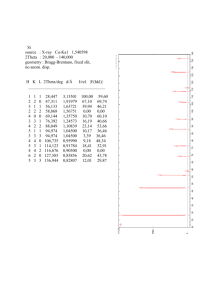
Crystal Structures of Interest • Elemental solids: • Binary compounds
advertisement

Crystal Structures of Interest
• Elemental solids:
– Face-centered cubic (fcc)
– Hexagonal close-packed (hcp)
– Body-centered cubic (bcc)
– Diamond cubic (dc)
• Binary compounds
– Fcc-based (Cu3Au,NaCl, ß-ZnS)
– Hcp-based (α-ZnS)
– Bcc-based (CsCl, Nb3Sn)
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
The Common Crystal Structures:
Body-Centered Cubic (BCC)
•
Atoms at the corners of a cube plus one atom in the center
•
Common in
MSE 200A
Fall, 2008
– Is a Bravais lattice, but drawn with 2 atoms/cell to show
symmetry
– Bcc is not ideally close-packed
– Closest-packed direction: <111>
– Closest-packed plane: {110}
– Alkali metals (K, Na, Cs)
– Transition metals (Fe, Cr, V, Mo, Nb, Ta)
J.W. Morris, Jr.
University of California, Berkeley
The Face-Centered Cubic (fcc) and
Hexagonal Close-Packed (hcp) Structures
•
Fcc: atoms at the corners of the cube and in the center of each face
•
Hcp: close-packed hexagonal planes stacked to fit one another
– Is a Bravais lattice, but drawn with 4 atoms/cell to show symmetry
– Found in natural and noble metals: Al, Cu, Ag, Au, Pt, Pb
– Transition metals: Ni, Co, Pd, Ir
– Does not have a primitive cell (two atoms in primitive lattice of hexagon)
– Divalent solids: Be, Mg, Zn, Cd
– Transition metals and rare earths: Ti, Zr, Co, Gd, Hf, Rh, Os
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
fcc and hcp from Stacking
Close-Packed Planes
A
A
B
A
A
A
B
B
C
A
A
A
B
C
A
A
BB
C
A
A
C
C
→
C
A
B
A
AB
A
A
A
B
→
C
C
A
A
B
A
B
C
A
A
ABA = hcp
A
B
C
C
A
A
A
B
B
C
A
A
A
• There are two ways to stack spheres
• The sequence ABA creates hcp
• The sequence ABC creates fcc MSE 200A
Fall, 2008
The image
cannot be
displayed. Your
computer may
not have
enough
memory to
open the
image, or the
image may
ABC = fcc
J.W. Morris, Jr.
University of California, Berkeley
Hexagonal Close-Packed
MSE 200A
Fall, 2008
•
HCP does not have a primitive cell
•
Common in
•
Anisotropy limits engineering use of these elements
– 2 atoms in primitive cell of hexagonal lattice
– 6 atoms in cell drawn to show hexagonal symmetry
– Divalent elements: Be, Mg, Zn, Cd
– Transition metals and rare earths: Ti, Zr, Co, Gd, Hf, Rh, Os
J.W. Morris, Jr.
University of California, Berkeley
Face-Centered Cubic Structure
ABC stacking
Fcc cell
View along diagonal (<111>)
•
FCC is cubic stacking of close-packed planes ({111})
•
Common in
– 1 atom in primitive cell; 4 in cell with cubic symmetry
– <110> is close-packed direction
– Natural and noble metals: Cu, Ag, Au, Pt, Al, Pb
– Transition metals: Ni, Co, Pd, Ir
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Interstitial Sites:
Octahedral Voids in fcc
MSE 200A
Fall, 2008
•
Octahedral interstitial site is equidistant from six atoms
•
There are 4 octahedral voids per fcc cell
– “Octahedral void”
– Located at {1/2,1/2,1/2} and {1/2,0,0}
– One per atom
J.W. Morris, Jr.
University of California, Berkeley
Interstitial Sites:
Tetrahedral Voids in FCC
• Tetrahedral site is equidistant from four atoms
– “tetrahedral void”
– Located at {1/4,1/4,1/4} - center of cell octet
• There are 8 tetrahedral voids per fcc cell
– Two per atom
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Interstitial Sites:
Voids between Close-packed Planes
A
B
C
C
A
A
A
B
B
C
A
A
A
A
B
C
C
A
A
A
B
B
C
A
A
•
In both FCC and HCP packing:
•
Stacking including voids:
A
– Tetrahedral void above and below each atom (2 per atom)
– Octahedral void in third site between planes
– Fcc: ...(aAa)c(bBb)a(cCc)b(aAa)…
– Hcp: ...(aAa)c(bBb)c(aAa)… (octahedral voids all on c-sites)
⇒ Size and shape of voids are the same in fcc and hcp
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
The Diamond Cubic Structure
•
Fcc plus atoms in 1/2 of tetrahedral voids
•
DC is the structure of the Group IV elements
– Close-packed plane stacking is ...AaBbCc… or ... aAbBcC...
- Each atom has four neighbors in tetrahedral coordination
- Natural configuration for covalent bonding
– C, Si, Ge, Sn (grey)
– Are all semiconductors or insulators
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Solid Solutions and Compounds
• Solid solution
– Solute distributed through solid
- Substitutional: solutes on atom sites
- Interstitial: solutes in interstitial sites
- Ordinarily small solutes (C, N, O, …)
• Ordered solution (compound)
– Two or more atoms in regular pattern
(AxBy)
– Atoms may be substitutional or interstitial
on parent lattice
– “Compound” does not imply
distinguishable molecules
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Atomic Resolution Image of Gum Metal
• “Gum metal”: Ti-23Nb-0.7Ta-2Zr-1.2O
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Binary Compounds: Examples
• Substitutional:
– Bcc: CsCl
– Fcc: Cu3Au
• Interstitial:
–
–
–
–
MSE 200A
Fall, 2008
Fcc octahedral: NaCl
Fcc tetrahedral: ß-ZnS
Hcp tetrahedral: α-ZnS
Bcc tetrahedral: Nb3Sn (A15)
J.W. Morris, Jr.
University of California, Berkeley
BCC Substitutional: CsCl
• BCC parent
–
–
–
–
Stoichiometric formula AB
A-atoms on edges
B-atoms in centers
Either specie may be “A”
• Found in:
– Ionic solids (CsCl)
• Small size difference
• RB/RA > 0.732 to avoid like-ion
impingement
– Intermetallic compounds
• CuZn (ß-brass)
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
FCC Substitutional: Cu3Au
• FCC parent
– Stoichiometric formula A3B
– B-atoms on edges
– A-atoms on faces
• Found in:
– Intermetallic compounds (Cu3Au)
– As “sublattice” in complex ionics
• E.g., “perovskites”
– BaTiO3 (ferroelectric)
– YBa2Cu3O7 (superconductor)
• Lattices of + and - ions must
interpenetrate since like ions cannot
be neighbors
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
FCC Octahedral Interstitial: NaCl
• FCC parent
–
–
–
–
Stoichiometric formula AB
A-atoms on fcc sites
B-atoms in octahedral voids
Either can be “A”
• Found in:
– Ionic compounds:
• NaCl, MgO (RB/RA ~ 0.5)
• “Perovskites” (substitutional
ordering on both sites)
– Metallic compounds
• Carbonitrides (TiC, TiN, HfC)
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
FCC Tetrahedral Interstitial: ß-ZnS
• Binary analogue of DC
– Stoichiometric formula AB
– A-atoms on fcc sites
– B-atoms in 1/2 of tetrahedral voids
• AaBbCc
– Either element can be “A”
• Found in:
– Covalent compounds:
• GaAs, InSb, ß-ZnS, BN
– Ionic compounds:
• AgCl
• Large size difference (RB/RA < .414)
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Hcp Tetrahedral Interstitial: α-ZnS
• Hexagonal analogue of ß-ZnS
– Stoichiometric formula AB
– A-atoms on hcp sites
– B-atoms in 1/2 of tetrahedral voids
• AaBbAaBb
– Either element can be “A”
• Found in:
– Covalent compounds:
• ZnO, CdS, α-ZnS, “Lonsdalite” C
– Ionic compounds:
• Silver halides
• Large size difference (RB/RA < .414)
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Interstitial Sites:
“Octahedral” Voids in Bcc Crystals
•
Octahedral voids in face center and edge center
•
Octahedral voids in bcc are asymmetric
MSE 200A
Fall, 2008
– Located at {1/2,1/2,0} and {1/2,0,0}
– Each has a short axis parallel to cube edge (Ox, Oy, Oz)
– Total of six octahedral voids, three of each orientation
J.W. Morris, Jr.
University of California, Berkeley
Interstitial Sites:
“Tetrahedral” Voids in Bcc Crystals
•
Tetrahedral voids in each quadrant of each face
– Located at {1/2,1/4,0}
– 12/cell => 6/atom
•
MSE 200A
Fall, 2008
Tetrahedral voids in bcc are asymmetric
J.W. Morris, Jr.
University of California, Berkeley
Bcc Tetrahedral Interstitial: Α15
• Complex BCC derivative
– Stoichiometric formula A3B
– B-atoms on bcc sites
– A-atoms in 1/2 of tetrahedral voids
• Form “chains” in x, y, and z
• Found in:
– A15 compounds:
• Nb3Sn, Nb3Al, Nb3Ge, V3Ga
– These are the “type-II”
superconductors used for wire in
high-field magnets, etc.
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Description of
Complex Crystal Structures
• Most crystals can be referred to a close-packed frame
– Fcc or hcp network
– Possibly plus small distortions along symmetry axes
• Cubic → tetragonal (edge unique),
• Cubic → rhombohedral (diagonal unique)
• Atoms in ordered configurations in
– Substitutional sites
– Interstital sites: octahedral or tetrahedral
– Vacancies are useful as “atoms” to complete the configuration
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Hierarchical Description of
Complex Crystal Structures
•
Construct planar layers
•
Identify ordered pattern
•
Order layers
– Network (fcc or hcp)
– Interstitial planes that contain atoms
– Primary and interstitial planes
– Pattern is the same on all planes
– Including vacancies, if necessary, as species
– Physical pattern (fcc or hcp)
– Chemical pattern
• composition may change from layer to layer (differentiation)
– Stacking pattern is the same for network and interstitial layers
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Substitutional X-Compounds
• Undifferentiated
– All atoms are the same: fcc, hcp, polytypes (e.g., ABCBABCBA…)
• Differentiated
– Planes of atoms alternate: CuPt, WC
– Note that cubic symmetry is broken in CuPt: rhombohedral
^
^
^
^
^
MSE 200A
Fall, 2008
^
= Cu
=W
= Pt
=C
J.W. Morris, Jr.
University of California, Berkeley
Octahedral Interstital X-Compounds
•
Undifferentiated
•
Differentiated
= Na
= As
= Cl
= Ni
– Fcc or hcp planes alternate with filled octahedral planes: NaCl, NiAs
– Note that o-sites in NiAs are ccc, can tell which atom is in octahedral hole
– Alternate lattice or interstitial planes differ
– CdI2: like NiAs but octahedral Cd planes alternate with vacant planes
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Tetrahedral(I) X-compounds
= Zn
= Zn
=S
=S
• Lattice planes plus alternate planes of tetrahedral voids
• Examples:
– Unary: diamond cubic, hexagonal diamond (Lonsdaleite)
– Binary: α-ZnS, β-ZnS
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley
Tetrahedral(II) X-Compounds
= Ca
=F
• Lattice planes plus planes on both tetrahedral sites
• Fcc-based: CaF2 (flourite) and Li2O
• Hcp-based: none known
MSE 200A
Fall, 2008
J.W. Morris, Jr.
University of California, Berkeley