745 cm -1 Aromatic CH bend
advertisement

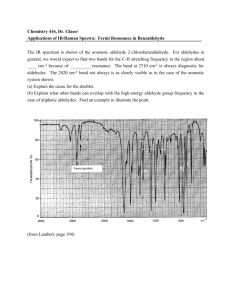
The Use of Infrared Microspectroscopy to Determine the Biotransformation of Benzotriazole by (Helianthus annuus L.) Sunflowers Kenneth M. Dokken1, Lawrence C. Davis1, Larry E. Erickson2, David L. Wetzel3, Nebojsa Marinkovic4, John A. Reffner5 1Department of Biochemistry, Kansas State University 2Department of Chemical Engineering, Kansas State University 3 Microbeam Molecular Spectroscopy Laboratory, Kansas State University 4Center for Synchrotron Biosciences, Albert Einstein College of Medicine 5SensIR Technologies, Danbury, CT ABSTRACT Infrared microspectroscopy has been extensively applied in plant cell wall analysis to monitor developmental changes. However, this technique is not widely used to study changes of plant structures induced by exposure to organic contaminants. Previous studies of finely ground secondary roots treated with various concentrations of benzotriazole used infrared spectroscopy of KBr pellets to examine absorptions in the region of 749-775 cm-1. Increased peak heights at 749 cm-1 were proportional to the concentration of benzotriazole to which the plant had been treated. Peaks from lignin near 870 cm-1 decreased simultaneously. In this study, sunflower plants were grown hydroponically in the presence of benzotriazole at concentrations less than 1mM. At low concentrations, treated sunflower plants develop nearly as well as untreated plants which allows optimal uptake and incorporation of benzotriazole into the plant. Changes in the secondary structure due to uptake, incorporation, and/or transformation of benzotriazole were monitored using diamond attenuated total reflectance (ATR) and reflection absorption microspectroscopy. Diamond ATR spectra displayed the same changes in spectra of secondary roots as seen in the traditional IR spectroscopy method of KBr pellets. False color intensity maps of treated secondary root sections were produced using absorption reflection infrared microspectroscopy at Brookhaven National Laboratories. These showed changes in plant structure as well as the presence of aromatic CH peaks due to incorporation of benzotriazole within the plant. The technique of IR microspectroscopy may be used as a tool in phytoremediation to study changes of plant structure induced by the presence of organic contaminants in soil and water. Objectives • Pinpoint the tissue location where benzotriazole is being concentrated and/or transformed • Determine the changes in plant structure induced by exposure to benzotriazole • Determine how the benzotriazole is being transformed and/or incorporated into the plant • To use this system as a model for determining the fate of organic compounds in all types of vegetation Rationale It is vital to understand the fate of organic contaminants within the plants used for waste cleanup (phytoremediation). It is well documented that many plants can biotransform organic contaminants. Major issues in the field are whether transformed contaminants are still bioavailable and whether they may have greater toxicity than the parent compound. As currently understood, plant detoxification of xenobiotics typically depends on oxidation, conjugation and sequestration. For various isotopically labeled compounds, a large portion usually ends up in the lignin fraction of plant cells. We propose to use benzotriazole as a model compound for which we can specifically map out the locations of sequestration, which often is initially found in the vacuole and ultimately in lignin. We know that benzotriazole is transformed upon entry to the plant. Our working hypothesis is that the peroxidatic system responsible for lignification ultimately incorporates benzotriazole into a polymeric material that is no longer bioavailable. BENZOTRIAZOLE Commonly used as a corrosion inhibitor in antifreeze, airplane deicing fluids, gasoline, oil, lubricants, and heat exchanger fluids Toxic to aquatic organisms and bacteria, possibly carcinogenic, UV resistant (high stability), complexes with metals Has been detected in ground and surface waters and has no known bacterial degradation pathway H N N N F.W. 119.13 Sunflowers grown in nutrient solutions containing various concentrations of Benzotriazole (BT) for 14 days Leaves of treated plants turn dark green and become crinkled control 10 mg/L 20 mg/L 30 mg/L BT BT BT Higher concentrations of BT lead to the production of small highly branched roots INTERNAL REFLECTING OBJECTIVE IlluminatIR® mounted between the body and the eyepiece of an infinity corrected microscope Diamond Attentuated Total Reflectance (ATR) Spectroscopy The diamond internal reflection objective design features the capability of visible light viewing of the specimen surface both in the survey mode and during data collection. A lens (see figure) inside of the secondary mirror is focused by adjusting its height independent of the reflecting surface The large ZnSe internal reflecting element surface is protected by a small diamond cap. The curved surface assures optical contact with the specimen. Sample Preparation for Diamond ATR Spectroscopy Dry treated and untreated secondary roots of sunflower plants were laterally dissected and placed on reflective slides The instrument used to obtain the diamond ATR spectral data was an IlluminatIR® (SensIR® Technologies, Danbury, CT) with a diamond ATR objective. 8cm-1 resolution was used with 256 scans coadded. Longitudinal section of dry secondary root Diamond ATR Spectra of xylem tissue of treated and untreated dried secondary roots Untreated root Roots treated w/ 60 mg/L BT Relative 745 cm-1 Aromatic CH bend Infrared Microspectroscopy at the Center for Synchrotron Biosciences, Brookhaven National Labs, Beamline U2B Nicolet Nic Plan IR Microscope Advantages of a Synchrotron IR Source • Brightness is 1000 times greater than conventional (globar) sources for mid-IR • High spatial resolution and spectral resolution • Decreased source noise • Able to determine changes in structure at a cellular level IR spectra of root xylem of untreated and treated Sunflower using a 100m diameter aperture 1635 cm-1 aromatic stretch 1735 cm-1 CO stretch 1250 cm-1 CO stretch of lignin aromatic rings 745 cm-1 Aromatic C-H out-of-plane bending Assignment of Important Spectral Bands for Lignin and Benzotriazole Frequency Assignment Comments 745 cm-1 Aromatic C-H out-of-plane bending Benzene ring in Benzotriazole 1250 cm-1 Guaiacyl ring breathing with CO stretching Guaiacyl –syringyl subunits in lignin 1635 cm-1 Aromatic stretch Aromatic rings of lignin 1750 cm-1 CO stretch Unconjugated ketone and carboxyl groups commonly found in lignin Sarkanen and Ludwig 1971, Lin-Vien et al. 1991 , Mohan and Settu 1993 Untreated Sunflower Control 1750 cm-1 Log µm Co ntro l S unfl owe r 01 : P o i nt 1 @ (-31 75, -29 53) 60 0 .5 40 20 0 .0 0 3 500 0 3 000 50 2 500 2 000 1 50 (c m -1 ) W ave num bers P o s i ti on (m i c rom eters ) 1 00 1 500 1 000 1 500 1 000 2 00 1250 cm-1 Log µm 6 0 Co ntro l S unfl owe r 01 : P o i nt 1 @ (-31 75, -29 53) 04 .50 20 0 .0 0 3 500 0 50 3 000 1 00 2 500 2 000 1 50 W ave num bers (c m -1 ) 2 00 P o s i ti on (m i c rom eters ) 745 cm-1 µm 60 Control plants do not contain these bands!!!! 40 20 0 0 50 1 00 1 50 P o s i ti on (m i c rom eters ) 2 00 4 m section of secondary root from untreated sunflower controls -0 .0 Log µm 1750 cm-1 02.40 0 0 .2 3 500 3 000 2 500 2 000 s u nflo wer 20 mg/L B T tip : P oint 1 @ (14 716 , 57 66) W ave num bers (c m-1 ) 0 1 00 Sunflowers treated 1 500 000 with 120 mg/L Benzotriazole 2 00 Po s iti on (mic rom eters ) -0 .0 1250 cm-1 3 500 3 000 2 500 2 000 1 500 1 000 µm W ave num bers (c m-1 ) 20 0 0 1 00 2 00 P o s iti on (mic rom eters ) µm 745 cm-1 20 0 0 1 00 2 00 Po s iti on (mic rom eters ) 4 m section of secondary root from sunflowers treated with 20 mg/L benzotriazole Conclusions • Diamond ATR and IR Microspectroscopy can be used as a technique to detect changes in plant structure due to exposure to organic contaminants. • Benzotriazole becomes transformed and incorporated into the sunflower plant presumably through lignin – Presence of 745 cm-1 due to C-H bending in Benzotriazole – More prominent bands at 1250 cm-1 and 1750 cm-1 due to increased lignin production References 1. K. M. Dokken, L. C. Davis, L. E. Erickson, S. Castro (2002) Fourier transform infrared spectroscopy as a tool to monitor changes in plant structure in response to soil contaminants. Proceedings of the 2002 Conference on Application of Waste Remediation Technologies to Agricultural Contamination of Water Resources, Kansas City, MO, 2002, pgs. 250-256, http://www.engg.ksu.edu/HSRC. 2. M. C. McCann, M. Bush, D. Milioni, P. Sado, N. J. Stacey, G.Catchpole, M. Defernez, N. C. Carpita, H. Hofte, P. Ulvskov, R. H. Wilson, K. Roberts (2001) Approaches to understanding the functional architecture of the plant cell wall. Phytochemistry 57:811-821. 3. G. Mouille, S. Robin, M. Lecomte, S. Pagant, H. Hofte (2003) Calssification and identification of Arabidopsis cell wall mutants using Fourier-Transform Infrared (FT-IR) microspectroscopy. The Plant Journal 35: 393-404. 4. N. S. Marinkovic, R. Huang, P. Bromberg, M. Sullivan, J. Toomey, L. M. Miller, E. Sperber, S Moshe, K.W. Jones, E. Chouparova, S. Lappi, S. Franzen, M.R. Chance (2002) Center for Synchrotron Biosciences’ U2B Beamline: an international resource for biological infrared spectroscopy. Journal of Synchrotron Radiation 9:189-197. 5. K.V. Sarkanen and C.H. Ludwig (1971) Lignins:Occurrence, Formation, Structure, and Reactions, John Wiley and Sons, Inc., New York, New York. 6. S. Mohan and K. Settu (1993) Vibrational spectra and analysis of 1,2,3-benzotriazole. Indian Journal of Pure Applied Physics 31: 850-854. 7. D. Lin-Vien, J. G. Grasselli, N.B. Colthup, W. G. Fateley (1991) The Handbook of Infrared and Raman Characteristic Frequencies of Organic Molecules, Academic Press, New York, New York. Acknowledgements The authors thank SensIR Technologies for the use of their instruments and facilities in Danbury, CT. This project was supported by U.S. Environmental Protection Agency under assistance agreement R-825550 through the Great Plains/Rocky Mountain Hazardous Substance Research Center, Kansas Agricultural Experiment Station and the KSU Microbeam Molecular Spectroscopy Laboratory. The authors would also like to thank Cindy Chard-Bergstrom and Deborah St. Cyr for their help with microtoming the root sections.