Chemical Equations & Stoichiometry Lesson
advertisement

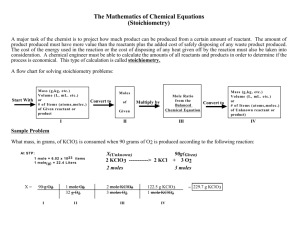
IN OUR PREVIOUS CLASS ABOUT CHEMICAL EQUATIONS, WE FOUND THAT AN EQUATION CAN BE USED TO DESCRIBE A CHEMICAL REACTION. IT GIVES US A GREAT DEAL OF INFORMATION: CH4 + 2O2 CO2 + 2H2O 1 MOLECULE 2 MOLECULES METHANE OXYGEN 1 MOLECULE 2 MOLECULES CARBON DIOXIDE WATER 1 MOLE 2 MOLES 1 MOLE 2 MOLES 16 g 64 g 44 g 36 g 6.02 x 1023 Molecules 2(6.02x1023) molecules 6.02 x 1023 molecules 2(6.02 x 1023) molecules THE MOST USEFUL RELATIONSHIIPS WILL BE THE MOLAR RATIOS INVOLVED IN THE REACTIONS AND THE MASS RATIOS. WE CAN USE THIS INFORMATION TO CALCULATE: 1. THE AMOUNTS OF STARTING MATERIALS NEEDED TO MAKE A GIVEN AMOUNT OF PRODUCT 2. THE THEORETICAL YIELD (100%) THAT GIVEN AMOUNTS OF PRODUCTS WOULD GIVE 3. THE LIMITING REAGENT IN A REACTION CONSIDER THE FOLLOWING PROBLEM. HOW MANY MOLES OF OXYGEN CAN YOU PRODUCE FROM 5 MOLES OF KClO3? 5 moles 2KClO3 2KCl 2 moles + X moles 3O2 3 moles THE EQUATION TELLS YOU THAT 2 moles OF KClO3 WILL GIVE YOU 3 moles OF O2. YOU HAVE 5 MOLES AND NEED TO FIND HOW MUCH O2 WILL BE PRODUCED. USE THE RATIO: 5/2 = X/3 X = 3(5/2) = 7.5 moles AND: YOU CAN ALSO USE YOUR EQUATIONS IN TERMS OF MASS. HOW MANY GRAMS OF WATER ARE PRODUCED IN THE OXIDATION OF 1 GRAM OF GLUCOSE? C6H12O6 + 6O2 6CO2 + 6H2O (combustion reaction) 1 MOLE GLUCOSE 6 MOLES WATER LET’S CALCULATE THE MOLAR MASSES OF WATER AND GLUCOSE SO WE CAN GET THE MASS RELATIONSHIPS. The molar mass of glucose is: 6 C = 6 x 12.0 = 72.0 g 12 H = 12 x 1.01 = 12.12 g 6 O = 6 x 16.0 = 96.0 g molar mass = 180.0 g/mole The molar mass of 2 H = 2 x 1.01 1 ) = 1 x 16.0 molar mass water is: = 2.02 g = 16.0 g = 18.02 g/mole 1.0 g glucose X g water C6H12O6 + 6O2 6CO2 + 6H2O 180 g/mole 6(18 g/mole) 1/180 = X/108 OR X = 108/180 = 0.6 grams WATER The decomposition of KClO3 (potassium chlorate) is commonly used to prepare small amounts of O2 in the laboratory: 2 KClO3 (s) 2 KCl (s) + 3 O2 (g) How many grams of O2 can be prepared from 4.50 g KClO3? FIRST, THE FORMULA MASS OF KCLO3 IS: 1 K = 1 x 39.1 = 39.1 g 1 Cl = 1 x 35.5 = 35.5 g 3 O = 3 x 16.0 = 48.0 g formula mass = 123 g/mole THE FORMULA MASS OF O2 IS 2 X 16 = 32 g/mole 4.5 g 2 KClO3 2(123 g/mole) 2 KCl + X g 3 O2 3(32 g/mole) SO, YOUR RATION IS: 4.5/246 = X/96 AND X = (4.5/246) x 96 = 1.76 g OXYGEN 2 KClO3 (s) 2 KCl (s) + 3 O2 (g) SOLID LITHIUM HYDROXIDE IS USED IN SPACE VEHICLES TO REMOVE EXHALED CARBON DIOXIDE. THE LITHIUM HYDROXIDE REACTS WITH GASEOUS CARBON DIOXIDE TO FORM SOLID LITHIUM CARBONATE AND LIQUID WATER. HOW MANY GRAMS OF CARBON DIOXIDE CAN BE ABSORBED BY 1.00 G OF LITHIUM HYDROXIDE? YOU NEED THE BALANCED EQUATION, SO 2 LiOH (s) + CO2 Li2CO3 (s) + H2O (l) The formula mass of LiOH is: 1 Li = 1 x 6.94 = 6.94 g 1 O = 1 x 16.0 = 16.0 g 1 H = 1 x 1.01 = 1.01 g formula mass = 24.0 g and you have 2, so 2 x 24.0 = 48.0 g The molar mass of CO2 = 12.0 + 2 x 16.0 = 44.0 g 1.0 g X g 2 LiOH (s) + CO2 Li2CO3 (s) + H2O (l) 48 g 44.0 g So, 44 / 48 = X / 1.00 and X = (44/48) x 100 X = 0.917 g http://www.gpb.org/chemistry-physics/chemistry/801