Poster Gallery Analysis Questions 1. a. Which group/family of

Poster Gallery Analysis Questions
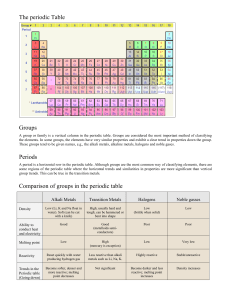
1. a. Which group/family of elements in the periodic table contains the most reactive metals? b. Which group/family of elements in the periodic table contains the most reactive nonmetals? c. Which group/family of elements is the least reactive?
2. Ductility and malleability are both properties of metals. a. Explain the difference between ductility and malleability b. Name some common practical uses of metal elements where ductility and/or malleability are necessary?
3. Silver, gold, and platinum are often used to make fine jewelry; they are also all transition metals. Why are these elements used for fine jewelry? (Consider both the chemical and physical properties of the transition metals)
4 a. What property makes semimetals useful as “switches” to turn a small electric current on and off?
b. As a result of this property, what are some common uses for the semimetals?
5 a. Which group contains mostly synthetic elements? b. What does it mean to be a synthetic element? c. What other chemical property do these elements share?
6. Why is hydrogen often classified in a group of its own?
7. Nitrogen and Oxygen are gases at room temperature; so are the inert gases. Compare the inert gases with that of
Nitrogen and Oxygen.
8. Why are Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorous, and Sulfur particularly important to us? (Hint #1: Did you notice any common uses? Hint #2: Think 7 th grade science)
9. Elements X, Y, and Z are each an element on the periodic table. Using the chart below, try to figure where each belongs on the periodic table. Write the letter “X” in ONE element box on the blank periodic table (Note: there may be multiple correct answers – go with one of them). Then repeat for “Y” and “Z.”
Unknown
Element
Physical Properties Chemical Properties
X Shiny, very malleable, soft solid
Very reactive with chlorine and fluorine; highly reactive with water
Y Shiny, solid; excellent electrical conductor
Less reactive; over many years will react with oxygen to produce a light green compound
Z
Dense gas; glows when electrical current is passed through
Very unreactive; is stable on its own