CE 403 Optional FE Review: Thermodynamics Chris Rehmann

CE 403 Optional FE Review:
Thermodynamics
Chris Rehmann
Thermodynamics
• 1 st and 2 nd laws of thermodynamics
• Energy, heat, and work
• Availability and reversibility
• Cycles
• Ideal gases
• Mixture of gases
• Phase changes
• Heat transfer
• Properties of enthalpy and entropy
Properties: PSU METBD 330 #11
Which of the following is an extensive property?
A. temperature
B. weight
C. composition
D. pressure
E. none of the above
See http://engr.bd.psu.edu/davej/classes/EIT_330.html
Properties: PSU METDB 330 #3
How much energy is required to heat 2.5 kg of water from 70°C to 95°C at 100 kPa pressure?
A. 105 kJ
B. 31 kJ
C. 211 kJ
D.262 kJ
E. 78 kJ
Properties: USF QOTD 12.2
See http://fe.eng.usf.edu/section12/q2/qtemplate.asp
. Change “q2” to get other questions.
Properties: PSU METBD 330 #5
What is the enthalpy of water at 165°C with a quality of
97%?
A. 4766 kJ/kg
B. 3761 kJ/kg
C. 2701 kJ/kg
D. 2066 kJ/kg
E. 2004 kJ/kg
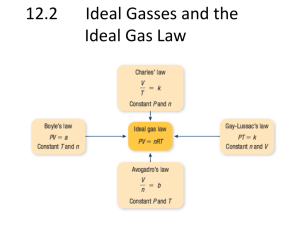
Ideal gas: PSU METBD 330 #6
Air at 205°C and 345 kPa is compressed isothermally to one-tenth of its original volume. What is the final pressure?
A. 690 kPa
B. 6900 kPa
C. 2585 kPa
D. 34.5 kPa
E. 3450 kPa
Ideal gas: PSU METBD 330 #13
When the pressure of an ideal gas is doubled and the absolute temperature is halved, its volume is:
A. constant
B. doubled
C. increased by 4 times
D. cut in half
E. one fourth of its original
Ideal gas: PSU METBD 330 #12
A 0.71 m 3 tank contains 4.5 kg of an ideal gas. The molecular weight of this gas is 44 kg/kmole and the gas temperature is 21 o C. What is the gas pressure ?
A. 352 kPa
B. 25 kPa
C. 190 kPa
D. 15497 kPa
E. 25 MPa
Ideal gas: USF QOTD 12.4
1 st Law—work: PSU METBD 330 #15
An ideal gas in a frictionless, vertical piston-cylinder device is heated and expands from a volume of 0.04 m 3 to 0.10 m 3 while the pressure remains constant at 200 kPa. What work is done by the system?
A. 8 kJ
B. 10 kJ
C. 12 kJ
D. 14 kJ
E. none of the above
1 st Law: isothermal compression
A bicycle pump compresses air from a volume of 200 cm 3 to a volume of 100 cm 3 , lets the entire air volume escape, draws in a new volume, and so on. The pressure is initially 10 5 N/m 2 and temperature is 20°C. If the pump is cooled as it operates so that the temperature is constant, what is the work done per stroke by the pumping?
A. 8 J
B. 10 J
C. 12 J
D. 14 J
E. 14 kJ
1 st Law: USF QOTD 12.5
1 st Law—isentropic process:
PSU METBD 330 #2
A 57 liter volume of air is compressed from 1 atmosphere to 1.60 atm. in an isentropic process. What is the final volume of the air if it is further compressed to 208 kPa in an isentropic process?
A. 34 L
B. 17 L
C. 10.5 L
D. 21 L
E. 42 L
1 st Law—polytropic process:
PSU METBD 330 #7
Four kilograms of nitrogen gas is compressed in a reversible, adiabatic process from 101.325 kPa and 20°C to 506.625 kPa. The compression process follows a polytropic relationship with n=1.4. What work is done on the gas during the compression?
A. -885 kJ
B. 508 kJ
C. 776 kJ
D. 885 kJ
E. -508 kJ
For closed systems:
Carnot cycle: PSU METBD 330 #8
What is the efficiency of an ideal Carnot heat engine cycle operating between 38°C and 482°C?
A. 90 %
B. 89 %
C. 59 %
D. 16 %
E. 11 %
Carnot cycle: PSU METBD 330 #9
A Carnot refrigeration cycle operates between -10°C and 23°C. What is its coefficient of performance?
A. 1.3
B. 3.7
C. 4.5
D. 7.9
E. 8.9
Isentropic process: AUC exam #114
Cycles: AUC exam #115
Ideal gas law: AUC exam #116
Superheated water: AUC exam #117
Carnot cycle: AUC exam #118
Gas constant: AUC exam #119
Adiabatic process: AUC exam #120