Oxidation-Reduction (Redox) Titration of Oxalate in K 3 Fe(C 2 O 4 )
advertisement

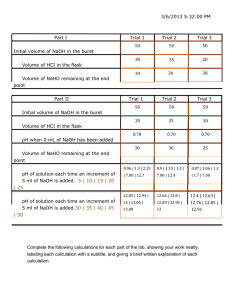
Oxidation-Reduction (Redox) Titration of Oxalate in K3Fe(C2O4)3·3H2O First things first… Safety: Put bags away Goggles Lab Jacket Gloves Acidic Reactants! Quiz! LAB! What is titration anyway? Using a buret, you can deliver a specific volume of a solution for a reaction To read a volume in a buret, look at the bottom of the meniscus Be sure to note the start volume and the final volume. No bubble in tip! Procedure WEAR GLOVES! Permanganate will stain Weigh 0.1g of the potassium trioxalatoferrate into 3 separate 125 or 250 mL Erlenmeyers Make sure to record mass to 0.00Xg (3 significant figures) Add ~20 mL of 1.0 M Sulfuric acid Rinse buret with two 5 mL portions KMnO4, and then fill halfway with (X.XX M) KMnO4 Titration! Repeat for a total of 3 titrations Actual Titration Place the Erlenmeyer on a hot plate and stir until it dissolves and temp 80-90ºC Titrate KMnO4 from the buret, temp >60ºC Keep stirring As the endpoint approaches, titrate SLOWLY, drop-wise until the sol’n becomes pink (reaches end-point) Waste: KMnO4 in buret goes to beaker back hood Titrated solution goes down the sink Calculations 2 5C2O4 2MnO4 16 H 2Mn Moles KMnO4 reduced 2 10CO2 8H 2O VolKMnO4 ( L) MolarityKM nO4 (mol ) XmolesKMnO 1 ( L) 2 Moles C2O4 oxidized Mass C2O4 XmolesKMnO4 5molC2O4 2 YmolC 2O4 1 2molKMnO4 YmolC 2O4 1 2 88.02 gC2O4 MW 2 C2O ZgC2O4 2 1molC2O4 4 2 Weight % 2 ZgC2O4 100% weight % __ gCordComp Sample mass originally weighed (~0.1xxg) Everything due before leaving lab Turn in today: pre-lab with calculations (yellow sheet) Finish calculations, including % error Include pages from lab manual if yellow pages are not easy to read Enter data into computer/spreadsheet Do initial log in Password: last 3 digits g number Then one individual member adds data Group members: group id is the individual id who entered data Due Next Class Freezing point depression quiz and pre-lab Lab Report: Data analysis (based upon lab handout “Redox Titration of Oxalate” on chem.gmu.edu/results site) 1-2 page double spaced discussion/conclusion page Do not use discussion ?s