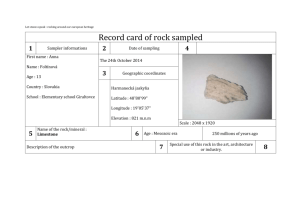
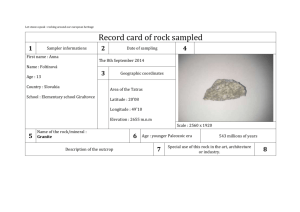
UCCS PES/ENSC 2500 Renewable Energy name: _________________________
advertisement

UCCS PES/ENSC 2500 Renewable Energy Chapter 15 Geothermal Energy name: _________________________ Problem 15.1 Calculate the ideal Carnot efficiency for a turbine operating with a hot reservoir at a temperature of 440°F and a cold reservoir at a temperature of 170°F. Converting temperatures to absolute degrees (Kelvin) gives Then the Carnot efficiency is Problem 15.4 Heat is extracted from hot rock at a temperature of 250°C and used to produce electricity with the ideal Carnot efficiency. If the temperature of the cold reservoir is 75°C, what mass of rock would be needed to yield 1 GWhe. See Chapter 8 for the thermal properties of typical rock, and assume a generator efficiency of 90%. The heat extracted from rock is Q = CmΔT. From Table 8.6, for rock C = 879 J/(kg ºC). We need a net energy of 1 GWhe and at 90% efficiency the total energy required is (1 GWhe)/(0.9) = 1.1 GWh. Converting this to Joules gives For the temperatures given for the hot and cold reservoirs in ºC, we calculate the equivalent temperatures in K as Thus the ideal Carnot efficiency is η = This means that the total energy required is For ΔT = _________________________________ the mass is Problem 15.6 Assume that geothermal heat transfer (at least near the surface of the earth) is by conduction through the crust rocks. 2 For an average geothermal heat flow of 0.087 W/m and typical thermal gradient of 100°C/km, calculate the thermal conductivity of the rock. Compare with the known thermal conductivities of similar materials given in Chapter 8. Equation (8.4) gives where k is the thermal conductivity, A is the area, ΔT is the temperature difference and l is the distance. Solving for k gives So in this case P/A = 0.087 W/m and ΔT/l = 100ºC/km or 0.001ºC/cm. 2 The thermal conductivity is then From Table 8.1, The values for concrete and brick are: 170 value for rock is in the proper range. and 71 respectively, so the calculated