31:692–700. 33. Willits K, Amendola A, Bryant D, et al. Operative versus nonoperative treatment of acute Achilles tendon ruptures: a multicenter randomized trial using accelerated functional rehabilitation. J Bone Joint Surg [Am] 2010;92-A:2767–2775. 34. Gartland JJ. Orthopaedic clinical research: deficiencies in experimental design and determinations of outcome. J Bone Joint Surg [Am] 1988;70-A:1357–1364. 35. Robinson JM, Cook JL, Purdam C, et al. The VISA-A questionnaire: a valid and reliable index of the clinical severity of Achilles tendinopathy. Br J Sports Med 2001;35:335–341. 36. Visentini PJ, Khan KM, Cook JL, et al. The VISA score: an index of severity of symptoms in patients with jumper's knee (patellar tendinosis): Victorian Institute of Sport Tendon Study Group. J Sci Med Sport 1998;1:22–28. 37. Dawson J, Fitzpatrick R, Carr A, Murray D. Questionnaire on the perceptions of patients about total hip replacement. J Bone Joint Surg [Br] 1996;78-B:185–190. 38. Dawson J, Fitzpatrick R, Carr A, Murray D. Questionnaire on the perceptions of patients about total knee replacement. J Bone Joint Surg [Br] 1998;80-B:63–69. 39. Hudak PL, Amadio PC, Bombardier C. Development of an upper extremity outcome measure: the DASH (disabilities of the arm, shoulder and hand): The Upper Extremity Collaborative Group (UECG). Am J Ind Med 1996;29:602–608. 40. Thermann H, Frerichs O, Biewener A, Krettek C, Schandelmeier P. Functional treatment of acute rupture of the Achilles tendon: an experimental biomechanical study. Unfallchirurg 1995;98:507–513 (in German). 41. Leppilahti J, Forsman K, Puranen J, Orava S. Outcome and prognostic factors of achilles rupture repair using a new scoring method. Clin Orthop 1998;346:152–161. 42. Nilsson-Helander K, Thomeé R, Silbernagel KG. The Achilles tendon Total Rupture Score (ATRS): development and validation. Am J Sports Med 2007;35:421–426. 43. Likert R. A technique for the measurement of attitudes. Archives of Psychology 1932;140:1–55. 44. Chiodo CP, Glazebrook M, Bluman EM, et al. American Academy of Orthopaedic Surgeons clinical practice guideline on treatment of Achilles tendon rupture. J Bone Joint Surg [Am] 2010;92-A:2466–2468. 45. Twaddle BC, Poon P. Early motion for Achilles tendon ruptures: is surgery important? A randomized, prospective study. Am J Sports Med 2007;35:2033–2038. 46. Healy B, Beasley R, Weatherall M. Venous thromboembolism following prolonged cast immobilisation for injury to the tendo Achillis. J Bone Joint Surg [Br] 2010;92B:646–650. 47. Keating JF, Will EM. Operative versus non-operative treatment of acute rupture of tendo Achillis: a prospective randomised evaluation of functional outcome. J Bone Joint Surg [Br] 2011;93-B:1071–1078. 48. Wallace RG, Heyes GJ, Michael AL. The non-operative functional management of patients with a rupture of the tendo Achillis leads to low rates of re-rupture. J Bone Joint Surg [Br] 2011;93-B:1362–1366. 49. Majewski M, Ochsner PE, Liu F, Flückiger R, Evans CH. Accelerated healing of the rat Achilles tendon in response to autologous conditioned serum. Am J Sports Med 2009;37:2117–2125. 50. Bolt P, Clerk AN, Luu HH, et al. BMP-14 gene therapy increases tendon tensile strength in a rat model of Achilles tendon injury. J Bone Joint Surg [Am] 2007;89A:1315–1320. 51. Chan BP, Chan KM, Maffulli N, Webb S, Lee KK. Effect of basic fibroblast growth factor: an in vitro study of tendon healing. Clin Orthop 1997;342:239–247. 52. Sharma P, Maffulli N. Tendon injury and tendinopathy: healing and repair. J Bone Joint Surg [Am] 2005;87-A:187–202.