2054, Chap. 9, page 1 I. A. Overview of metabolism
advertisement

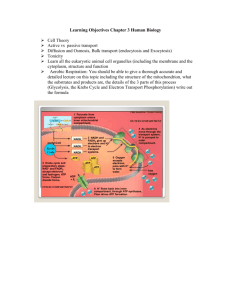
2054, Chap. 9, page 1 I. Metabolism: Energy Release and Conservation (Chapter 9) A. Overview of metabolism 1. metabolism = total of all chemical reactions in the cel 2. catabolism = molecules broken down into simpler molecules with the release of energy a. some energy trapped and made available for work (ATP) b. produces precursor compounds for biosynthesis (e.g., pyruvate, "-ketoglutarate) c. generates reducing equivalents for energy or biosynthesis (NADPH) 3. anabolism = synthesis of complex molecules from simpler ones with an input of energy 4. amphibolic pathways function catabolically and anabolically a. glycolysis and TCA cycle b. most reactions are reversible or other enzymes exist to bypass irreversible catabolic steps c. two separate enzymes are important for independent regulation 5. energy sources (Fig. 9.1) a. light (phototrophs) b. chemicals (1) organic (chemoorganotrophs) (2) inorganic (chemolithotrophs) 6. electron acceptors a. compound derived from the substrate (fermentation) b. O2 (aerobic respiration) c. other external electron acceptors (anaerobic respiration) 7. ATP generation a. substrate-level phosphorylation (SLP) is the main mechanism in fermentative microorganisms b. oxidative phosphorylation (OP) is the main mechanism in respiring microorganisms 8. catabolism can be broken down to 3 stages (Fig 9.3) a. stage 1 = large nutrient molecules are hydrolyzed to simpler molecules b. stage 2 = products of 1st stage degraded to simpler compounds that are key intermediates in amphibolic pathways (1) acetyl CoA, pyruvate, TCA intermediates (2) ATP, NADH, FADH2 2054, Chap. 9, page 2 c. stage 3 = molecules completely oxidized to CO2 in TCA cycle (1) high yield of energy (ATP, NADH, FADH2) (2) can use oxygen or other electron acceptor 9. substrate level phosphorylation = phosphorylation of ADP coupled to exergonic breakdown of substrate molecule B. Breakdown of glucose to pyruvate (Appendix II) 1. glycolysis (Emden-Meyerhoff pathway) (Fig. 9.5) a. most common pathway for glucose degradation to pyruvate (1) stage 2 of catabolism (2) functions in presence or absence of O2 (3) occur in all major groups of microorganisms b. can be divided into 2 parts, 6-carbon and 3-carbon (1) 6-C, glucose phosphorylated twice to fructose-1,6-bisphosphate (2) no energy yield; 2 ATP consumed (3) 3-C stage starts with cleavage of F-1,6-P (4) 2 ATP/3-C compound (= 4/glucose) (5) also generates 2 NADH c. 9 steps from glucose to pyruvate (1) activation of glucose; phosphorylated by ATP to G-6-P (2) G-6-P converted to isomer F-6-P (3) ATP phosphorylates to F-1,6-diphosphate (a) so far 2 ATP consumed (b) no energy released (c) no oxidation-reduction (4) F-1,6-DP split into glycerol-3-phosphate (G-3-P) and dihydroxyacetone phosphate (DHAP) (a) isomers (b) DHAP converted into G-3-P (5) G-3-P oxidized and phosphorylated (Pi) to diphosphoglyceric acid (DPGA) (a) NAD reduced to NADH (b) single oxidation-reduction step of glycolysis (c) aerobes use NADH for energy; anaerobes reoxidize it, using an organic 2054, Chap. 9, page 3 compound as the acceptor (6) DPGA becomes 3-phosphoglyceric acid; ADP phosphorylated to ATP (7) 3PG converted to 2PG (8) 2PG dehydrated to phosphoenolpyruvate (9) PEP dephosphorylated (ADP ÷ ATP), pyruvate formed 2. pentose phosphate or hexose monophosphate pathway = another pathway for sugar breakdown to pyruvate (Fig 9.6) a. can operate at same time as EMP pathway b. produces high levels of NADPH (for synthetic reactions) 3. Entner-Doudoroff pathway = another hexose to pyruvate pathway, substitutes for EMP pathway (Fig 9.8) a. generally in Pseudomonas, Rhizobium, Azotobacter, Agrobacterium, and a few other gram negative bugs b. rare in gram positives; Streptococcus faecalis C. Fermentation 1. fermentation = all metabolic processes that release energy from a sugar or other organic molecule, do not require oxygen or an electron transport system, and use an organic molecule as the final electron acceptor a. uses organic compounds (part of substrate) as terminal electron acceptors (1) substrate level phosphorylation (2) reducing equivalents recovered in organic endproducts b. yields small amount of ATP 2. other definitions in use a. any process that produces alcoholic beverages or acidic dairy products (general use) b. any spoilage of food by microorganisms (general use) c. any large-scale microbial process occurring with or without air (industrial definition) d. any energy-releasing metabolic process that takes place only under anaerobic conditions (oversimplification) 3. typical products of pyruvate fermentation a. alcohol (beer, wine, whiskey) b. solvents (acetone, butanol) c. organic acids (lactic, acetic) 2054, Chap. 9, page 4 d. dairy products, soy sauce D. tricarboxylic acid cycle 1. stage 3 of catabolism 2. pyruvate degraded to CO2 a. oxidized to CO2 and acetyl-CoA by pyruvate dehydrogenase complex b. acetyl-CoA = acetic acid and coenzyme A joined by high energy thiol ester bond c. acetyl-CoA arises from many other pathways 3. tricarboxylic acid (TCA) cycle or citric acid cycle a. substrate, acetyl CoA, condensed with 4 carbon intermediate, oxaloacetate, to form citrate (3o alcohol) and begin the 6 carbon stage b. citrate rearranged to give isocitrate, a more readily oxidized 2o alcohol c. isocitrate oxidized and decarboxylated to "KG d. "KG oxidized and decarboxylated to succinyl-CoA (1) cycle enters 4 carbon stage (2) two oxidation steps yield FADH2 and NADH for each acetyl-CoA added originally (3) GTP produced from succinyl-CoA by substrate level phosphorylation e. eventually oxaloacetate is reformed, ready for next cycle 4. overall, the TCA cycle generates 2 CO2, 3 NADH, 1 FADH2, and 1 GTP for each acetyl CoA oxidized 5. TCA cycle enzymes widely distributed among microorganisms a. even microorganisms that lack complete cycle b. one major function is to provide compounds for biosynthesis E. Electron transport 1. respiration = ATP-generating process in which molecules are oxidized and the final electron acceptor is (almost always) an inorganic molecule a. electron transport chain is an essential part of aerobic respiration (1) O2 is final electron acceptor aerobically (2) anaerobically, final electron acceptor is an inorganic molecule other than O2 or, rarely, an organic molecule (a) sulfate, nitrate, carbonate (b) fumarate b. electron transport chain = sequence of membrane associated electron carrier molecules 2054, Chap. 9, page 5 that are capable of oxidation and reduction (1) ETS have two basic functions (a) accept electrons from a donor and pass them to an acceptor (b) conserve some of the energy released during electron transport for ATP synthesis (2) electrons passed through the chain, in direction of higher reducing potential, with stepwise release of energy (3) energy released drives the chemiosmotic generation of ATP (4) located in plasma membrane of prokaryotes, mitochondrial membrane of eucaryotes c. several classes of carrier molecules (1) NADH dehydrogenases (transfer hydrogen atoms from NADH) (a) bound to inside of membrane (b) accept electrons from NADH (c) pass 2 hydrogens to flavoproteins (2) flavoproteins (a) contain flavins (derived from riboflavin, vitamin B2) in the prosthetic group (b) accept hydrogens, donate electrons (c) FMN, FAD (3) iron-sulfur proteins (nonheme iron-sulfur proteins) (a) several arrangements, Fe2S2 and Fe4S4 are most common (b) ferredoxin (Fe2S2) is common (c) reduction potentials vary, so they occur at different points of the ETS (d) carry electrons only (4) quinones (a) lipid-soluble, nonprotein carriers (b) Q (coenzyme Q) (c) some related to vitamin K (a growth factor for higher animals) (d) accept hydrogen, donate electrons (5) cytochromes (a) proteins containing an iron group in a porphyrin ring, in an oxidized (Fe3+) or reduced (Fe2+) form, called a heme (b) transfer single electrons 2054, Chap. 9, page 6 (c) several classes, differing in reduction potentials (d) cyt a, cyt a3, cyt b, cyt c, cyt c1 (e) can form tight complexes with other cytochromes or iron-sulfur proteins d. most information known about mitochondrial ETC (1) electrons transferred from NADH to FMN (2) FMNH2 passes electrons to Q and two H+ through the membrane (3) Q transports two H+ from inside to outside, passes electrons to cytochromes (4) typical order is cyt b, cyt c1, cyt c, cyt a, cyt a3 (5) cyt a3 reduces O2 to H2O (6) FADH2 can transfer electrons into the ETC at a lower level than NADH, so it produces only 2/3 as much energy for ATP generation (7) note that some carriers (cytochromes, iron-sulfur proteins) transport electrons only, while others (FMN, Q) transfer protons as well F. Oxidative phosphorylation 1. chemiosmotic hypothesis a. electron transport chain (ETC) functions as a proton pump (1) H+ + e- move from inside to outside of cell (2) e- transferred to carrier and moves back towards cytoplasm while H+ gets excreted (3) on cytoplasmic side, e- picks up H+ and moves to outer layer (4) overall, a proton gradient is formed (more outside), with potential energy as a proton motive force (PMF) 2. respiratory inhibitors block electron transport in ETC, preventing formation of PMF 3. uncouplers make membrane permeable to H+, collapsing the gradient G. Yield of ATP in glycolysis and aerobic respiration 1. glycolysis (EMP) yields 2 ATP (via SLP) 2. most fermentative pathways do not generate additional ATP a. purpose of fermentation is to oxidize NADH back to NAD b. pathways producing acetate or butyrate can produce additional ATP 3. respiration can yield a net of 38 ATP (Table 9.2) from glucose a. oxidation of 10 NADH for 30 ATP b. oxidation of 2 FADH2 for 4 ATP c. 4 ATP from SLP (2 from glycolysis and 2 from TCA cycle) 2054, Chap. 9, page 7 4. P/O ratio is the number of ATP formed per atom of oxygen reduced by 2 electrons in electron transport a. eukaryotes, P/O typically 3 for NADH and 2 for FADH2 b. P/O tends to be lower in bacteria 5. Pasteur effect = rate of sugar catabolism is greater under anaerobic than aerobic conditions H. anaerobic respiration = respiration in which electron acceptors other than O2 are used 1. typically NO3-, SO42-, CO32-, as well as some organics (e.g., fumarate) 2. less energy generated than with O2, due to lower potentials I. chemolithotrophy = energy generation from inorganic molecules 1. lithothroph = rock eating 2. typically H2, H2S, NH3 3. usually involves aerobic respiratory processes 4. possess ETS components and generate a PMF 5. most are autotrophs (use CO2 as carbon source)(vs. heterotrophs, which use organic compounds as their source of C) J. phototrophy 1. photosynthesis = conversion of light energy into chemical energy a. carbon fixation = synthesis of sugars from CO2 b. electrons come from hydrogens in water 2. light reactions = light energy converts ADP to ATP (photophosphorylation) and NADP is reduced to NADPH 3. dark reactions = electrons and energy used to reduce CO2 to sugar 4. light energy is absorbed by chlorophyll molecules a. chlorophyll a used by green plants, algae, cyanotobacteria (1) membranous thylakoids in chloroplasts of plants, algae (2) thylakoids in cyanobacteria b. bacteriochlorophylls used by other bacteria c. bacteriorhodopsin = pigment in Halobacterium 5. excited electrons jump from chlorophyll through an electron transport chain a. similar to respiration b. ADP converted to ATP by chemiosmosis 6. green and purple bacteria are anoxygenic 2054, Chap. 9, page 8 a. use H2, H2S, So, reduced organics as electron source b. can also grow chemotrophically in dark K. catabolism of carbohydrates, lipids, and proteins 1. carbohydrates are hydrolyzed and/or converted to glucose and fed into a glycolytic pathway 2. lipids a. glycerol moieties enter glycolytic pathway b. fatty acids $-oxidized to acetyl CoA, which is fed into TCA cycle or used for biosynthesis 3. proteins are hydrolyzed external to plasma membrane; amino acids enter cell by specific transport mechanisms a. amino acids are deaminated b. resulting organic acid converted to pyruvate, acetyl CoA, or a TCA intermediate