PPT
advertisement

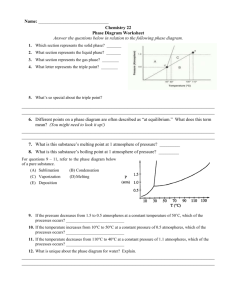
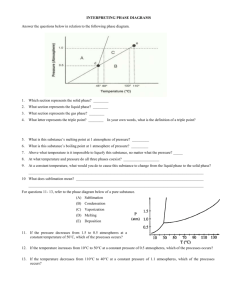
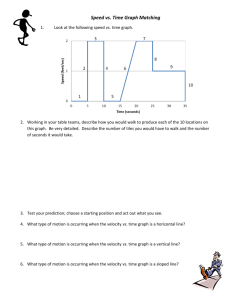
Dust Storms Mars and Earth Bit of Administration …. • Reading – BSNV Chaps. 10, 11 • 12-week exam! – Tonight, 7:15-8:30 pm, 3425 Sterling Hall • Mathieu office hours for pre-test questions – All day today Nuclear Fusion - A Digression Thermonuclear Hydrogen Fusion 4 H => He Why “Thermonuclear”? Bringing two positive charges together Requires high velocity = high temperature Hydrogen Deuterium Positron Neutrino Helium 3 Helium 4 Nuclear Fusion - A Digression Thermonuclear Hydrogen Fusion D + T => He + n Nuclear Fusion - A Digression Thermonuclear Hydrogen Fusion D + T => He + n Nuclear Fusion - A Digression Thermonuclear Hydrogen Fusion D + T => He + n Nuclear Fusion - A Digression Thermonuclear Hydrogen Fusion D + T => He + n Advantages • • • • Fuel source - deuterium, tritium from water - tritium also from reactors No risk of nuclear accident No air pollution Minimal radioactivity? The “Big Idea” of Solar System Evolution The Second Law of Thermodynamics Heat can never pass spontaneously from a colder to a hotter body. As a result of this fact, natural processes that involve energy transfer must have one direction, and all natural processes are irreversible. Profound Consequence: Equilibrium requires an Energy Source Planetary Atmospheres Earth 77% N2, 21% O2 15 oC 1 atmosphere Mars Venus Mercury Trace of He, Na, O 95% CO2 -50 oC 0.01 atmospheres 96% CO2 470 oC 90 atmospheres Moon Trace of He, Na, Ar Planetary Atmospheres • Retention of Atmosphere • Atmospheric particles (atoms, molecules, dust) respond to gravity in same way as any body Atmosphere will escape if velocities of atmospheric particles (THERMAL VELOCITY) are greater than ESCAPE VELOCITY of planet Planetary Atmospheres • Escape Velocity - “a threshold velocity” • Escape Velocity - Minimum speed at which two objects will not be held together by their mutual gravity 2GM Vescape R M R Planetary Atmospheres • Thermal Velocities of Atmospheric Particles Values for speeds are just for example - depends on temperature Planetary Atmospheres • Peak Thermal Velocity • Note Vthermal increases with higher T • Note Vthermal decreases with higher mparticle 2kT Vthermal m particle mparticle = mass of atom or molecule T = temperature in oK k = Boltzmann constant = 1.4 x 10-23 joules/K Planetary Atmospheres • Peak Thermal Velocity Same mass particles Low temperature Medium temperature High temperature 0 Velocity of particles Planetary Atmospheres • Peak Thermal Velocity Same temperature High mass particles (e.g., CO2) Medium mass particles (e.g., H2O) Low mass particles (e.g., H) 0 Velocity of particles Planetary Atmospheres • Peak Thermal Velocity Planetary Atmospheres • Retention of Atmosphere Rule of Thumb - if Vesc > 5 x Vthermal, then retain an atmosphere Surface Escape Tension Velocity of drop of Water Escape Velocity Low temperature Medium temperature High temperature Escape Evaporation from Escape fromPlanet Planet 0 Velocity of particles Planetary Atmospheres • Retention of Atmosphere • Example - consider Earth and Moon - T ≈ 300 K H = 1.7 x 10-27 kg O = 2.7 x 10-26 kg For H, For H2O, H2O = 3.0 x 10-26 kg Vthermal = 2 x 1.4x10 -23 x 300 2300 m/sec = 2.3 km/sec -27 1.7 x 10 Vthermal = 2 x 1.4x10 -23 x 300 500 m/sec = 0.5 km/sec -26 3.0 x 10 Planetary Atmospheres • Retention of Atmosphere • Example - consider Earth and Moon - T ≈ 300 K Vescape, Earth = 11.2 km/sec Vescape, Moon = 2.4 km/sec No H, retains H20 No H, no H20 For H, Vthermal = 2.3 km/sec For H2O, Vthermal = 0.5 km/sec ConcepTest! The escape velocity of Mercury is 4.3 km/sec. The escape velocity of Mars is 5.0 km/sec. Mercury does not have a substantial atmosphere; Mars has an atmosphere. This is primarily because A) B) C) D) Mars is further from the Sun so any gases are much colder Mars has a 20% higher escape velocity CO2 has more mass on Mars than on Mercury Mars is a dynamically active planet with active volcanoes Planetary Atmospheres • Greenhouse Effect 90 atmospheres! Planetary Atmospheres • Greenhouse Effect 240 watts/m2 in Small fraction escapes into space 240 watts/m2 out 50% reflected by clouds and surface CO2 H20 Planetary Atmospheres • Greenhouse Effect Energy In 100 energy units 100% gate Energy Out 200 energy units stored in equlibrium 100 energy units 50% gate Planetary Atmospheres • Greenhouse Effect Energy In 100 energy units 100% gate Energy Out 10, 000 energy units stored in equlibrium 100 energy units 1% gate Planetary Atmospheres • Greenhouse Effect