Biology
Slide
1 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Slide
2 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
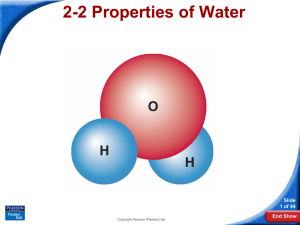
The Water Molecule
The Water Molecule
Like all molecules, a water molecule is neutral.
Slide
3 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Why are water molecules polar?
Slide
4 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Polarity
However an oxygen atom, with 8 protons in its
nucleus, has a much stronger attraction for
electrons than does the hydrogen atom with a
single proton.
Thus, there is a greater probability of finding the
shared electrons near the oxygen atom than near
the hydrogen atom.
Slide
5 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
As a result, the oxygen end of the molecule has a
slight negative charge and the hydrogen end has a
slight positive charge.
A water molecule is polar because there
is an uneven distribution of electrons
between the oxygen and hydrogen
atoms.
Slide
6 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Water Molecule
Slide
7 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Hydrogen Bonds
Because of their partial positive and negative
charges, polar molecules can attract each other.
Slide
8 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
The attraction between the hydrogen atoms on one
molecule and the oxygen atom on another water
molecule is an example of a hydrogen bond.
Hydrogen bonds are not as strong as covalent or
ionic bonds.
Slide
9 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
A single water molecule may be involved in as many
as four hydrogen bonds at a time.
Water’s ability to form multiple hydrogen bonds is
responsible for many of its special properties.
Slide
10 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Hydrogen bonds form
between water
molecules.
Slide
11 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Cohesion is an attraction between molecules of the
same substance.
Because of hydrogen bonding, water is extremely
cohesive.
Slide
12 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Water's cohesion causes molecules on the surface
of water to be drawn inward, which is why drops of
water form beads on a smooth surface.
Cohesion also explains why some insects and
spiders can walk on a pond's surface.
Slide
13 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Adhesion is an attraction between molecules of
different substances.
The surface of water in a graduated cylinder dips in
the center because adhesion between water
molecules and glass molecules is stronger than
cohesion between water molecules.
Slide
14 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Adhesion between water and glass causes water to
rise in a narrow tube against the force of gravity.
This effect is called capillary action.
Slide
15 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
The Water Molecule
Capillary action is one of the forces that draw water
out of the roots of a plant and up into its stems and
leaves.
Cohesion holds the column of water together as it
rises.
Slide
16 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
Solutions and Suspensions
A mixture is a material composed of two or more
elements or compounds that are physically mixed
but not chemically combined.
Slide
17 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
Two types of mixtures can be made with water
• solutions
• suspensions
Slide
18 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
Solutions
All the components of a solution are evenly
distributed throughout the solution.
In a salt–water solution, table salt is the solute—the
substance that is dissolved.
Water is the solvent—the substance in which the
solute dissolves.
Slide
19 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
When a crystal of table salt is placed in warm water,
sodium and chloride ions are attracted to the polar
water molecules.
Cl
-
Cl Na+
Na+
Water
Water
Slide
20 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
Ions break away from the crystal and are surrounded
by water molecules.
Cl -
Cl -
Na+
Na+
Water
Water
Slide
21 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
The ions gradually become dispersed in the water,
forming a solution.
Cl -
Cl -
Na+
Na+
Water
Water
Slide
22 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
Water's polarity gives it the ability to dissolve both
ionic compounds and other polar molecules, such as
sugar.
Water is the greatest solvent on Earth.
Slide
23 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
Suspensions
Some materials do not dissolve when placed in
water but separate into pieces so small that they
do not settle out.
Slide
24 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
The movement of water molecules keeps the small
particles suspended.
Such mixtures of water and nondissolved material
are known as suspensions.
Slide
25 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Solutions and Suspensions
Some of the most important biological fluids are both
solutions and suspensions.
The blood that circulates through your body is mostly
water, which contains many dissolved compounds.
Blood also contains cells and other undissolved
particles that remain in suspension as the blood
moves through the body.
Slide
26 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
What are acidic solutions? What are basic
solutions?
Slide
27 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
Acids, Bases, and pH
A water molecule can react to form hydrogen and
hydroxide ions.
Water is neutral because the number of positive
hydrogen ions (H+) produced is equal to the
number of negative hydroxide ions (OH-)
produced.
Slide
28 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
The pH scale
Chemists devised a measurement system called
the pH scale to indicate the concentration of H+
ions in solution.
The pH scale ranges from 0 to 14.
Slide
29 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
The pH Scale
At a pH of 7, the
concentration of H+
ions and OH- ions is
equal.
Pure water has a pH
of 7.
Sea water
Human blood
Pure water
Milk
Normal rainfall
Slide
30 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
The pH Scale
Solutions with a pH
below 7 are called acidic
because they have more
H+ ions than OH- ions.
The lower the pH, the
greater the acidity.
Acid rain
Tomato juice
Lemon juice
Stomach acids
Slide
31 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
The pH Scale
Solutions with a pH
above 7 are called basic
because they have more
OH- ions than H+ ions.
Oven cleaner
Bleach
Ammonia solution
Soap
The higher the pH, the
more basic the solution.
Slide
32 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
Acids
An acid is any compound that forms H+ ions in
solution.
Acidic solutions contain higher
concentrations of H+ ions than pure water
and have pH values below 7.
Slide
33 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
Strong acids tend to have pH values that range from
1 to 3.
The hydrochloric acid produced by the stomach to
help digest food is a strong acid.
Slide
34 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
Bases
A base is a compound that produces hydroxide ions
(OH- ions) in solution.
Basic, or alkaline, solutions contain
lower concentrations of H+ ions than
pure water and have pH values above 7.
Slide
35 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
Strong bases, such as lye, tend to have pH values
ranging from 11 to 14.
Slide
36 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
Buffers
The pH of the fluids within most cells in the human
body must generally be kept between 6.5 and 7.5.
If the pH is lower or higher, it will affect the
chemical reactions that take place within the cells.
Slide
37 of 44
Copyright Pearson Prentice Hall
2-2 Properties of Water
Acids, Bases, and pH
Controlling pH is important for maintaining
homeostasis.
One of the ways that the body controls pH is through
dissolved compounds called buffers.
Buffers are weak acids or bases that can react with
strong acids or bases to prevent sharp, sudden
changes in pH.
Slide
38 of 44
Copyright Pearson Prentice Hall
2-2
Click to Launch:
Continue to:
- or -
Slide
39 of 44
Copyright Pearson Prentice Hall
2-2
A molecule in which the charges are unevenly
distributed is called a
a. polar molecule.
b. cohesive molecule.
c. hydrogen molecule.
d. covalent molecule.
Slide
40 of 44
Copyright Pearson Prentice Hall
2-2
A dissolved substance is called a
a. solvent.
b. solution.
c. solute.
d. suspension.
Slide
41 of 44
Copyright Pearson Prentice Hall
2-2
A compound that produces hydroxide ions in
solution is called a(an)
a. base.
b. buffer.
c. acid.
d. salt.
Slide
42 of 44
Copyright Pearson Prentice Hall
2-2
Hydrogen bonds between water molecules
result from
a. adhesion between water molecules.
b. magnetic attractions between water
molecules.
c. uneven electron distribution in each water
molecule.
d. ionic bonds in the water molecule.
Slide
43 of 44
Copyright Pearson Prentice Hall
2-2
On a pH scale, a value of 2 means that the
solution has
a. equal concentrations of H+ and OH- ions.
b. the same concentration of H+ ions as pure
water.
c. higher concentration of H+ than in pure
water.
d. lower concentration of H+ than in pure water.
Slide
44 of 44
Copyright Pearson Prentice Hall
END OF SECTION