CHEM 109A CLAS 1. Predict the products of the following reactions
advertisement

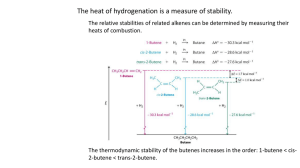
CHEM 109A CLAS Elimination Reactions - KEY 1. Predict the products of the following reactions (a-c E2, d-f E1 – KEY focuses only on elimination products, in most cases there will also be substitution products.) Br O -O a. Br O LG must be axial O HO Br- -O H are the same H -O b. Br H HO H -O Br- major P (E > Z and more substituted over less substituted alkene) Br -O c. Cl H HO -O Cl- major P (more substituted alkene) Cl LG must be axial H O d. I H H H I O H IH 3O+ major P Page 1 of 7 CHEM 109A CLAS Elimination Reactions - KEY Br H O e. H Br H Me shift H O H 2o carbocation 3o carbocation H 3O + Br- major P HO Br f. H Hydride shift Br H O H H 2O +CH 3 Br- major P 2. Which reactant in each of the following pairs will undergo an elimination reaction more rapidly? Explain your choice and show the mechanism. OH- OH Cl Br H 2O H 2O a. or E2 conditions and Br better LG so more rapid reaction. Page 2 of 7 CHEM 109A CLAS Elimination Reactions - KEY CH3O - CH3O - CH 3OH Br CH 3OH Br b. or E2 conditions and LG must be axial for E2 reaction. 3. For each of the following, give the major elimination product; if the product can exist as stereoisomers, indicate which stereoisomer is obtained in greater yield. a. (R)-2-bromohexane + high concentration HO- → H H -OH Br H Br- H 2O H trans is major P (cis and monosub alkene are minor) E2 b. (R)-2-bromohexane + H2O → H Br O H H H H H 3O + BrH trans is major P (cis and monosub alkene are minor) E1 c. trans-1-chloro-2-methylcyclohexane + high concentration CH3O- → Cl H Will remove H on C6 b/c it is axial -OCH 3 E2 d. trans-1-chloro-2-methylcyclohexane + methanol → Page 3 of 7 CHEM 109A CLAS Elimination Reactions - KEY H O Cl H Hydride shift H2O +CH3 Cl - E1 e. (R)-2-bromohexane + high concentration sodium tert-butoxide → Na+ H Br H H H H H H -O H H H H O (R)-2-bromohexane Na+ H Br- E2 f. (2-Bromo-3-methyl-butyl)-benzene/2-bromo-3-methyl-1-phenylbutane + high concentration methoxide → H Br -O CH3 H H H (2-Bromo-3-methyl-butyl)-benzene H O Br- CH 3 H H H major P (dbl bond is less substituted, conjugated with benzene ring) E2 Additional Information: Definitions Elimination rxn: in general, removal of H-X molecule and formation of double bond (alkene). A.K.A. β-elimination, 1,2-elimination Zaitsev’s (Saitseff’s) Rule: The more substituted alkene is obtained when a proton is removed from the β-C w/fewest Hs. EXCEPTIONS: If R has double bond or benzene ring, the conjugated alkene (NOT more sub) will be more stable/major P. If bulky B is used, alkene w/ bulky groups opposite each other (NOT more sub) will be the more stable/major P. Page 4 of 7 CHEM 109A CLAS Elimination Reactions - KEY E2 Mech: Bimolecular, 1 step/concerted reaction: B removes proton from β-C, e- go to form double bond and α-C – X bond breaks heterolytically so LG leaves as X-. Br H 2O Br- H H H -OH Rel Reactivity: Based on LG: RI > RBr > RCl > RF Based on R: 3o > 2o > 1o Regioselectivity: Usu. follows Zaitsev’s rule Br H -OH major P, more substituted alkene unless opportunity for formation of conjugated double bonds I major P, conjugated diene minor P, more substituted, but isolated diene Page 5 of 7 CHEM 109A CLAS Elimination Reactions - KEY major P, dbl bond conjugated w/ benzene ring Cl minor P, more substituted, but dbl bond isolated from benzene ring OR steric hinderence for B reacting w/ H on β-C (i.e. bulky base) Br major P, b/c less steric hinderence -O Tbl 9.1 If alkyl halide (R) is NOT sterically hindered and Base is… Little/Moderately hindered Very hindered More sub P Less sub P W/ alkyl fluoride major P is the less substituted alkene b/c F is such a poor LG that a neg charge develops on the α-C in the T.S. ~ carbanion (1o > 2o > 3o, carbanion stability b/c destabilized by e- donating alkyl groups). Stereoselectivity: Since elimination of H and X occurs in 1 step, the H-C and X-C bonds have to be in the same plane (a.k.a. periplanar) Syn-elimination (from eclipsed conformer) – “front attack” Anti-elimination (from staggered conformer) – “back attack”, favored b/c it is the more stable conformer & allows for back-side attack so base doesn’t feel the LG as much → get more E than Z. E1 Mech: 2-step rxn 1) LG leaves to form carbocation (r.d.s.) 2) B removes H from β-C (delocalization of e- b/c of hyperconjugation dec B.D.E. of H attached to sp3 C). H 3O+ Br Br- H O H H *WATCH for 1,2-hydride shifts, 1,2-methyl shifts (carbocation rearrangements) to form more stable carbocation before deprotonation. Page 6 of 7 CHEM 109A CLAS Elimination Reactions - KEY Rel Reactivity: Based on LG: RI > RBr > RCl > RF Based on R = rel stability of carbocations: 3o benzylic ~ 3o allylic > 2o benzylic ~ 2o allylic ~ 3o > 1o benzylic ~ 1o allylic ~ 2o > 1o > vinyl Regioselectivity: Same as E2 Stereoselectivity: W/ carbocation intermediate, both anti and syn elim possible → major P has bulkiest substituents opposite each other rel to the double bond. Cyclohexanes E2 – H and X must be axial (more stable conformer will NOT go E2) E1 – carbocation intermediate w/ possibility of rearrangement, usually follows Zaitsev’s Rule. Substitution and Elimination Compete So look at rxn conditions Tbl 9.4, 9.5and 9.6 Rxn Conditions Alkyl Halide 1o 2o 3o Rel Reactivity Stereochemistry SN1 vs. E1 SN2 vs. Poor Nuc/wk B N/A Favored Both Both Both N/A o o o o o o o 3 > 2 > 1 1 > 2o > 3o 3 >2 >1 Both E & Z (more w/ Inverted bulky Both R & S only groups opposite) E2 Strong Nuc/strong B W/ sterically hindered R or Nuc/B W/stronger B, bulkier B, higher T Only o 3 > 2o > 1o Both E & Z (more w/ bulky groups opposite, unless β-C bound to 1 H and then only single stereoisomer formed) Page 7 of 7