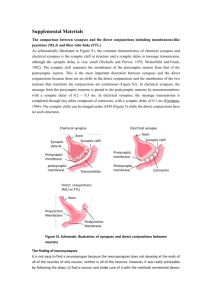
Chemical Electrical
advertisement

SYNAPTIC TRANSMISSION I Tim Murphy NRSC 500, 2011 The definition of synaptic transmission is simply the communication between two nerve cells. Communication believed to involve specialized structures termed "synapses". We will focus on: 1) The discovery of synaptic transmission 2) The form of transmission, i.e. chemical or electrical 3) Criteria for a chemical transmitter 4) Ionic requirement for release 5) Quantal aspects of release: vesicle theory Discovery of synaptic transmission • Cajal's golgi staining methods suggested the presence of contacts between cells that were used for communication ~1900’s. • Sherrington proposed the term "synapse" meaning to clasp to describe the structure, 1890’s. Cajal’s drawings of golgi stain. Shepherd 1997 TINS Sherrington’s insights 1890s. Modern golgi staining, YFP mouse cortical fluorescence, can be bred to other KO’s, transgenics Feng et al. 2000. From Kristin Harris Lectures. http://synapses.mcg.edu/lab/harris/lectures.htm Otto Loewi, chemical transmitter. • 1936 Nobel prize for Medicine • Showed that vagus nerve stimulation liberates a diffusible transmitter. • Perfusate from one stimulated frog heart could be transferred to another and have an effect on beat frequency. Debate on synaptic transmission chemical or electrical. • Otto Loewi showed that acetylcholine could mimic the effect of vagal nerve stimulation. • What additional experiment would have been nice to prove that the vagal nerve released acetylcholine. • The results of Loewi's experiments sparked debate about whether chemical and electrical transmission was occurring. • Subsequently shown that both chemical and electrical transmission exist. From Kristin Harris Lectures. http://synapses.mcg.edu/lab/harris/lectures.htm Chemical Electrical From Kristin Harris Lectures. http://synapses.mcg.edu/lab/harris/lectures.htm From Kristin Harris Lectures. http://synapses.mcg.edu/lab/harris/lectures.htm Electrical Synapses • Gap junction-type communication important for rapidly synchronizing syncytia of cells as is observed in astrocytes, heart, and developing brains. Present in some invertebrates to promote rapid defensive secretions. • Problems with electrical: difficult to modulate gating of channels (exceptions exist cAMP, pH). • Can't change sign, i.e. charge always flows "down hill." • Electrical synaptic transmission requires that the presynaptic cell or terminal be larger than the postsynaptic cell for it to inject considerable charge, no real amplification mechanism. Electrical synaptic transmission. Fundamental Neuro. Chemical transmission inhibitors do not block transmission in developing Zebrafish. Saint-Amant and Drapeau Neuron 2001 Gap junction inhibitors block transmission in developing Zebrafish. Saint-Amant and Drapeau Neuron 2001 Chemical transmission. • Contrary to electrical transmission multiple steps are required to release transmitter chemicals and for them to act on postsynaptic receptors, resulting in a time delay (can be as short as 0.2 msec, from Ca2+ entry to secretion). • Directional, select localization of release machinery to presynaptic terminals and receptors to postsynaptic specializations. • Can change sign by release of inhibitory transmitter. • Highly modulatable as it has many steps presynaptic terminal and at the postsynaptic sites. Chemical Synaptic Transmission. • Definition: Communication between cells which involves the rapid release and diffusion of a substance to another cell where it binds to a receptor (at a localized site) resulting in a change in the postsynaptic cells properties. A hall mark of chemical transmission is a delay between presynaptic Ca2+ elevation and secretion. The delay can be as short as 0.2 ms, but is usually longer due to a variety of factors. Fundamental Neuro. 2002 Steps to chemical synaptic transmission. • First need to bring the presynaptic neuron to threshold at axon hillock. • Conduction down axon, length, R*C dependent. • Opening of voltage gated Ca channels. • Diffusion and action of Ca at release machinery. • Exocytosis and diffusion of transmitter in cleft. • Activation of postsynaptic receptors. Synaptic delays can be less than 0.2 ms from calcium entry (Fund. Neurosci. Chap 8) to the beginning of secretion, but are typically longer when all steps (below) are considered. From Sudhof 2004 Chemical synapse types. • Axosomatic, axoaxonic, axodendritic, and dendrodendritic. • Excitatory (type I) and inhibitory (type II) synapses have different structure in CNS neurons. • CNS synapses usually have one or small number of release sites while nerve muscle synapses have up to 300 active zones. See http://synapses.mcg.edu/atlas Type I Excitatory spherical Type II Inhibitory flattened See http://synapses.mcg.edu/atlas Josef Spacek From Kristin Harris Lectures. http://synapses.mcg.edu/lab/harris/lectures.htm Synapse structure like real estate location, location, location!! Multiple release sites NMJ From Squire et al. Fund. Neurosci 2nd ed. Criteria for a chemical transmitter, make a case for glutamate. • The transmitter substance must be synthesized in the presynaptic neuron. Experiment? • It must be present in the presynaptic terminal and released in amounts sufficient to result in the level of response produced by the endogenous transmitter. experiment? • When applied exogenously the substance should mimic the effect of the endogenous transmitter. experiment? • A specific mechanism must exist for removing the transmitter from the synaptic cleft. experiment? Ionic requirement for release. • Calcium influx is the trigger for fast evoked transmitter release • extracellular calcium (Ca2+) is typically about 1-2 mM, intracellular calcium at rest is <0.1 mM. • An elevation in intracellular calcium concentration is an absolute requirement for transmitter release. Na+ and K+ ions not necessary for release. • How do you test this hypothesis? Ion substitution and pharmacology experiments. • The influx of calcium is triggered by voltage gated ion channels. • Depolarization itself is not needed to stimulate release, as calcium can be elevated by other means (caged calcium chelators). • The relationship between calcium influx and release is related to the "power (exponent)" of calcium entry and is highly nonlinear. For example for a 4th power relationship a doubling of calcium entry can produce a 16 fold increase in release. Slope on a log-log plot indicates power relation, a small change in calcium produces a large change in release. Release~[Ca]^3-5 Slope=5.0 Slope=1.6 from Delaney Enc. of Sci. Neurotransmitter release is triggered by a locally-activated low affinity sensor since bulk cytosolic [Ca2+]i rarely exceeds 10-6 M, yet transmitter release requires much higher [Ca2+]. G. Augustine Curr. Op. in Neurobiol. Squid giant axon and release • Due to its large size the squid giant axon has been used to examine the calcium dependence of transmitter release. • Squid studies using different Ca2+ buffers indicate high concentrations of Ca2+ that are reached for less than 1 ms trigger release (Adler et al. 1991 J. Neurosci.). • Most calcium entry which triggers release occurs during the falling phase of action potential. Why is this advantageous? Rate of calcium binding by buffer (chelator) provides insight into release machinery. • Fast BAPTA (kon 8x108 M -1 *sec -1) buffers block release whereas, • slow EGTA (kon 1x107 M-1*sec -1) buffers do not (Adler et al. 1991 J. Neurosci.). • Time for equilibration of EGTA with calcium~1000 ms versus 12.5 ms for BAPTA. • To estimate the buffer equilibration time take 1/(koff +kon*[Ca2+]), use koff of 8x101 sec –1 for both buffers and 1x10-4 for [Ca2+] at peak. Temporal requirements. • Calcium trigger must be able to act within 0.1 ms of presynaptic stimulation. This requirement restricts the class of chemical events that may be involved in the evoked release process. • Phosphorylation, protein synthesis, gene expression all out. • Everything must be ready and localized- diffusion could not move transmitter vesicles or calcium very far. Calcium channels are clustered on the release face (side with release) of the chick caylx synapse. Patch config. Outside of synapse Release face of synapse Reviewed in EF Stanley TINS 1997 Diffusion time of Ca2+ limits release latency. • Buffered calcium diffusion coeff. are on the order of 200 mm2/sec (D) so calcium could only diffuse a small distance at the most (~0.2 mm) during the synaptic delay (0.0001 s, 0.1 ms), so Ca2+ channels need to be very close to the release machinery. • distance=Sqrt(2Dt) D=diffusion coeff., t=time The diffusion time is dependent on the square of the distance (d). • t=d2/(2*D), where t=time, d=distance, and D=diffusion coefficient. • For 0.1 mm the time is 0.025 ms. • However for 1.0 mm the time jumps by the square to 2.5 ms (100 times longer!), way longer than the release observed latency. These equations are for reference, the concept is of interest. Kd or Km, concentration at ½ max binding or activity. Kd=Km=kdissoc/kassoc affinity=1/km, low affinity means a big km which usually means a large kdissoc Note koff=kdissoc and kon=kassoc Low affinity binding gives rapid off rate. • Concentration for ½ max activity is ~1/affinity and is termed the Kd or Km, if a binding site has a low affinity more ligand is needed to get ½ saturation. • Therefore Kd=koff/kon, assuming a diffusion limited kon of 5x108 M –1 s –1 then koff must be 5x104 s-1 if the Kd is 100 mM. • To estimate the dissociation time constant take 1/(koff +kon*[Ca]) or 20 ms. Note after the channels close [Ca2+] is ~ 1x10-7 and the kon*[Ca] is small compared to koff. These equations are for reference. For reference For a simple reaction. [E]+[S] kon*[S] [ES] koff The time constant for Ca2+ dissoc will be: t=1/(koff+kon*[S]) 20 ms=1/(5x104 s-1 + 5x108 M –1 s –1 *1x10-7 M) Local domains of Ca2+ near channel mouths control transmitter release. It is unclear whether release is always triggered by a single channel or whether multiple ones cooperate. EF Stanley TINS 1997 Transmitter release microdomains. Activation of Ca channels 1.8 mM outside Resting state just after channel closure, bulk Ca is up only 10%. To make full use of microdomains the vesicle must be bound to the calcium channels. Atwood & Shanker Karunanithi DIVERSIFICATION OF SYNAPTIC STRENGTH: PRESYNAPTIC ELEMENTS Nature Reviews Neuroscience 3, 497 -516 (2002). BAPTA, a faster buffer than EGTA more readily blocks synaptic transmission. Given 100 mM Ca2+, BAPTA equilibrates in <20 ms while EGTA takes ~1000 ms. B Atwood & Shanker Karunanithi DIVERSIFICATION OF SYNAPTIC STRENGTH: PRESYNAPTIC ELEMENTS Nature Reviews Neuroscience 3, 497 -516 (2002). Calcium as a local messenger. • Fast channel activation, Ca2+ diffuses to couple excitation to synaptic chemistry. • Low affinity, rapid off rate, and restricted microdomains are characteristics of the calcium flux that evokes release. • Rapid off rate allows for re-loading of release mechanism when Ca2+ levels fall. Affinity=kon/koff • high off rate = low affinity. Quantal aspects of release at the neuromuscular junction. • At the neuromuscular junction small spontaneous potentials (depolarizations) termed miniature end plate potentials are observed. Analogous to CNS miniature excitatory postsynaptic currents (mEPSCs). • The amplitude of evoked responses (due to calcium influx) is always an integer multiple of the unitary response. • Shown that calcium increases the probability of observing a unitary response and not its amplitude. • These data suggested the existence of transmitter quanta or packets. • Evoked transmission mediated by the release of ~ 150 quanta over a 1-3 ms period. Each quantum leads to about 0.5 mV depolarization. From Kristin Harris Lectures. http://synapses.mcg.edu/lab/harris/lectures.htm From Kristin Harris Lectures. http://synapses.mcg.edu/lab/harris/lectures.htm From Kristin Harris Lectures. http://synapses.mcg.edu/lab/harris/lectures.htm Stimulation mini Evoked amplitudes. 1X Miniature event histogram. 2X 4X 1X 3X 4X 2X 1 mV Squire Fund. Neurosci. 2002 CNS synapses and quanta. • At CNS synapses with only a single release site, changing the probability of release (i.e. changing calcium concentration) does not effect the amplitude of the response (as only zero or one vesicle is released in theory). • At CNS synapses with multiple release sites, changing release probability can change the postsynaptic response amplitude as more transmitter is released (graded quantal levels). • At the NMJ a single nerve can elicit a postsynaptic AP given multiquantal release, while at the CNS synapse (with low numbers of release sites) multiple synapses must cooperate, forces a network. CNS synapses and miniature release. • Miniature release is produced in the absence of action potential stimulation. • Thought to reflect the release of single vesicles or transmitter quanta. • Can be stimulated by calcium entry, but may not necessarily require calcium for release. • Commonly studied to gain insight into changes in receptors or release probability during synaptic plasticity experiments, although can be difficult to interpret. Define the number of readily releasable vesicles a synapse has available. A consequence of having of limited number is depletion at high stimulus frequency, CNS synapses may have only a small number of docked vesicles on the order of 5-10 vesicles for a hippocampal CA1 synapse (Harris and Sultan, 1995; Schikorski and Stevens, 1997). From Kristin Harris Lectures. http://synapses.mcg.edu/lab/harris/lectures.htm Many vesicles in the RRP. Few vesicles in the RRP, but Pr high undergoes depression. Remember depression over short timescales can also be caused by other mechanisms including desensitization and autoreceptors. Fundamental Neuro. 2002 Short term plasticity, history dependent changes in responsiveness, typically studied over 10-200 ms intervals. Stim. Residual Ca in the terminal could facilitate transmission if not all quanta are released on the first stimulus (A). If transmission is robust on the first stimulus most readily releasable vesicles will be gone and depression results (B). Squire Fundamental Neurosci. 2002 Response types at single CNS synapses with different #s of release sites. 1 release site 2 release sites 2 vesicles 1 vesicle failure all or postsyn response? 1 vesicle failure Voltage Time postsyn amplitude variation? Electrode See sum of all synapses When multiple synapses (or release sites) are involved facilitation can reflect an increase in release probability (all or none secretion) at single synapses that can look like a graded increase in release. Electrode sum of all synapses Trial 1 Electrode See sum of all synapses Trial 2, 25 ms later Readings • Neuroscience 4th Ed. Purves Chapters 4-6 optional • Fundamental Neuroscience 1st Ed. Chapters 7 and 8 (for Neurochem also), 2nd Ed. Chapters 7 and 8, 3rd Ed. Chapters 7,8. • Delaney, Kerry R (March 2000 ) Calcium and Neurotransmitter Release. In: Encyclopedia of Life Sciences, London: Nature Publishing Group, http://www.els.net/doi:10.1038/npg.els.0000027 • Harold L. Atwood & Shanker Karunanithi Diversification of synaptic strength: presynaptic elements. Nature Reviews Neuroscience 3, 497 -516 (2002). Advanced review comprehensive. • For great EM pictures of synapse see Josef Spacek’s site http://synapses.mcg.edu/atlas