Solutions & Gases: Reading Solubility Tables F & G
advertisement

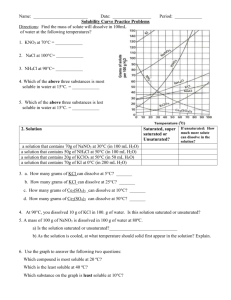
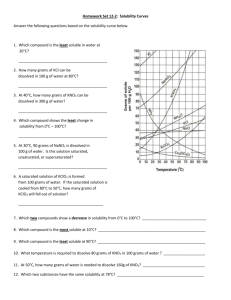
UNIT 6 SOLUTIONS AND GASES 6.3 How do we read table F and G? April 11, 2011 AIM: How do we know if a substance is soluble in another, and to what extend? DO NOW: 1. Define solubility. How much of a solute will dissolve in a certain amount of solvent 2. What factors affect solubility? TEMPERATURE PRESSURE POLARITY OF SUBSTANCES TABLE F WORD DEFINITION SOLUBILITY How much of a solute will dissolve in a certain amount of solvent SOLUBLE Materials with high solubility INSOLUBLE Materials with low solubility PRECIPITATE Formation of a solid in a solution CATEGORY Dissolves or does not dissolve? SOLUBLE Dissolves INSOLUBLE Dissolves Formation of Precipitate? No Yes Is NH4Cl soluble in H2O? All ammonium ions are soluble and halides (Cl-, Br-, I) are soluble. Therefore, NH4Cl is soluble in H2O. Is silver chloride soluble in H2O? All halides are soluble. However, the EXCEPTIONS are when combined with Ag+, Pb2+, and Hg22+. Since silver (Ag) is listed as an exception, silver chloride is insoluble. PRACTICE LiF HNO3 NaOH Ca(OH)2 AgBr Fe3(PO4)2 PbCl2 H2SO4 AgI CaS (NH4)2S KClO4 TABLE G WORD SATURATED “Just enough” DEFINITION a solution that contains a maximum amount of solute that will dissolve at a specific temperature SUPERSATURATED a solution holds more solute than is present in a saturated solution at a specific temperature UNSATURATED the solution holds less solute than the maximum it can hold “More than it can handle” “Less than it can handle” USING TABLE G Table G tells us the grams of solute that can be dissolved in H2O at various temperatures Point on Graph Saturation ON THE LINE SATURATED BELOW THE LINE UNSATURATED OVER THE LINE SUPERSATURATED a) 60 grams of KCl at 80C: SUPERSATURATED _____ i) 60 grams of HCl at 45C _____ SATURATED b) 70 grams of KNO3 at 50C: UNSATURATEDc ___ j) 110 grams at NaNO3 at 45C ____ SATURATED c) 40 grams of NaCl at 90C: SATURATED ____ k) 1 gram of SO2 at 10C _____ UNSATURATED d) 140 grams of KI at 20C UNSATURATED _____ l) 140 grams of KI at 10C _____ SUPERSATURATED e) 90 grams of NH4Cl at SUPERSATURATED 100C___ m) At how many grams of NH3 at 100C would the solution be supersaturated? ANYTHING ABOVE 8g _________________ f) 70 grams of NH3 at 10C SATURATED _____ n) At how many grams of KNO3 at 40C would the solution be unsaturated? ANYTHING BELOW 63g __________________ g) 10 grams of NH3 at 80C UNSATURATED _____ o) At how many grams of KCl at 60C would the solution be saturated? AT 45g ____________________ h) 20 grams of KClO3 at 30C SUPERSATURATED ___ p) At how many grams of NaCl at 90C would the solution be saturated? AT 40g ______________________ INDEPENDENT PRACTICE • Complete the Independent Practice • PLACE COMPLETED IP IN HOMEWORK BIN