Science SCI.IV.1.3 Grade: 6
advertisement

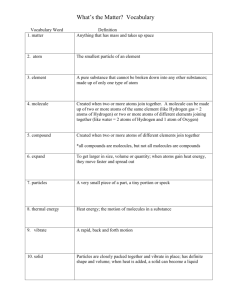
Science SCI.IV.1.3 Grade: 6 Strand IV: Using Scientific Knowledge in Physical Science Standard 1: Matter and Energy - All students will explain what the world around is made of. Benchmark 3: Classify substances as elements, compounds, or mixtures and justify classifications in terms of atoms and molecules. Constructing and Reflecting: SCI.I.1.2 SCI.I.1.3 SCI.I.1.4 SCI.I.1.5 SCI.II.1.5 SCI.II.1.6 - Design and conduct scientific investigations. Use tools and equipment appropriate to scientific investigations. Use metric measurement devices to provide consistency in an investigation. Use sources of information in support of scientific investigations. Develop an awareness of and sensitivity to the natural world. Recognize the contributions made in science by cultures and individuals of diverse backgrounds. Vocabulary / Key Concepts Context • element • compound • mixture • molecule • atom Common substances such as those listed above, including elements such as • copper • aluminum • sulfur • helium • iron • compounds, such as • water • salt • sugar • carbon dioxide See IV.1.MS.4 Molecular structure of solids, liquids and gases mixtures, such as • soil • salt and pepper • salt water • air • Iron filings, sand, and salt Resources Knowledge and Skills Students will classify substances as elements, matter that is made up of atoms with identical chemical properties. Aluminum, gold, copper iron, hydrogen, and oxygen are examples of elements. Compounds exist when different elements are joined together to form a new chemical substance. Water (H2O), table salt (NaCl), and chalk (CaCO) are examples of compounds. Mixtures are a combination of two or more substances that keep their chemical properties. Soil, salt and pepper, KoolAid, and sugar water are examples of mixtures. Mixtures are combinations of elements and/or compounds. Mixtures can be physically separated into original components. Benchmark Clarification: Matter consists of extremely small, invisible particles called atoms. Atoms cannot be broken down into their smaller parts during a physical change, or during a chemical reaction. They can be broken into smaller particles during nuclear reactions. All elements are made of one kind of atom. (See periodic table of the Coloma Resources: Lab: Classifying common substances as element, compound, mixture Matter & Molecules (see handouts) Other Resources: • MSU Instructional Unit Matter & Molecules • MDE Instructional Units Steamed Up and Hard As Ice • Bill Nye: Chemical Phases in Matter • Science Explosion: “Elements, Compounds, Mixtures,” “Elements” • Michigan Teacher Network Resources elements) • BCISD Physical Science Resources Atoms may exist alone or be combined together. When two or more atoms join together, they form molecules. When different kinds of elements join together, they form compounds. • Oxford Science Encyclopedia – Atoms and Molecules • Jefferson Lab – Questions and Answers Atoms, Elements and Molecules For example, oxygen exists as a molecule containing two oxygen atoms in the atmosphere. When three oxygen atoms join together, they form the molecule called ozone. Water is a substance / a compound made of hydrogen and oxygen atoms. Each water molecule is made of two atoms of hydrogen and one atom of oxygen. These molecules/compounds do not break down into individual atoms/elements when they are heated or cooled during phase changes. Molecules can be broken down into separate atoms or simpler molecules when exposed to electrical current or during a chemical reaction. Examples of chemical reactions include: • a reaction with acids • decomposition • burning (a chemical reaction with oxygen). When two or more elements and/or compounds are physically combined together, they are called a mixture. Mixtures can be physically separated into their original components. Videoconferences Available For more information, see www.remc11.k12.mi.us/dl or call Janine Lim 471-7725x101 or email jlim@remc11.k12.mi.us IV.1.MS.3 Mixed-up Mixtures and Solutions from Hook's Discovery and Learning Center Heart of the Matter from COSI Toledo 6th Grade Science Curriculum Technology Resources IV.1.MS.3 Classify substances as elements, compounds, or mixtures and justify classifications in terms of atoms and molecules. Assessment Instruction Focus Question: How are elements, compounds Optional Assessment and mixtures classified in terms of atoms and Create a chart, arranging the following items into molecules? the appropriate classification as an element, Each student will construct at least one model of an compound or mixture. Explain why you placed element and one model of a compound by using each item in its category. (Water is compound broken toothpicks and raisins, colored minibecause it is made of the elements hydrogen and marshmallows, or gumdrops. oxygen and cannot be easily separated. Salt water is a mixture because it is made up of salt Students will work in small groups and create a and water and can be separated back into its concept map to classify elements, compounds, and parts. Iron is element because it is made of only mixtures in terms of atoms and molecules. They will one kind of atom which can be found on the glue and label the models they are creating onto the periodic chart.) concept map. Students will choose a single color item to represent one kind of atom. They will label this on the concept map as atom 1. They should repeat this step for atom 2 and atom 3. Then they should combine individual atoms (single color items) to form elements (all atoms the same color and property) and glue their element samples onto their concept maps and label them. Item bank: Kool-Aid, salt, aluminum foil, salad dressing, copper wire, soil, chalk, salt water, carbon dioxide, sugar They should form compounds by attaching elements with toothpicks (toothpicks represent bonds) and glue these samples onto their concept maps and label them. Scoring Rubric They should form mixtures by combining two or more elements and/or compounds (these are not bonded; do not use toothpicks) and glue these samples onto their concept maps and label them. They should add definitions of terms and real-world examples for each element and compound. NOTE TO TEACHER: Check Benchmark Clarification for proper classification. Criteria: Completeness of chart: Apprentice - Creates a chart with few headings and some missing information. Basic - Creates a complete chart with correct headings but some missing information. Meets - Creates a complete and correct chart with proper headings. Exceeds - Creates a complete and correct chart with proper headings and detailed explanations. Criteria: Correctness of identification: Apprentice - Identifies three or fewer items. Basic - Identifies four to six items. Meets - Identifies seven to eight items correctly and completely. Exceeds - Identifies all nine items correctly and completely. Criteria: Correctness of justification: Apprentice - Justifies three or fewer items. Basic - Justifies four to six items. Meets - Justifies seven to eight items correctly and completely. Exceeds - Justifies all nine items correctly and completely. Teacher Notes: “All matter is made up of atoms, which are far too small to see directly through a microscope. The atoms of any element are alike but are different from atoms of other elements. Atoms may stick together in well-defined molecules or may be packed together in large arrays. Different arrangements of atoms into groups compose all substances.” (BSL) “Chemical elements do not break down during normal laboratory reactions involving such treatments as heating, exposure to electric current, or reaction with acids. There are more than 100 known elements that combine in a multitude of ways to produce compounds, which account for the living and nonliving substances that we encounter.” (NSES) “Atoms and molecules are perpetually in motion. Increased temperature means greater average energy of motion, so most substances expand when heated. In solids, the atoms are closely locked in position and can only vibrate. In liquids, the atoms or molecules have higher energy, are more loosely connected, and can slide past one another; some molecules may get enough energy to escape into a gas. In gases, the atoms or molecules have still more energy and are free of one another except during occasional collisions.” (BSL) Matter consists of extremely small, invisible particles called atoms. Atoms may exist alone (e.g. inert gases), or combined together. Atoms combine in different ways to form all substances. All elements are made of one kind of atom. (See Periodic Table). These elements do not breakdown during lab reactions involving heating, exposure to electrical current, or reaction with acids. (However, they can be broken down by nuclear reactions!) These elements can also combine to form compounds. Focus Questions • • • How are elements, compounds and mixtures different from each other in terms of atoms and molecules? What is the arrangement and motion of molecules in solids, liquids and gases? How can you use the idea of atoms and molecules to explain why solids are hard, liquids flow and gases are invisible? (JCISD) Notes Students often hold the following misconceptions about atoms and molecules: 1. There is no empty space between molecules; rather students believe there is dust, germs, or “air” between the particles of air. 2. Particles possess the same properties as the materials they compose. For example, copper atoms are “orange” and molecules of a solid are “hard.” (JCISD)