31 Atomic Physics Outline
advertisement

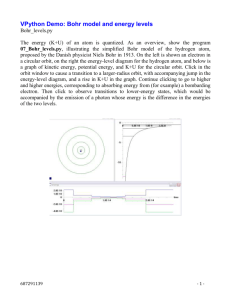
31 Atomic Physics Outline 31-1 Early Models of the Atom 31 2 31-2 The Spectrum of Atomic Hydrogen 31-3 Bohr’s Model of the Hydrogen Atom 31-3 Bohr’s Model of the Hydrogen Atom Bohr′s model of Hydrogen Atom n = 1, 2, 3, ….. Figure 31-7. The First Three Bohr Orbits 31-3 Bohr’s Model of the Hydrogen Atom The spectrum of Hydrogen How to create spectrum? • Electron jumps from one orbit to the another: it gives up or absorbs photons (as a result of energy difference) difference). • Different orbit has different energy level. • The p photon wavelength g is determined by y hf. Electron Energies include: • Electric Potential energy. • Kinetic energy. 31-3 Bohr’s Model of the Hydrogen Atom Assumptions of the Bohr model: • The electron in a hydrogen atom moves in a circular orbit around the nucleus. • Only certain orbits are allowed, where the angular momentum in the nth allowed orbit is • Electrons in allowed orbits do not radiate photons. Radiation is emitted when an electron changes from one orbit to another, with frequency given by the energy difference: 31-3 Bohr’s Model of the Hydrogen Atom In order for an electron to move in a circle of radius r at speed v, the electrostatic force must provide the 2 2 mv e required centripetal force. Hence: ( = k 2 ) r 2 e mv 2 = k rn r 31 − 3 Adding the angular momentum requirement gives: According Eq.11 − 12 L = rmv, mrn vn = nh / 2π , nh vn = , n = 1, 2,3,... 2π mrn 31 − 4 31-3 Bohr’s Model of the Hydrogen Atom Solving for rn and vn from these two equations, the allowed radii are: h2 2 rn = ( 2 n ) , n = 1,, 2,3,... , , 2 4π mke k 31 − 5 The speeds at the allowed radii are: 2π ke vn = , n = 1, 2,3,... nh 2 31 − 6 Example p 1 Find the radius of the Hydrogen at the smallest orbit (Bohr Orbit). Solution: h2 2 ) , rn = ( 2 n 2 4π mke with n = 1 (6.626 ×10−34 ) 2 2 ) 1 =( 2 × 4π (9.109 ×10−31 kg )(8.988 ×109 N .m 2 / C 2 )(1.602 ×10−19 ) 2 = 5.29 × 10−11 m Examples Find the speed and kinetic energy of the electron in the Second Bohr orbit (n=2). Solution: 2π ke k vn = with n=2 , nh 2π (8.99 ×109 N ⋅ m 2 / C 2 )(1.60 ×10−19 ) 2 v2 = 2 × (6.63 ×10−34 J .s ) 2 = 1.09 × 10−6 m / s 1) Speed 2π ke vn = , with n=2 nh 2π (8.99 ×109 N ⋅ m 2 / C 2 )(1.60 ×10−19 ) 2 v2 = 2 × (6.63 (6 63 ×10−34 J .s ) 2 = 1.09 × 10−6 m / s 2) Kinetic energy 1 2 K 2 = mv2 2 1 = (9.11 (9 11×10−31 kg k )(1.09 )(1 09 × 106 m / s ) 2 = 55.4 4 × 10−19 J 2 31-3 Bohr’s Model of the Hydrogen Atom The total mechanical energy of an electron in a Bohr orbit is the sum of its kinetic and potential energies. After some algebraic manipulation, and substituting known values of constants, we find for hydrogen atom: 1 En = −(13.6 ⋅ eV ) 2 , n = 1, 2,3,... n 1 eV = 1.60x10-19 Joule The lowest energy is called the ground state. Most atoms at room temperature are in i the h groundd state. The Origin of Spectral Series in Hydrogen: Lyman Series Figure 31-9a The Origin of Spectral Series in Hydrogen: Balmer Series Figure 31-9b The Origin of Spectral Series in Hydrogen: Paschen Series Figure 31-9c Active Exam 31-2 Absorbing a photon and what is the wavelength? Find the wavelength of a photon that will be absorded, if a electron i tto raise is i iin a h hydrogen d atom t ffrom th the n=3 3 state t t tto the th n=5 5 state. t t Solution: 1) The energy at n=5 state is 1 1 = − (13 (13.6 6 ⋅ eV ) = −0.544 0 544 eV 2 2 n 5 = −0.544 ×1.6 × 10−19 J = −0.87 × 10−20 J E5 = −(13.6 (13 6 ⋅ eV ) 2) Th The energy att n=3 3 state t t is i E3 == −(13.6 ⋅ eV ) 1 = −2.42 × 10−19 J 2 5 3) The energy difference is ΔE5,3 = 1.55 × 10−19 J 4) The wavelength is hf = ΔE5,3 = 1.55 × 10−19 J ⇒ f = 2.34 × 1014 Hz f λ = c ⇒ λ = 1.28 × 10−6 m Chapter Summary The total mechanical energy of an electron in a Bohr orbit is the sum of its kinetic and potential energies. For Hydrogen atom, the total energy is 1 En = −(13.6 (13 6 ⋅ eV ) 2 , n = 11, 22,3,... 3 n