Chapter 10
Energy
Chapter 10
Table of Contents
10.1 The Nature of Energy
10.2 Temperature and Heat
10.3 Exothermic and Endothermic Processes
10.4
Thermodynamics
10.5 Measuring Energy Changes
10.6
10.7
10.8
10.9
Thermochemistry (Enthalpy)
Hess’s Law
Quality Versus Quantity of Energy
Energy and Our World
10.10Energy as a Driving Force
Copyright © Cengage Learning. All rights reserved
2
Section 10.1
The Nature of Energy
Energy
•
•
Ability to do work or produce heat.
That which is needed to oppose natural
attractions.
• Law of conservation of energy – energy
can be converted from one form to
another but can be neither created nor
destroyed.
The total energy content of the universe
is constant.
Return to TOC
Copyright © Cengage Learning. All rights reserved
3
Section 10.1
The Nature of Energy
Energy
• Potential energy – energy due to position or
composition.
• Kinetic energy – energy due to motion of the
object and depends on the mass of the object
and its velocity. Energy is the capacity to do
work.
• Radiant energy - comes from the sun and is
earth’s primary energy source
• Thermal energy - is the energy associated with
the random motion of atoms and molecules
• Chemical energy - is the energy stored within the
bonds of chemical substances
• Nuclear energy is the energy stored within the
collection of neutrons and protons in the atom
Copyright © Cengage Learning. All rights reserved
Return to TOC
4
Section 10.1
The Nature of Energy
Initial Position
•
In the initial position, ball A has a higher
potential energy than ball B.
Return to TOC
Copyright © Cengage Learning. All rights reserved
5
Section 10.1
The Nature of Energy
Final Position
•
After A has rolled down the hill, the potential
energy lost by A has been converted to random
motions of the components of the hill (frictional
heating) and to the increase in the potential
energy of B.
Return to TOC
Copyright © Cengage Learning. All rights reserved
6
Section 10.1
The Nature of Energy
Energy
•
Heat involves the transfer of energy between
two objects due to a temperature difference.
• Work – force acting over a distance.
• Energy is a state function; work and heat are
not:
State Function – property that does not
depend in any way on the system’s past or
future (only depends on present state).
Changes independently of its pathway
Return to TOC
Copyright © Cengage Learning. All rights reserved
7
Section 10.1
The Nature of Energy
State functions are properties that are determined by the state of
the system, regardless of how that condition was achieved.
energy, pressure, volume, temperature
DU = Ufinal - Uinitial
DP = Pfinal - Pinitial
DV = Vfinal - Vinitial
DT = Tfinal - Tinitial
Potential energy of hiker 1 and hiker 2 is the same even though
they took different paths.
Return to TOC
Copyright © Cengage Learning. All rights reserved
8
Section 10.2
Temperature and Heat
Temperature
•
A measure of the random motions of the components
of a substance.
Temperature is a measure of the thermal energy.
Return to TOC
Copyright © Cengage Learning. All rights reserved
9
Section 10.2
Temperature and Heat
Heat
•
A flow of energy between two objects due to a
temperature difference between the objects.
Heat is the way in which thermal energy is
transferred from a hot object to a colder object.
Return to TOC
Copyright © Cengage Learning. All rights reserved
10
Section 10.3
Exothermic and Endothermic Processes
•
•
System – part of the universe on which we
wish to focus attention.
Surroundings – include everything else in
the universe.
Return to TOC
Copyright © Cengage Learning. All rights reserved
11
Section 10.3
Exothermic and Endothermic Processes
open
Exchange: mass &
energy
closed
isolated
Energy exchange
No exchange
Return to TOC
Copyright © Cengage Learning. All rights reserved
12
Section 10.3
Exothermic and Endothermic Processes
Energy Changes Accompanying the Burning of a Match
Return to TOC
Copyright © Cengage Learning. All rights reserved
13
Section 10.3
Exothermic and Endothermic Processes
•
Endothermic Process:
Heat flow is into a system.
Absorb energy from the surroundings.
• Exothermic Process:
Energy flows out of the system.
• Energy gained by the surroundings must
be equal to the energy lost by the system.
Return to TOC
Copyright © Cengage Learning. All rights reserved
14
Section 10.3
Exothermic and Endothermic Processes
Concept Check
Is the freezing of water an endothermic or
exothermic process? Explain.
Return to TOC
Copyright © Cengage Learning. All rights reserved
15
Section 10.3
Exothermic and Endothermic Processes
Concept Check
Classify each process as exothermic or
endothermic. Explain. The system is
underlined in each example.
Exo
a)
Endo
Endo
b)
c)
Exo
Endo
d)
e)
Your hand gets cold when you touch
ice.
The ice gets warmer when you touch it.
Water boils in a kettle being heated on a
stove.
Water vapor condenses on a cold pipe.
Ice cream melts.
Copyright © Cengage Learning. All rights reserved
Return to TOC
16
Section 10.3
Exothermic and Endothermic Processes
Concept Check
For each of the following, define a system
and its surroundings and give the direction
of energy transfer.
a)
b)
Methane is burning in a Bunsen burner in a
laboratory.
Water drops, sitting on your skin after
swimming, evaporate.
Return to TOC
Copyright © Cengage Learning. All rights reserved
17
Section 10.3
Exothermic and Endothermic Processes
Concept Check
Hydrogen gas and oxygen gas react
violently to form water.
Which is lower in energy: a mixture
of hydrogen and oxygen gases, or
water? Explain.
Return to TOC
Copyright © Cengage Learning. All rights reserved
18
Section 10.3
Exothermic and Endothermic Processes
H2 + O2 have higher potential energy than H2O
higher
energy
potential
is absorbed
energy
Electrolysis of Water
Copyright © Cengage Learning. All rights reserved
lower
energy
potential
is given
energy
off
Burning of
Hydrogen in Air
Return to TOC
19
Section 10.4
Thermodynamics
•
•
Study of energy
Law of conservation of energy is often
called the first law of thermodynamics.
The energy of the universe is constant.
Return to TOC
Copyright © Cengage Learning. All rights reserved
20
Section 10.5
Measuring Energy Changes
•
The common energy units for heat are the
calorie and the joule.
calorie – the amount of energy (heat)
required to raise the temperature of one
gram of water 1oC.
Joule – 1 calorie = 4.184 joules
1Cal (food) = 1000 calories
Return to TOC
Copyright © Cengage Learning. All rights reserved
21
Section 10.5
Measuring Energy Changes
Example
Convert 60.1 cal to joules.
60.1 cal
4.184 J
= 251 J
1 cal
Return to TOC
Copyright © Cengage Learning. All rights reserved
22
Section 10.5
Measuring Energy Changes
Energy (Heat) Required to Change the Temperature of a
Substance Depends On:
1. The amount of substance being heated
(number of grams).
2. The temperature change (number of
degrees).
3. The identity of the substance.
Specific heat capacity is the energy required
to change the temperature of a mass of one
gram of a substance by one Celsius degree.
Return to TOC
Copyright © Cengage Learning. All rights reserved
23
Section 10.5
Measuring Energy Changes
Specific Heat Capacities of Some Common Substances
Return to TOC
Copyright © Cengage Learning. All rights reserved
24
Section 10.5
Measuring Energy Changes
To Calculate the Energy Required for a Reaction:
•
Energy (heat) required, Q = s × m × ΔT
Q = energy (heat) required (J)
s = specific heat capacity (J/°C·g)
m = mass (g)
ΔT = change in temperature (°C)
Return to TOC
Copyright © Cengage Learning. All rights reserved
25
Section 10.5
Measuring Energy Changes
Example
Calculate the amount of heat energy (in
joules) needed to raise the temperature of
6.25 g of water from 21.0°C to 39.0°C.
Where are we going?
• We want to determine the amount of energy
needed to increase the temperature of 6.25 g of
water from 21.0°C to 39.0°C.
What do we know?
• The mass of water and the temperature increase.
Return to TOC
Copyright © Cengage Learning. All rights reserved
26
Section 10.5
Measuring Energy Changes
Example
Calculate the amount of heat energy (in
joules) needed to raise the temperature of
6.25 g of water from 21.0°C to 39.0°C.
What information do we need?
• We need the specific heat capacity of water.
4.184 J/g°C
How do we get there?
Q = s m DT
Q = 4.184 J/g C 6.25 g 39.0 C 21.0 C
Q = 471 J
Copyright © Cengage Learning. All rights reserved
Return to TOC
27
Section 10.5
Measuring Energy Changes
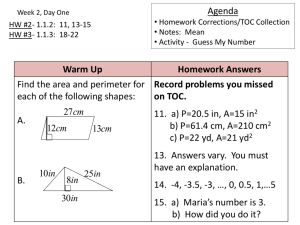
Exercise
A sample of pure iron requires 142 cal of energy to raise its
temperature from 23ºC to 92ºC. What is the mass of the sample?
(The specific heat capacity of iron is 0.45 J/gºC.)
a)
b)
c)
d)
0.052 g
4.6 g
19 g
590 g
Q = s m DT
Q
m=
s DT
142 cal
4.184 J
1 cal
m=
= 19 g
0.45 J/g C 92 C 23 C
Return to TOC
Copyright © Cengage Learning. All rights reserved
28
A
Section 10.5
B
Measuring Energy Changes
100 g water
200 g water
30oC
20
30oC
20
4184 J
8368 J
Twice as much
heat energy is
required to raise
the temperature
of 200 g of
water 10oC as
compared
to
100 g of water.
temperature
heat beakers
rises 10oC
Return to TOC
Copyright © Cengage Learning. All rights reserved
29
Section 10.5
Measuring Energy Changes
Concept Check
A 100.0 g sample of water at 90°C is added
to a 100.0 g sample of water at 10°C.
The final temperature of the water is:
a) Between 50°C and 90°C
b) 50°C
c) Between 10°C and 50°C
Return to TOC
Copyright © Cengage Learning. All rights reserved
30
Section 10.5
Measuring Energy Changes
Concept Check
A 100.0 g sample of water at 90.°C is added to a
500.0 g sample of water at 10.°C.
The final temperature of the water is:
a) Between 50°C and 90°C
b) 50°C
c) Between 10°C and 50°C
Calculate the final temperature of the water.
23°C
Copyright © Cengage Learning. All rights reserved
Return to TOC
31
Section 10.5
Measuring Energy Changes
Concept Check
You have a Styrofoam cup with 50.0 g of water at
10.C. You add a 50.0 g iron ball at 90.C to the
water. (sH2O = 4.18 J/°C·g and sFe = 0.45 J/°C·g)
The final temperature of the water is:
a) Between 50°C and 90°C
b) 50°C
c) Between 10°C and 50°C
Calculate the final temperature of the water.
18°C
Copyright © Cengage Learning. All rights reserved
Return to TOC
32
Section 10.6
Thermochemistry (Enthalpy)
Calorimetry
•
Enthalpy, H is
measured using a
calorimeter.
Return to TOC
Copyright © Cengage Learning. All rights reserved
33
Section 10.6
Thermochemistry (Enthalpy)
An
energy
transformation
whenever a chemical change occurs.
occurs
•
If energy is absorbed during a chemical change,
the products will have more chemical potential
energy than the reactants.
•
If energy is given off in a chemical change, the
products will have less chemical potential
energy than the reactants.
Return to TOC
Copyright © Cengage Learning. All rights reserved
34
Section 10.6
Thermochemistry (Enthalpy)
A sample of a metal with a mass of 212 g is heated to 125.0oC and then dropped
into 375 g of water at 240.0oC. If the final temperature of the water is 34.2oC, what
is the specific heat of the metal?
When the metal enters the water, it begins to cool,
losing heat to the water. At the same time, the
temperature of the water rises. This process continues
until the temperature of the metal and the temperature of
the water are equal, at which point (34.2oC) no net flow
of heat occurs.
Return to TOC
Copyright © Cengage Learning. All rights reserved
35
Section 10.6
Thermochemistry (Enthalpy)
A sample of a metal with a mass of 212 g is heated to 125.0oC
and then dropped into 375 g of water at 240.0oC. If the final
temperature of the water is 34.2oC, what is the specific heat
of the metal?
• Calculate the heat gained by the water.
• Calculate the final temperature of the metal.
• Calculate the specific heat of the metal.
Return to TOC
Copyright © Cengage Learning. All rights reserved
36
Section 10.6
Thermochemistry (Enthalpy)
A sample of a metal with a mass of 212 g is heated to 125.0oC
and then dropped into 375 g of water at 240.0oC. If the final
temperature of the water is 34.2oC, what is the specific heat
of the metal?
Heat Gained by the Water
temperature rise
of the water
Δt = 34.2oC – 24.0oC = 10.2oC
heat gained (375g ) 4.184 J
o
4
(10.2
C)
=
=
1.60 x 10 J
o
by the water
gC
Return to TOC
Copyright © Cengage Learning. All rights reserved
37
Section 10.6
Thermochemistry (Enthalpy)
A sample of a metal with a mass of 212 g is heated to 125.0oC and then
dropped into 375 g of water at 240.0oC. If the final temperature of the
water is 34.2oC, what is the specific heat of the metal?
Heat Lost by the Metal
Once the metal is dropped into the water, its temperature
will drop until it reaches the same temperature as the
water (34.2oC).
temperature drop
of the metal
Δt = 125.0oC – 34.2oC = 90.8oC
heat lost
heat gained
=
= 1.60 x 104 J
by the metal
by the water
Copyright © Cengage Learning. All rights reserved
Return to TOC
38
Section 10.6
Thermochemistry (Enthalpy)
A sample of a metal with a mass of 212 g is heated to 125.0oC and then
dropped into 375 g of water at 240.0oC. If the final temperature of the
water is 34.2oC, what is the specific heat of the metal?
The heat lost or gained by the system is given by:
(mass) (specific heat) (Δt) = energy change
rearrange
heat
specific heat =
mass x Δt
specific heat
=
of the metal
Copyright © Cengage Learning. All rights reserved
1.60 x 10 J 0.831 J
(212g)(90.8oC) g oC)
4
Return to TOC
39
Section 10.6
Thermochemistry (Enthalpy)
Homework
• Reading assignment
– Pages 288 through 302 and chapter review
• Homework Questions and Problems: pages 317 - 319
– 3, 5, 9, 13, 17, 19, 25, 27, 29, 31, 33, 35.
• Due on
Return to TOC
Copyright © Cengage Learning. All rights reserved
40