BThayerTalk4 - FSU High Energy Physics
advertisement

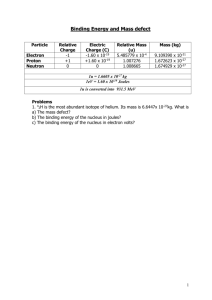
Basic Concepts of Nuclear Physics by Benjamin Thayer Contents Binding Energy Mass Energy Equivalence Binding Energy vs. Atomic mass Applications: Fission and Fusion Binding Energy Binding energy is the energy that is lost when a nucleus is created from protons and neutrons. Binding energy speaks of an nucleus’ stability The greater the binding nrg, the more stabile. Binding Energy Cont. If you added up the mass of the proton and neutrons seperately and then added them up together collectively (i.e. the mass of the nucleus,) you would find their sum is not equal. This difference in mass, together with Einstein’s mass energy relationship, defines the magnitude of the binding energy. Nuclear binding energy = change (m c^2) The Nuclear Binding Energy Curve One of the most important graphs in all physics!!! Definition: tightly bound = less mass per neucleon Applications: Fission and Fusion Applications Cont. The yields of uranium fission and tritiumdeuterium fusion