Bacillus bingmayongensis sp. nov., isolated from the pit
advertisement

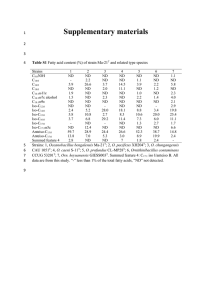
Bacillus bingmayongensis sp. nov. 1 2 1 Bacillus bingmayongensis sp. nov., isolated from the pit soil of Emperor Qin's Terracotta Warriors in China 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 Bo Liu1, Guohong Liu1,2, Naiquan Lin2 , Jianyang Tang1, Yingzhi Lin1, 44 3 g beef extract and 18 g agar (pH 7.0), with 0.5% NaCl solution and incubated at 30 ℃ for 48 h. 45 The isolated strains were subcultured several times to obtain a purified culture, and were then 1 2 Agricultural Bio-resource Institute, Fujian Academy of Agricultural Sciences, Fuzhou, Fujian 350003; PR China. Biological Control Institute, Fujian Agricultural and Forest University, Fuzhou, Fujian 350002, PR China. Author for correspondence: Bo Liu. Tel: + 86 591 87884601. Fax: +86 591 87884262. e-mail: fzliubo@163.com A slightly-halophilic endospore-forming bacterium is isolated from the No.1 pit soil of Emperor Qin's Terracotta Warriors in Xi’an City, Shanxi Province, China. The novel isolate FJAT-13831T is rod-shaped, motile, Gram-stain-positive, catalase- and oxidase-positive, grew aerobically at 0–5% NaCl (w/v) (optimum 2%), 15-45 º, (optimum 35 º5), pH 4.0-10.0 (optimum pH 7.0) and produced acid from various sugars. The colony was dull white, rough, flat, circular in the nutrient agar (NA). The DNA G+C content is 36.5 mol%. Cell wall peptidoglycan contains meso-diaminopimelic acid. The predominant menaquinone is MK-7; MK-5 and MK-4 are a minor component. The major fatty acids are iso-C15:0, iso-C17:0, C16:0, iso-C13:0, anteiso-C15:0, iso-C17:1 ω 5c with values of 21.03%, 11.49%, 9.83%, 7.66%, 7.39%, 5.12%, respectively. Phylogenetic analyses based on both 16S rRNA and gyrB (DNA gyrase B subunit gene) gene sequences showed that the novel isolate FJAT-13831T falls into the genus Bacillus cluster, validated by significant bootstrap values. The similarity of 16S rDNA between the novel isolate FJAT-13831T and the most closely reference species Bacillus pseudomycoides DSM12442T in the cluster was 99.72%, that of the gyrB gene was 93.8%, that of ANI (the average nucleotide identity) of 2881 core genes was 91.47%. The DNA–DNA hybridization value between the novel isolate FJAT-13831T and phylogenetically related speceis of Bacillus pseudomycoides DSM12442T was 69.1%. less than 70%, indicating that the novel isolate FJAT-13831T represents a distinct species. On the basis of these results, the novel isolate FJAT-13831T is reported as a novel Bacillus species. The name Bacillus bingmayongensis sp. nov. is proposed for this organism. The type strain is FJAT-13831T ( = CGMCC 1.12043T = DSM 25427T). The genus Bacillus consisted of aerobic, facultatively anaerobic, Gram-positive, spore-forming, or rod-shaped bacterium that had a wide range of physiological adaptations to the harsh environments, such as in desert sands (Zhang et al., 2011), hot springs (Nazina et al., 2004), forest soils (Chen et al., 2011), freshwater (Baik et al., 2010), marine sediments (Jung et al., 2011) and ancient tomb (Gatson et al., 2006). Liu et al. (2012) reported the phylogenetic analysis of Bacillus species isolated from No.1 pit of the First Emperor Qin's Terracotta Warriors in Xi’an City, Shanxi Province, China, finding a strain FJAT-13831T to be possible a new Bacillus species. Thus, this paper dealt with the taxonomic characterization of a novel Bacillus strain FJAT-13831T isolated soil samples existed in situ of the ancient mausoleum more than 2,222 years (i.e., The First Emperor of Qin Shi Huang, 259-210 BC). The detail researches will be summaried as follow. The strain FJAT-13831T was isolated from the soil sample in No.1 pit, using the dilution plating technique on a solid medium of nutrient agar (NA) ((NA; Atlas, 1993), containing (L-1) 10 g peptone, The GenBank/EMBL/DDBJ accession number for the 16S rRNA gene sequence of strain FJAT-13831T was JN 885201, and the accession numbers of the gyrB gene sequences of this strain was JN874726. 1 Bo Liu and others 46 further characterized. Strains were cultured routinely on nutrient agar (NA) under identical 47 conditions and stored in a deep freezer (-80 ℃) with 20% (v/v) glycerol suspensions. The 48 49 50 51 52 53 54 55 reference strains were Bacillus pseudomycoides DSM12442T, Bacillus cereus DSM31T and Bacillus mycoides DSM2408T, from DSMZ (Deutsche Sammlung von Mikroorganismen und Zellkulturen, Braunschweig, Germany). The biomass for chemotaxonomic and molecular systematic studies was prepared to incubate the strains in shake flasks of nutrient broth (NB, containing (L-1) 10 g peptone, 3 g beef extract, pH 7.0). The cells were harvested by centrifugation, washed with double distilled water (ddH2O) before using for further studies. 56 the novel isolate and other three reference strains were observed by microscop on NA at 30 ℃ 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 72 73 for 48 h. The cell morphology of tested strains was examined by a scanning electron microscope (SEM, JSM-6380; Jeol, Japan) with the cells fixed in a 2.5% paraformaldehyde/ glutaraldehyde mixture as well as coated with gold in a sputter coater (Polaron SC502 Siemens Simatic, Japan). The colony and cell photographs were demonstrated in Figs. 1 and 2, respectively. It was obvious that the colonial morphologies showed significantly differences among the tested strains, e.g. B. bingmayongensis FJAT-13831T with greyish-white in colour, irregular round with undulate margins (Fig. 1a), B. pseudomycoides DSM12442T with pale yellow in colour and irregularly shape with branching margins (Fig. 1b), B. mycoides DSM2408T with pale yellow in colour and irregular round with dentate margins (Fig. 1c), B. cereus DSM31T with pale yellow in colour and irregular round with smooth margins (Fig. 1d). The colony of the novel isolate grew more slowly than any of the reference strains with the shapes of colonies identified each other obviously. Furthermore, the scanning electron micrographs of cell morphologies of tested strains displayed greatly diversity from which it was easy to distinguish one another (Fig. 2a, 2b, 2c and 2d). Because of colony morphotypes and growth conditions affecting colony morphology, the colony morphology was not a reliable attribute to be used to speciate members of the genus Bacillus. The physiological and molecular characteristics were needed for further analysis. For analysis on colony and cell morphology of the tested Bacillus strains, the colonial properties of 1a 2 Bacillus bingmayongensis FJAT-13831T 1b Bacillus pseudomycoides DSM 12442T Bacillus bingmayongensis sp. nov. Fig 1 1c Bacillus mycoides DSM 2408T 1d Bacillus cereus DSM 31T Colonny pictures of four Bacillus strains cultured on a solid medium of nutrient agar (NA) ( Seveno et al., 2001) incubated at 30 ℃ for 48 h. e.g. Bacillus bingmayongensis FJAT-13831T with greyish-white in colour, irregular round with undulate margins (1a), Bacillus pseudomycoides DSM12442T with pale yellow in colour and irregularly shape with branching margins (1b), Bacillus mycoides DSM2408T with pale yellow in colour and irregular round with dentate margins (1c), Bacillus cereus DSM31T pale yellow in colour and irregular round with smooth margins (1d). 74 2a Bacillus bingmayongensis FJAT-13831T 2b Bacillus pseudomycoides DSM 12442T 2c Bacillus mycoides DSM 2408T 2d Bacillus cereus DSM 31T Fig 2 Cell pictures of four Bacillus strains e.g. Bacillus bingmayongensis FJAT-13831T(2a), Bacillus pseudomycoides DSM12442T(2b), Bacillus mycoides DSM2408T (2c) and Bacillus cereus DSM31T (2d) in tests. 75 76 For determination of optimum physical and chemical conditions, the strains were cultured in 77 nutrient agar (NA) for 72 h at temperatures ranging from 5 to 50 ℃ with 5 ℃ unit increment, in 78 79 80 81 NaCl concentrations ranging from 0 to 8% with interval 2% (w/v), at pH conditions ranging from 4 to 10 with 1 pH unit increment. The physiological and biochemical characterizations, e.g., Gram-staining, spore test, indole production, Voges-Proskauer, oxidase, catalase, urease, DNase activity, nitrate reduction, hydrolysis of starch, gelatin, arginine dihydrolase, lysine decarboxylase, 3 Bo Liu and others 82 83 84 85 86 87 ornithine decarboxylase, the utilization of Koser citrate broth, Triple Sugar Iron and KCN were performed under the identical conditions of growth temperature and culture medium were assessed, according to the standard procedures (Gregersen,1978; Smibert & Krieg, 1994). Acid production profiles from carbohydrates were obtained with the API 50 CH system (bioMérieux) after growth in 50 CHB medium as described by Logan & Berkeley (1984). The results of the novel isolate FJAT-13831T and Bacillus reference strains were compared in Table 1. About 18 88 characteristics marked with ( ) symbol for the novel isolate were differed from that for the most 89 90 91 closely species B. pseudomycoides DSM 12442T, for instance, the adaptation of high temperature, tolerances of NaCl or pH for B. bingmayongensis FJAT-13831T was 45oC, 4% NaCl or pH 10, respectively, quite different from that of B. pseudomycoides DSM12442T with 40oC, 2% NaCl or 92 pH 9. Acid productions marked with ( ) symbol from D-lactose, D-glucose, D-fructose, Erythritol, 93 94 95 96 97 98 99 100 101 102 103 D-saccharse, D-turanose and potassium gluconate of B. binamayongensis FJAT-13831T were also shown the difference with that of B. pseudomycoides DSM 12442T (Table 1). Based on the biological, physiological and biochemical characteristics, Bacillus species could be preliminarily identified from each other (Priest et al.,1988). ※ ※ Table 1. Investigations on the biological, physiological, biochemical characteristics and DNA G+C content among the novel isolate FJAT-13831T (Bacillus bingmayongensis sp. nov.) and the reference strains of Bacillus species. The characteristics marked with “※”were different between Bacillus bingmayongensis FJAT-13831T and Bacillus pseudomycoides DSM12442T, that with “†” meant the data for the type strains of Bacillus cereus, Bacillus mycoides, Bacillus pseudomycoides were obtained from “Bergy’s Mannual of Systematic Bacteriology” second edition. +, growth; -, no growth; w, weak growth. Bacillus Bacillus Bacillus Bacillus bingmayongensis pseudomycoides mycoides cereus Characteristics FJAT-13831T DSM12442T DSM2408T DSM31T Growth conditions ※ + + + Aerobic growth Temperature for growth (ºC) 5 10 15 20 30 35 40 45 ※ 50 Growth in NaCl 0 2% 4% ※ 6% ※ 8% pH value for growth 4 5 6 7 8 9 4 ※ + + + + + + + + + + - + + + + + + + - + + + + + + + - - - + + + + + + W + + + + + + W - + + - - - + + - - - + + + + + + + + + + + + + + + + + + Bacillus bingmayongensis sp. nov. Characteristics 10 Bacillus bingmayongensis FJAT-13831T Bacillus pseudomycoides DSM12442T Bacillus mycoides DSM2408T Bacillus cereus DSM31T + - + + + - - + + - + + + + + - + + + - + ※ Oxidase ※ Catalase ※ Acetoin production (V.P.) Enzyme production: DNase activity Arginine dihydrolase ONPG (b-Galactosidase) Ornithine decarboxylase Lysine decarboxylase Urease activity Hydrolysis of Gelatin ※ Starch Esculine Utilization of ※ - + + + + + + + + - - + ※ Triple Sugar Iron + - - - KCN growth H2S production Indole production + - + + - - Koser citrate broth Nitrate reduction ※ Acid production from (using API CH50) ※ D-lactose ※ D-glucose ※ D-fructose ※ D-saccharose D-turanose ※ D-arabinose L-arabinose D-ribose D-xylose L-xylose D-galactose D-mannose L-sorbose L-rhamnose D-cellobiose D-maltose D-melibiose D-trehalose D-melezitose D-raffinose Glycogene Gentiobiose D-tagatose D-fucose L-fucose ※ Erythritol - + - - + - + + + - + + + - + + + - - - + + + + + + + + + + + - + + + + - + + + + + + 5 Bo Liu and others Characteristics Glycerol Dulcitol Inositol Xylitol D-mannitol D-sorbitol D-arabitol L- arabitol Potassium gluconate ※ Amygdaline Arbutine Inulin Salicine N-acetylglucosamine Methyl-βD-xylopyranoside Methyl-αD-mannopyranoside Methyl-αD-glucopyranoside Potassium 2-cetogluconate Potassium 5-cetogluconate DNA G+C content (mol%)(Tm) ※ † Bacillus bingmayongensis FJAT-13831T + + Bacillus pseudomycoides DSM12442T + - Bacillus mycoides DSM2408T - Bacillus cereus DSM31T + - + + - + + - + + + - + + + + - 36.5 34.0–36.0 34.2 35.7 104 105 106 107 For detection of DNA base composition, the G + C content of the DNA was determined from the midpoint value of the thermal denaturation profile (De Ley, 1970) obtained with a model UV-Vis 5515 spectrophotometer (Perkin-Elmer) at 260 nm; this instrument was programmed for 108 temperature increases of 1.0 ℃ min-1. The G+C content was calculated from the thermal 109 110 111 112 113 114 115 116 117 118 denaturation temperature with the equation of Owen & Hill (1979). The result showed that the DNA G+C content of the novel isolate FJAT-13831T was 36.5 mol% (Table 1), comparing to the contents that existed the B. cereus group with the ranges of 31.7-40.1 mol% (Priest et al., 1988). It was clear that the taxonomic position of the novel isolate corresponding to the member of this group in the genus Bacillus. 119 (pH 7.0) at 28 ℃ for 24 h. Extracts were analyzed using a gas chromatograph (Agilent 7890N) 120 and identified using the Microbial Identification Sherlock software package. All tested strains 121 exhibited typical fatty acid profiles for the genus Bacillus(showed in Table 3), with a lot of 122 123 124 125 126 127 128 129 branched chain components (Kaneda, 1977). Kämpfer (1994) specified fatty acid profiles of members in the genus Bacillus, containing amounts of iso-C15:0, iso-C17:0, C16:0, iso-C13:0, anteiso-C15:0, iso-C17:1 ω5c and low amounts of unsaturated fatty acids (<3%). Fatty acid profiles of the tested strains complied with this profile. Major fatty acids of iso-C15:0, iso-C17:0, C16:0, iso-C13:0, anteiso-C15:0, iso-C17:1 ω 5c for the novel isolate FJAT-13831T were 21.03%, 11.49%, 9.83%, 7.66%, 7.39%, 5.12%, that for B. pseudomycoides DSM 12442T were 15.26%, 14.04%, 10.49%, 7.75%, 3.91%, 3.08%, that for B. cereus DSM 31T were 29.19%, 11.84%, 6.11%, 6.62%, 4.40%, 5.53%, that for B. mycoides DSM 2048T were 15.95%, 10.09%, 11.01%, 10.36%, 4.13%, For testing cellular fatty acid profiles, the novel isolate and several Bacillus reference species were subjected to cellular fatty acid methyl ester analysis to confirm the genus classification. Fatty acids were extracted and analysed according to the standard protocol of the Microbial Identification System (Sherlock Microbial Identification System; MIDI) (Sasser, 1990) with cells grown on TSA 6 Bacillus bingmayongensis sp. nov. 130 131 132 133 134 2.36%. In this study, Bacillus species can be easily differentiated from one another based on amounts of fatty acid profiles. Table 3 Cellular fatty acid composition of the novel isolate FJAT-13831T and the closely related reference species from genus Bacillus B. bingmayongensis sp. B. pseudomycoides B. cereus B. mycoides nov. FJAT-13831T DSM 12442T DSM 31T DSM 2048T Fatty acid (%) mean SE mean SE mean SE mean SE iso-C15:0 iso-C17:0 C16:0 iso-C13:0 anteiso-C15:0 iso-C17:1 ω5c C14:0 iso-C16:0 Iso-C14:0 anteiso-C17:0 anteiso-C13:0 C18:0 anteiso-C17:1 a Iso-C12:0 C15:0 2OH C16:1 ω11c alcohol-C16:1 ω7c iso-C17:1 ω10c 21.03 11.49 9.83 7.66 7.39 5.12 4.13 3.62 2.86 2.84 2.23 1.68 0 0 0 0 0 0 0.24 0.22 0.31 0.17 0.24 0.17 0.72 0 0.07 0.17 0.12 0.27 0 0 0 0 0 0 15.26 14.04 10.49 7.75 3.91 3.08 2.17 8.37 3.10 3.35 4.37 0 1.15 4.87 0 0 0 0 1.84 0.85 1.30 0.67 0.54 0.84 0.14 1.19 0.37 0.54 0.63 0 0.05 0.50 0 0 0 0 29.19 11.84 6.11 6.62 4.40 5.53 2.38 5.99 2.97 2.11 0 0 1.06 0 1.17 0 0 4.61 0.96 0.91 0.49 0.28 0.67 0.49 0.07 0.26 0.15 0.13 0 0 0.04 0 0.12 0 0 0.31 15.95 10.09 11.01 10.36 4.13 2.36 2.91 6.67 3.03 1.95 2.11 1.38 0 1.10 1.20 2.06 1.72 9.82 0.58 0.18 0.25 0.29 0.35 0.08 0.02 0.07 0.06 0.08 0.18 0.42 0 0.06 0.14 0.05 0.03 0.37 135 136 137 138 139 140 141 142 143 For detection of cell wall composition, analysis of the cell wall peptidoglycan was performed by DSMZ. Respiratory quinones were also examined by DSMZ as described previously (Groth et al., 1996) using TLC and HPLC. The cell-wall peptidoglycan contained meso-diaminopimelic acid as the diagnostic cell-wall diamino acid, and also contained alanine and glutamic acid. The menaquinone composition of the novel isolate FJAT-13831T is the following: MK-7, MK-5, MK-4 (ratio of peak areas: 89:8:2, respectively); this was in accordance with all other members of the genus Bacillus (Shida et al., 1997). 145 146 147 148 149 150 151 152 153 154 155 156 157 158 For the phylogenetic and genetic analyses, genomic DNA was extracted using standard methods (Sambrook et al., 1989) . The 16S rRNA and gyrB genes were PCR-amplified with the universal primer sets described byYoon et al. (1997) and Yamamoto & Harayama (1995) respectively, and sequenced by Beijing Genomics Institute, China. The identity of a given PCR product was verified by bidirectional sequencing analysis. The phylogenetic relationships of the microorganisms examined in this study were determined by comparing individual 16S rRNA or gyrB gene sequences with sequences in the public databases using the BLAST algorithm (Altschul et al., 1990). The construction of phylogenetic trees by the neighbour-joining method (Saitou & Nei, 1987) were performed using the Mega5 soft (Tamura et al., 2011). Alignment gaps, primer regions for PCR amplification and unidentified base positions were not taken into consideration in the calculations. The topological robustness of the phylogenetic trees was evaluated by a bootstrap analysis involving 1000 replications (Felsenstein, 1985). A phylogenetic tree based on the 16S rRNA gene (Fig. 3) showed that the novel isolates clustered with members of the genus Bacillus, the nearest neighbour being B. pseudomycoides DSM 12442T (99.72% sequence similarity). Since several reports have been published showing that strains with>99% 16S rRNA 7 Bo Liu and others 159 160 161 162 163 164 165 166 167 168 169 170 171 172 173 gene sequence similarity may not belong to the same species (La Duc et al., 2004a; Satomi et al., 2002; Venkateswaran et al., 1999; Stackebrandt & Goebel, 1994), comparative gyrB gene sequence analyses were carried out to reveal that the closest phylogenetic similarity for the novel isolate FJAT-13831T was B. pseudomycoides DSM 12442T (93.8%). As had been observed in previous studies (La Duc et al., 2004b) the gyrB gene sequence-based phylogenetic topology proved more highly discriminative, grouping these strains monophyletically in a cluster separate from B. pseudomycoides DSM 12442T, clearly delineating them as a distinct species (Fig. 4). The sequence similarity values required to separate species on the basis of the gyrB gene vary according to the genus (Venkateswaran et al., 1998; Satomi et al., 2002, 2003, 2004). Additional reputable genetic analyses are therefore necessary to confirm the novelty of these isolates. Yet, bacterial strains with a difference in gyrB gene sequence of less than 5% cannot be allocated to the same species without support from DNA–DNA hybridization experiments (Stackebrandt & Ebers, 2006). T 62 Bacillus cereus ATCC 14579 (AE016877) Bacillus thuringiensis ATCC 10792T(ACNF01000156) 39 71 Bacillus anthracis ATCC 14578T (AB190217) Bacillus weihenstephanensis KBAB4(CP000903) 99 Bacillus mycoides DSM 2048T(ACMU01000002) 100 Bacillus pseudomycoides DSM 12442T(ACMX01000133) 73 71 Bacillus bingmayongensis FJAT-13831T (JN 885201) Bacillus megaterium DSM 319T (NC 014103) Bacillus aquimaris TF-12T(AF483625) Lysinibacillus sphaericus C3-41 (CP000817) 100 174 175 176 177 178 Lysinibacillus fusiformis B14905 (NZ AAXV01000000) 0.005 Fig. 3. Phylogenetic tree of members of the genus Bacillus, based on 16S rRNA gene sequences. The tree was constructed using the neighbour-joining method, and genetic distances were computed by using Jukes-Cantor model. Numbers at nodes indicate percen-tages of occurrence in 1000 bootstrapped trees. Lysinbacillus species was used as the outgroup. Accession numbers are given in parentheses. Bar, genetic distance of 0.005. Bacillus cereus ATCC 14579T (NC_004722) 77 Bacillus thuringiensis ATCC 10792T (FR850503) 63 Bacillus anthracis ATCC14578T (NC_003997.3) 84 Bacillus mycoides DSM 2048T(ACMU01000094) 96 100 Bacillus weihenstephanensis KBAB4 (CP000903) Bacillus bingmayongensis FJAT-13831T(JN874726) 93 Bacillus pseudomycoides DSM 12442T (CM000740) Bacillus aquimaris SG-1 (NZ ABCF01000000) 63 Bacillus megaterium DSM 319T (NC 014103) Lysinibacillus sphaericus C3-41 (CP000817) 100 179 180 Lysinibacillus fusiformis B14905 (NZ AAXV01000000) 0.02 Fig. 4. Phylogenetic tree of the novel isolates FJAT-13831T, based on gyrB gene sequences. The tree was 8 Bacillus bingmayongensis sp. nov. 181 182 183 184 185 186 187 188 189 190 constructed using the neighbour-joining method, and genetic distances were computed by using Jukes-Cantor model. Numbers at nodes indicate percen-tages of occurrence in 1000 bootstrapped trees. The reference strains of Lysinbacillus species served as the outgroup. Accession numbers are given in parentheses. Bar, genetic distance of 0.02. Table 2 Relateness values of 16S rRNA gene, gyrB gene and DNA-DNA hybridization between the novel isolate FJAT-13831T and the reference Bacillus species Relateness values (%) between Bacillus bingmayongensis FJAT-13831T and the reference Bacillus species Species gyrB 16S rRNA DNA-DNA relateness Bacillus bingmayongensis FJAT-13831T 100.00 100.0 99.8 Bacillus pseudomycoides DSM 12442T 99.72 93.8 69.1 Bacillus mycoides DSM 2048T 99.24 86.4 63.7 T Bacillus cereus DSM31 99.44 84.7 62.4 Bacillus thuringiensis ATCC 10792T 99.17 84.7 ND* Bacillus weihenstephanensis KBAB4 99.17 87.0 ND* Bacillus anthracis ATCC 14578 99.58 84.1 ND* Lysinibacillus fusiformis 93.47 73.4 ND* Bacillus megaterium DSM 319T 94.79 73.2 53.9 Lysinibacillus sphaericus 93.68 71.6 52.8 Bacillus aquimaris DSM16205T 95.27 71.5 52.7 *ND, not done. In the present study, DNA–DNA hybridization was performed using fluorometric method as 191 described by Gonzalez et al. 2005. A hybridization temperature of 62 ℃ (calculated with 192 193 194 195 196 197 198 199 200 201 202 203 204 205 206 207 208 209 210 211 212 213 215 216 correction for the presence 50% formamide) was used. An overview of DNA–DNA hybridization relatedness values between the strains was given in Table 2. DNA–DNA hybridization relatedness values between the novel isolate FJAT-13831T and the closest reference strain, B. pseudomycoides DSM 12442T, was 69.1%, which was below the 70% cut-off value recommended by Wayne et al. (1987) for the delineation of separate species. Because the novel isolate FJAT-13831 and the most closely strain exhibited a DNA-DNA hybridization value near the 70% threhold (Wayne et al., 1987; Roselló-Mora and Amann, 2001), the average nucleotide identity (ANI) of core genes in the relative species was introduced to represent a robust measure of the pairwise distance between them (Sorokin et al., 2006). The whole genomics of the novel isolate FJAT-13831T was sequenced by Liu et al. (2012) with the accession number AKCS0000000, and 6 relative reference species in the Bacillus cereus group were collected from NCBI (listed in Table 4). The ANI of 2881 core genes identified by bidirectional best blasp with 30% identity and 60% coverage of shorter proteins for the relative species were calculated( ), the similarity matrix of ANI values is shown in Table 5. The distance matrix (Table 6) counted with 1-ANI/100 was then used in Splitstree to construct a Neighbor-Joining tree (Fig. 5) (Huson and Bryant, 2006). The results showed that the ANI value obtained between the novel isolate FJAT-13831T and the most closely strain B. pseudomycoides DSM 12442T was 91.74%. This high value confirms the similarity between the two sequences, but is clearly lower than 95%, which corresponds to the established threshold for species delineation (Goris et al., 2007). Table 4 Genomic information for Bacillus bingmayongensis sp. nov. FJAT-13831T and the relative type stains in the Bacillus cereus group from NCBI 9 Bo Liu and others Symbol 13831 ban1 bce1 bmy1 bpm bth1 bwe 217 218 219 220 221 222 223 Gene Number 5657 5328 5234 5658 5851 6243 5155 GenBank ID AKCS0000000 NC_003997.3 NC_004722.1 NZ_CM000742.1 NZ_CM000745.1 NZ_CM000753.1 NC_010184.1 Name Bacillus bingmayongensis sp. nov. FJAT-13831T Bacillus anthracis ATCC 14578T Bacillus cereus ATCC 14579T Bacillus mycoides DSM 2048T Bacillus pseudomycoides DSM 12442T Bacillus thuringiensis berliner ATCC 10792T Bacillus weihenstephanensis KBAB4 Table 5 The average nucleotide identity (ANI %) based on 2881 core genes between Bacillus bingmayongensis sp. nov. FJAT-13831T and the relative type stains in the Bacillus cereus group, respectively. It was showed that the ANI between 13831= Bacillus bingmayongensis sp. nov. FJAT-13831T and bpm= Bacillus pseudomycoides DSM 12442T was 91.47%. species 13831 ban1 bce1 bmy1 bpm bth1 bwe 13831 100.00 ban1 82.77 100.00 bce1 82.77 92.49 100.00 bmy1 83.22 90.26 90.37 100.00 bpm 91.47 82.64 82.75 83.26 100.00 bth1 82.84 92.41 97.31 90.46 82.73 100.00 bwe 83.27 90.28 90.31 98.25 83.18 90.47 Table 6 100.00 The distance matrix counted with 1-ANI/100 among the relative stains in the Bacillus cereus group. species 13831 ban1 13831 0.0000 ban1 0.1723 0.0000 bce1 bmy1 bpm bth1 bce1 0.1723 0.0751 0.0000 bmy1 0.1677 0.0974 0.0962 0.0000 bpm 0.0852 0.1735 0.1724 0.1674 0.0000 bth1 0.1715 0.0758 0.0269 0.0954 0.1727 0.0000 bwe 0.1673 0.0971 0.0969 0.0175 0.1681 0.0953 bwe 0.0000 226 Fig. 5 Relationships between the novel isolate FJAT-13831T and other select reference species using a distance matrix counted with 1-ANI/100 from 2881 core genes based on the whole genomics. The phylogenic tree was constructed using the neighbor joining method and percent bootstrap confidence levels were calculated using 1000 resamplings of the original data (Priest et al. 2004). The novel isolate FJAT-13831T was highlighted in black 229 230 231 On the basis of colony and cell morphologies, physiological and biochemical characteristics, 16S rRNA and gyrB analyses, DNA-DNA hybridization values, ANI of core gene in the whole genomes, 10 Bacillus bingmayongensis sp. nov. 232 233 234 235 236 DNA G+C content, fatty acid pattern, cell wall peptidoglycan type, major quinone MK-7, the novel isolate FJAT-13831T can be differentiated from members of the genus Bacillus as a novel species, for which the name Bacillus bingmayongensis sp. nov. is proposed. 237 Bacillus bingmayongensis (bing.ma. yong. en'sis. Pinyin n. Bīng Mǎ Yǒng, literally "military 238 239 servants" (Terra-cotta Warriors and Horses, a collection of 8,099 life-size terra cotta figures of warriors and horses located in the Mausoleum of the First Qin Emperor thousand years ago in 240 China); N.L. masc. adj. bingmayongensis of belonging to Bīng Mǎ Yǒng, a mausoleum in Xi'an 241 242 city of China, where the type strain was isolated. 243 Cells are rods (1.8–6.8×0.9–1.4 μm), Gram-positive, facultatively anaerobic, capable of forming 244 ellipsoidal endospores and motile. Colonies are flat with undulate margins and greyish-white 245 grown aerobically at 15-45 ℃ (optimum 35 ℃), pH 4.0-10.0 (optimum pH 7.0), 0–5% NaCl (w/v) 246 247 248 249 250 251 252 253 254 255 256 257 258 259 260 261 263 264 265 266 267 268 269 270 271 272 273 274 275 276 277 278 279 (optimum 0%) in the nutrient broth(NB). Positive for catalase and oxidase, but negative for ONPG(b-galactosidase), DNase, urease, arginine dihydrolase, lysine decarboxylase and ornithine decarboxylase. Nitrate is not reduced to nitrite or nitrogen. Acetoin H2S and indole are not produced. Cells do hydrolyse starch, but not gelatin and esculine. Utilizes Koser citrate broth and triple sugar iron, not KCN. Acid is produced from D-glucose, D-cellobiose, D-maltose, D-fructose, D-ribose, D-saccharose, D-trehalose, D-turanose, glycogene, glycerol, erythritol, N-acetylglucosamine, salicin, and potassium gluconate, but no acid is produced from D-arabinose, L-arabinose, D-lyxose, L-xylose, methyl b-D-xylopyranoside, D-galactose, D-mannose, L-sorbose, L-rhamnose, adonitol, inositol, D-mannitol, methyl a-D-mannopyranoside, methyl a-D-glucopyranoside, amygdaline, arbutin, dulcitol, D-sorbitol, inulin, D-melezitose, D-lactose, D-melibiose, D-tagatose, starch, xylitol, Gentiobiose, D-fucose, L-fucose, D-arabitol, L-arabitol, Potassium 2-cetogluconate and Potassium 5-cetogluconate. The DNA G+C content is 36.5 mol%. Cell wall peptidoglycan contains meso-diaminopimelic acid in the API 50CHB system. The predominant menaquinone is MK-7; and MK-5 and MK-4 are as a minor component. The major fatty acids are iso-C15:0, iso-C17:0, C16:0, iso-C13:0, anteiso-C15:0, iso-C17:1 ω5c with values of 21.03%, 11.49%, 9.83%, 7.66%, 7.39%, 5.12%, respectively. Description of Bacillus bingmayongensis sp. nov. Acknowlegement: We would like to thank Professor J. P. Euzéby for his suggestion on the spelling of the specific epithet. This work was supported by agricultural bioresources institute, Fujian Academy of Agricultural Sciences, PR China. The work was financed by the 948 project (2011-G25) from Chinese Ministry of Agriculture as well as by the 973 program earlier research project (2011CB111607), the project of agriculture science and technology achievement transformation (2010GB2C400220), the international cooperation project (2012DFA31120) from Chinese Ministry of Science and Technology, respectively. Reference: Ahmed, I., Yokota, A., Yamazoe, A., & Fujiwara, T. (2007). Proposal of Lysinibacillus boronitolerans gen. nov., sp. nov., and transfer of Bacillus fusiformis to Lysinibacillus fusiformis comb. nov. and Bacillus sphaericus to Lysinibacillus sphaericus comb. nov. Int. J. Syst. Evol. Microbiol. 57, 1117-1125. Antwerpen, M. H., Schellhase, M. E., Ehrentreich-Forster, F., Bier, W. & Nubel, U. (2007). DNA microarray for detection of antibiotic resistance determinants in Bacillus anthracis and closely related Bacillus cereus. Mol. 11 Bo Liu and others 280 281 282 283 284 285 286 287 288 289 290 291 292 293 294 295 296 297 298 299 300 301 302 303 304 305 306 307 308 309 310 311 312 313 314 315 316 317 318 319 320 321 322 323 324 325 326 327 328 329 330 331 332 333 Cell. Probes Probes, 21, 152-160.v Ash, C., Farrow, J. A., Wallbanks, S. & Collins, M. D. (1991). Phylogenetic heterogeneity of the genus Bacillus revealed by comparative analysis of small-subunit-ribosomal RNA sequences. Lett. Appl. Microbiol. 13, 202-206. Claus, D. & Berkeley, R. C. W. (1986). Genus Bacillus Cohn 1872, p. 1105-1140. In P.H.A. Sneath, N.S. Mair, M.E. Sharpe, and J.G. Holt (eds.), Bergey’s manual of systematic bacteriology, vol. 2. The Williams and Wilkins Co., Baltimore, USA. Fitch, W. M. (1971). Toward defining the course of evolution: minimum change for a specific tree topology. Syst. Zool. 20, 406-416. Hasegawa, T., Takizawa, M. & Tanida, S. (1983). A rapid analysis for chemical grouping of aerobic actinomycetes. J Gen Appl Microbiol 29, 319-322. Kaneko, T., Nozaki, R., & Aizawa, K. (1978). Deoxyribonucleic acid relatedness between Bacillus anthracis, Bacillus cereus, and Bacillus thuringiensis. Microbiol. Immunol. 22, 639-641. Maughan, H., Van der Auwera, G. (2011). Bacillus taxonomy in the genomicera finds phenotypes to be essential though often misleading. Infection, Genetics and Evolution 11, 11789–797. Nakamura, L. K. (1998). Bacillus pseudomycoides sp. nov. Int J Syst Bacteriol 48, 1031-1035. Staneck, J. L. & Roberts, G. D. (1974). Simplified approach to identification of aerobic actinomycetes by thin-layer chromatography. Appl Microbiol 28, 226–231. Tamaoka, J. & Komagata, K. (1984). Determination of DNA base composition by reversed-phase high-performance liquid chromatography. FEMS Microbiol Lett 25, 125-128. Tamura, K., Nei, M. & Kumar, S. (2004). Prospects for inferring very large phylogenies by using the neighbor-joining method. Proceedings of the National Academy of Sciences (USA) 101, 11030-11035. Thompson, J. D., Gibson, T. J., Plewniak, F., Jeanmougin, F. & Higgins, D. G. (1997). The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25, 4876–4882. Tiago, I., Pires, C., Mendes, V., Morais, P. V., Da Costa, M. S. & Veríssimo, A. (2006). Bacillus foraminis sp. nov., isolated from a non-saline alkaline groundwater. Int. J. Syst. Evol. Microbiol. 56, 2571-2574. Torsvik, V., Daae, F. L. Daae, & Goksyr, J. Goksyr. (1995). Extraction, purification, and analysis of DNA from soil bacteria. In, Nucleic Acids in the environment: Methods and Applications. Trevors, J.T. and J. D. van Elsas (eds.), Berlin, Heidelberg, New York, Springer Verlag, pp. 29-48. Vandamme, P., Pot, B., Gillis M., de Vos, P., Kersters, K., & Swings, J. (1996). Polyphasic taxonomy, a consensus approach to bacterial systematics. Microbiol Rev 60, 407–438. Vogler, A. J., Busch, J. D., Percy-Fine, S. Tipton-Hunton, C., Smith, K. L., & Keim, P. (2002). Molecular analysis of rifampin resistance in Bacillus anthracis and Bacillus cereus. Antimicrob. Agents Chemother. 46, 511-513. Yoon, J. H., Lee, J. K., Shin, Y. K., Park, Y. H., & Lee, S. T. (1997). Reclassification of Nocardioides simplex ATCC 13260, ATCC 19565, and ATCC 19566 as Rhodococcus erythropolis. Int J Syst Bacteriol 47, 904-907. Zhang, T., Fan, X., Hanada S., Kamagata Y. & Fang H. H. P. (2006). Bacillus macauensis sp. nov., a long-chain bacterium isolated from a drinking water supply. Int. J. Syst. Evol. Microbiol. 56, 349-353. Seveno, N. A. Morgan, J. A. W. Wellington, E. M. H. (2001) Growth of Pseudomonas aureofaciens PGS12 and the dynamics of HHL and phenazine production in liquid culture, on nutrient agar, and on plant roots. Microbial Ecology; 2001. 41: 4, 314-324. Altschul S F, Gish W, Miller W, Myers E W, Lipman D J. (1990). Basic local alignment search tool. J Mol Biol. 1990, 215(3):403-10. Atlas, R. M. (1993). Handbook of Microbiological Media. Edited by L. C. Parks. Boca Raton, FL: CRC Press. Baik, K. S., Lim, C. H., Park, S. C., Kim, E. M., Rhee, M. S., & Seong, C. N. (2010). Bacillus rigui sp. nov., isolated from wetland fresh water. Int J Syst Evol Microbiol 60, 2204-9. Bo Liu, Guohong Liu, Naiquan Lin, Jianyang Tang. (2012). Bacillus identification and phylogenetic analysis, isolated from the 1st pit soil of Emperor Qin's Terracotta Warrior. [J]. Fujian Journal of agricultural sciences, 27 (6): 563-573. Chen, Y. G., Hao, D. F., Chen, Q. H., Zhang, Y.Q., Liu, J. B., He, J. W., Tang, S. K., & Li, W. J. (2011). Bacillus hunanensis sp. nov., a slightly halophilic bacterium isolated from non-saline forest soil. Antonie Van Leeuwenhoek, 99(3):481-8. De Ley, J., Cattoir, H., Cattoir, Reynaerts, & Reynaerts, A. (1970). The quantitative measurement of DNA 12 Bacillus bingmayongensis sp. nov. 334 335 336 337 338 339 340 341 342 343 344 345 346 347 348 349 350 351 352 353 354 355 356 357 358 359 360 361 362 363 364 365 366 367 368 369 370 371 372 373 374 375 376 377 378 379 380 381 382 383 384 385 386 hybridization from renaturation rates. Eur. J. Biochem 12, 133-142. Felsenstein, J. (1985). Confidence limits on phylogenies: an approach using the bootstrap. Evolution 40, 783-791. Gatson, J. W., Benz, B. F., Chandrasekaran, C., Satomi, M., Venkateswaran, K., & Hart, M. E. (2006). Bacillus tequilensis sp. nov., isolated from a 2000-year-old Mexican shaft-tomb, is closely related to Bacillus subtilis. Int J Syst Evol Microbiol 56, 1475-84. Gonzalez, J. M. Gonzalez & Saiz-Jimenez, C. (2005). A simple fluorimetric method for the estimation of DNA–DNA relatedness between closely related microorganisms by thermal denaturation temperatures. Extremophiles 9, 75–79. Goris, J., Konstantinidis, K. T., Klappenbach, J. A., Coenye, T., Vandamme, P. & Tiedje, J. M. (2007). DNA–DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol 57, 81–91. Gregersen, T. (1978). Rapid method for distinction of GramGram -negative from Gram -positive bacteria. Eur J Appl Microbiol Biotechnol 5, 123–127. Groth, I., Schumann, P., Weiss, N., Martin, K. & Rainey, F. A. (1996). Agrococcus jenensis gen. nov., sp. nov., a new genus of actinomycetes with diaminobutyric acid in the cell wall. Int J Syst Bacteriol 46, 234-239. Guohong Liu, Bo Liu, Naiquan Lin, Weiqi Tang, Jianyang Tang, and Yingzhi Lin. (2012). Genome Sequence of the Aerobic Bacterium Bacillus sp. Strain FJAT-13831. J. Bacteriol. 194(23):6633. Jukes, T. H. and Cantor, C. R. (1969). Evolution of protein molecules. In Munro HN, editor, Mammalian Protein Metabolism, pp. 21-132, Academic Press, New York. Jung, M. Y., Kim, J. S., Paek, W. K., Lim, J., Lee, H., Kim, P.I., Ma, J.Y., Kim, W., & Chang, Y. H. (2011). Bacillus manliponensis sp. nov., a new member of the Bacillus cereus group isolated from foreshore tidal flat sediment. J Microbiol49(6):1027-32. Kämpfer, P., Blasczyk, K., & Auling, G. (1994). Characterization of Aeromonas genomic species by using quinone, polyamine, and fatty acid patterns. Can. J. Microbiol. 40, 844-850. Kaneda, T. (1977). Fatty acids of the genus Bacillus: an example of branched-chain preference. Bacteriol Rev. 41(2): 391–418. La Duc, M. T., Satomi M., Agata, N., Venkateswaran, K. (2004a). gyrB as a phylogenetic discriminator for members of the Bacillus anthracis-cereus-thuringiensis group. J Microbiol Methods. 56(3):383-94. La Duc, M. T., Satomi, M. & Venkateswaran, K. (2004b). Bacillus odysseyi sp. nov., a round-spore-forming bacillus isolated from the Mars Odyssey spacecraft. Int J Syst Evol Microbiol 54, 195–201. Logan, N. A. & Berkeley, R. C. W. (1984). Identification of Bacillus strains using the API system. J Gen Microbiol 130, 1871–1882. Nazina, T. N, Lebedeva, E. V., Poltaraus, A. B., Tourova, T. P., Grigoryan, A. A., Sokolova, D. S.h, Lysenko, A. M., & Osipov, G. A. (2004). Geobacillus gargensis sp. nov., a novel thermophile from a hot spring, and the reclassification of Bacillus vulcani as Geobacillus vulcani comb. nov. Int J Syst Evol Microbiol. 54, 2019-24. Owen, R. J. & Hill, L. R. (1979). The estimation of base compositions, base pairing and genome size of bacterial deoxyribonucleic acids. In Identification Methods for Microbiologists, 2nd edn, pp. 277–296. Edited by F. A. Skinner & D. W. Lovelock. London: Academic Press. Priest, F. G., Goodfellow Goodfellow, M., &and Todd, C. (1988). A numerical classification of the genus Bacillus. J. Gen. Microbiol. 134, 1847-1882. Rosellóo-Mora, R., & Amann, R. (2001). The species concept for prokaryotes. FEMS Microbiol Rev 25, 36-67. Saitou, N. & Nei, M. (1987). The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol Biol Evol 4, 406-425. Sambrook J, Fritschi E F and Maniatis T (1989). Molecular cloning: a laboratorymanual, Cold Spring Harbor Laboratory Press, New York. Sasser, M. (1990). Identification of bacteria by gas chromatography of cellular fatty acids. USFCC News 20, 16. Satomi, M., Kimura, B., Hamada, T., Harayama, S., Fujii, T. (2002). Phylogenetic study of the genus Oceanospirillum based on 16S rRNA and gyrB genes: emended description of the genus Oceanospirillum, description of Pseudospirillum gen. nov., Oceanobacter gen. nov. and Terasakiella gen. nov. and transfer of Oceanospirillum jannaschii and Pseudomonas stanieri to Marinobacterium as Marinobacterium jannaschii comb. nov. and Marinobacterium stanieri comb. no. Int. J. Syst. Evol. Microbiol. 52, :739-47. Satomi, M., Kimura, B., Hayashi, M., Okuzumi, M. & Fujii, T. (2004). Marinospirillum insulare sp. nov., a novel halophilic helical bacterium isolated from kusaya gravy. Int J Syst Evol Microbiol 13 Bo Liu and others 387 388 389 390 391 392 393 394 395 396 397 398 399 400 401 402 403 404 405 406 407 408 409 410 411 412 413 414 415 416 417 418 419 420 421 422 423 424 425 426 427 428 429 430 431 54, 163–167. Satomi, M., La Duc, M. T. and Venkateswaran, K. (2006). Bacillus safensis sp. nov., isolated from spacecraft and assembly-facility surfaces. Int. J. Syst. Evol. Microbiol. 56, 1735-1740. Satomi, M., Oikawa, H. & Yano, Y. (2003). Shewanella marinintestina sp. nov., Shewanella schlegeliana sp. nov. and Shewanella sairae sp. nov., novel eicosapentaenoic-acid-producing marine bacteria isolated from sea-animal intestines. Int J Syst Evol Microbiol 53, 491–499. Shida, O., Takagi, H., Kadowaki, K., Nakamura, L. K. & Komagata, K. (1997). Transfer of Bacillus alginolyticus, Bacillus chondroitinus, Bacillus curdlanolyticus, Bacillus glucanolyticus, Bacillus kobensis, and Bacillus thiaminolyticus to the genus Paenibacillus and emended description of the genus Paenibacillus. Int J Syst Bacteriol 47, 289-298. Smibert, R.M. & Krieg, N. R. (1994). Phenotypic characterization. In Methods for General and Molecular Bacteriology, pp. 607–654. Edited by P. Gerhardt, R. G. E. Murray, W. A. Wood & N. R. Krieg. Washington, DC: American Society for Microbiology. Sorokin A, Candelon B, Guilloux K, Galleron N, Wackerow-Kouzova N, Ehrlich SD, Bourguet D, Sanchis V. (2006). Multiple-locus sequence typing analysis of Bacillus cereus and Bacillus thuringiensis reveals separate clustering and a distinct population structure of psychrotrophic strains. Appl Environ Microbiol. 72(2):1569-78. Stackebrandt, E. & Ebers, J. (2006). Taxonomic parameters revisited: tarnished gold standards. Microbiol. Today 33, 152-155. Stackebrandt, E. & Goebel, B. M. (1994). Taxonomic note: a place for DNA-DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. Int J Syst Bacteriol 44, 846–849. Tamura, K., Peterson, D., Peterson, N., Stecher, G., Nei, M., & Kumar, S. (2011). MEGA5: Molecular Evolutionary Genetics Analysis using Maximum Likelihood, Evolutionary Distance, and Maximum Parsimony Methods. Molecular Biology and Evolution (In Press). Venkateswaran K., Dohmoto N., and Harayama S. (1998). Cloning and Nucleotide Sequence of the gyrB Gene of Vibrio parahaemolyticus and Its Application in Detection of This Pathogen in Shrimp. Appl Environ Microbiol. 64(2): 681–687. Venkateswaran, K., Moser, D. P., Dollhopf, M. E. & 10 other authors (1999). Polyphasic taxonomy of the genus Shewanella and description of Shewanella oneidensis sp. nov. Int J Syst Bacteriol 49, 705–724. Wayne, L. G., Brenner, D. J., Colwell, R. R., Grimont, P. A. D., Kandler, O., Krichevsky, M. I., Moore, L. H., Moore, W. E. C., Murray, R. G. E. & other authors. (1987). International Committee on Systematic Bacteriology. Report of the ad hoc committee on reconciliation of approaches to bacterial systematics. Int J Syst Bacteriol 37, 463–464. Yamamoto, Satoshi, & Harayama, S. (1995). PCR Amplification and Direct Sequencing of gyrB Genes with Universal Primers and Their Application to the Detection and Taxonomic Analysis of Pseudomonas putida Strains. Appl Environ Microbiol 61,1104–1109. Yoon J H, Lee J S, Shin Y K, Park Y H and Lee S T. (1997). Reclassification of Nocardioides simplex ATCC 13260, ATCC 19565, and ATCC 19566 as Rhodococcus erythropolis. International jounal of systematic bcteriology, 47(3): 904-907. Zhang, L., Wu, G. L., Wang, Y., Dai, J., & Fang, C.X. (2011). Bacillus deserti sp. nov., a novel bacterium isolated from the desert of Xinjiang, China. Antonie Van Leeuwenhoek 99(2):221-9. Alexei Sorokin,1,* Benjamin Candelon,1,† Kévin Guilloux,1 Nathalie Galleron,1 Natalia Wackerow-Kouzova,1,‡ S. Dusko Ehrlich,1 Denis Bourguet,2,§ and Vincent Sanchis2. 2006. Multiple-Locus Sequence Typing Analysis of Bacillus cereus and Bacillus thuringiensis Reveals Separate Clustering and a Distinct Population Structure of Psychrotrophic Strains. Appl Environ Microbiol. 2006 February; 72(2): 1569–1578. Thompson, J. D., T. J. Gibson, F. Plewniak, F. Jeanmougin, and D. G. Higgins. 1997. The ClustalX windows 14 Bacillus bingmayongensis sp. nov. interface: flexible strategies for multiple sequence alignment aided by the quality analysis tools. Nucleic Acids Res. 24:4876-4882. Perrière G, Gouy M. 1996.WWW-query: an on-line retrieval system for biological sequence banks. Biochimie. 1996;78(5):364-9. Jolley KA, Feil EJ, Chan MS, Maiden MC. 2001. Sequence type analysis and recombinational tests (START). Bioinformatics. 2001 Dec;17(12):1230-1. Morgan N Price,1,2,* Paramvir S Dehal,1,2 and Adam P Arkin1,2,3,Orthologous Transcription Factors in Bacteria Have Different Functions and Regulate Different Genes. PLoS Comput Biol. 2007 September; 3(9): e175. Published online 2007 September 7. doi: 10.1371/journal.pcbi.0030175. PMCID: PMC1971122 Yan Ming Zhang,1,2,3 Chang Fu Tian,1,2,3,* Xin Hua Sui,1,2,3 Wen Feng Chen,1,2,3 and Wen Xin Chen1,2,3. Robust Markers Reflecting Phylogeny and Taxonomy of Rhizobia. PLoS One. 2012; 7(9): e44936. Published online 2012 September 17. doi: 10.1371/journal.pone.0044936. PMCID: PMC3444505 Priest FG, Barker M, Baillie LW, Holmes EC, Maiden MC. Population structure and evolution of the Bacillus cereus group. J Bacteriol. 2004 Dec;186(23):7959-70. 432 15