281-Calendar
advertisement

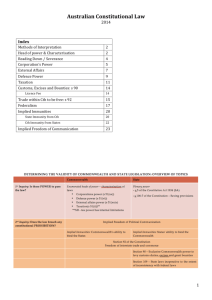
CHEM 281(001) Winter 2013-14, Inorganic Chemistry Class Activities and Deadlines Lectures: Tu, Th: 10:00-11:15 a.m. CTH 227 Labs: W: 2:00 - 6:15 p.m. CTH 303 Last updated on Nov 15, 2013 Monday 1. December 2: Tuesday December 3: Wednesday December 4: No labs 2. Thursday December 5: Discuss the syllabus Nuclear chemistry The nucleosynthesis of elements elements ( 1.1, 1.2, 1.3) Frid Decembe December 9: December 10: Periodic table Atomic spectroscopy and quantum mechanics Electron configuration. Sec 1.4, 1.5, 1.6 December 11: 2:00- 6:15 CTH 303 Lab 1 glassblowing December 12: Decembe Multi-electron atoms Sec 1.7, 1.8 & 1.9 Penetration and shielding page The building-up principle page, Penetration and Atomic Parameters. December16: December 17: Octet Rule, Lewis Structures and Structure and bond properties Sec. 2.1, 2.2 December 18: 2:00- 6:15 CTH 303 Lab 1 glassblowing December 19: VSEPR model Sec 2.3. Valence Bond (VB) theory and hybridization Sec 2.4, 2.5, 2.6 GA 1 due in class Decembe December 23: December 24: No class No class Christmas Christmas Break!! Break!! December 25: No class Christmas Break!! December 26: No class Christmas Break!! Decembe No class Christma Break!! December 30: January 31: No class No class Christmas Christmas Break!! Break!! January 1: No class Christmas Break!! January 2: No class Christmas Break!! January No class Christma Break!! January 6: January 7: An introduction to the MO theory Homonuclear diatomic molecules and Bond Properties. Sec 2.7, 2.8 & 2.10 January 8: 2:00- 6:15 CTH 324 MO theory Lab January 9: MO theory continued Exam #1 Review (Ch 1 & 2) January 5. January 13: January 14: Exam #1. (Ch 1 & 2) January 15: CTH 306 2:00- 6:15 Magnetic properties Lab January 16: 3.1 Unit cells and crystals 3.2 The close packing 3.3 Holes in close-packed structures January 6. January 20: No class Martin Luther King Day!! January 21: January 22: CTH 303 Structure of metals Solid-state Lab 1 3.4 Polytypism Basic lattice types of 3.5 Non-close-packed metallic compounds 3.6 Polymorphism of metals 3.7 Atomic radii of metals 3. 4. January 23: Ionic Solids 3.9 Characteristics of ionic solids 3.10 The rationalization of structures January : 3.8 Alloys 7. 8. January 27: January 28: January 29: CTH 303 Solid-state Lab 2 Basic lattice types of ionic compounds January 30: January February 3: February 4: Exam #2. (Ch 2 & 3) February 5: NMR Lab 1 CTH February 6: 4.1 Proton transfer equilibria 4.2 Solvent levelling 4.3 Characteristics of Brønsted acids 4.4 Simple oxoacids 4.5 Anhydrous oxides 4.6 Polyoxo compound formation February February 13: 5.1 Redox half-reactions 5.2 Standard potentials 5.3 Trend in Standard potentials 5.4 The electrochemical series February February 20: The diagrammatic presentation of potential data Latimer diagrams Bonus paper due February February 27: Comprehensive Make-up Test February 324/306 Proton and Carbon Theory and practice 9. 10. 11. February 10: February 17: February 24: February 11: 4.7 Examples of Lewis acids and bases 4.8 Group characteristics of Lewis acids Reactions and properties of Lewis acids and bases 4.9 The fundamental types of reaction 4.10 Hard and soft acids and bases February 12: NMR Lab 2. February 18: Redox reactions, Redox stability In water and oxygen, Disproportionation & comproportionation February 19: CTH 303/306 Cyclic Voltammetry Lab February 25: Exam #3. (Ch 4 & 5) CTH 306/CTH324 Data acquisition and processing CTH 306/CTH324 February 26: No lab