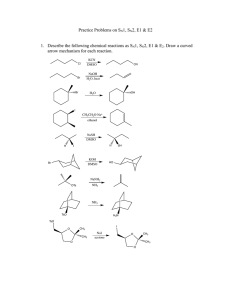
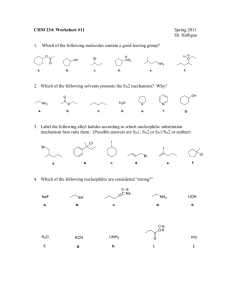
Topic 20. Organic chemistry
advertisement
Topic 20 - Organic chemistry • • • • • • Introduction- functional groups and naming Nucleophilic substitution reactions Elimination reactions Condensation reactions Reaction pathways Stereoisomerism 20.1 Ester • • • • Functional group: -COOCCondensation reaction or esterification Many fruit-smelling esters The bond in triglycerides (lipids) • Alcohol + carboxylic acid ester + water • CH3-OH + HOOC-CH3 CH3OOC-CH3 + H2O (The underlined forms water) Esterification Butanoic acid Methanol Name: – The alcohol part: Stem + yl: Methyl– The acid part: the salt name; -buthanoate => Methyl-buthanoate Which names? Which names? Ethylethanoate 2-butylethanoate propylethanoate penthylpropanoate Amine • Relatives to ammonia => weak bases • Functional group –NH2 • H-bonds => higher bp’s, smaller ones are water-soluble etc. • Name: stem + suffix: -ylamine (or prefix amino-) – Methylamine CH3-NH2 – Ethylamine CH3-CH2-NH2 Amide • Functional group: -CONH • Name: stem + suffix: -anamide – Methanamid H-CONH2 – Ethanamid CH3-CONH2 • Peptide bond in proteins Nitrile • Functional group: -CN • Former name: cyanides • Name: stem + suffix: nitrile – Metanenitrile: HCN – Ethanenitrile: CH3-CN Nucleophiles and electrophiles- often needed in organic reactions • Nucleophile- nucleus lover • Has free electronpair and whole or part negative charge • The larger the negative charge - the better the nucleophile • Eg: C=C, H2O, -OH, -CN, NH3 • Electrophile-electron lover • Has whole or part positive charge • The larger the positive charge - the better the Electrophile • Eg: C=O, H+, C-Cl, H H H H C 20.2 Nucleophilic substitution reactions H C P r im a r y C w it h C l a n d 2 H Cl Nu: + R-X Nu-R + X: H H H Cl H C C C H H H H Cl H C C C H C H3 H H S e c o n d a r y C w it h C l a n d 1 H H T e r t ia r y C w it h C l a n d 0 H It’s important to know the difference of these because they will undergo different forms of nucleophilic substitution reactions SN2 or SN1? • • • • • S N2 Bimolecular = two species in the rate determining step. Rate = k [org]*[Nu] Primary halogenoalkanes Steric hindrance One-step with transition state Inversion of configuration S N1 • Monomolecular = one species in the rate determining step. Rate = k [org] • Tertiary halogenalkanes • Heterolytic fission of substrate rate determining step • Formation of inermediate carbocation • Racemix mixture formed Factors affecting the rate • The halogen in the halogenoalkane is important for the reaction rate: C-I > C-Br > C-Cl > C-F (cf. bond strength) • The stronger the nucleophile, the faster the SN2-reaction CN- > OH- > NH3 > H2O SN2 substitution reaction with ammonia H3N: + R-X R-NH2 + HX Forming an amine SN2 substitution reaction with CN- as nucleophile R-X + CN- R-CN + X- Forming a nitrile The carbon chain will be prolonged with one carbon atom Reactions with nitriles • Nitrile + H2 Ni catalyst Amine CH3CH2-CN + H2/Ni CH3CH2-NH2 • Nitrile + Acidic water Carboxylic acid CH3CH2-CN + H+/H2O CH3CH2-COOH 20.3 Elimination reactions • CH3CH2-Br + OH- CH2=CH2 + H2O + BrCondition: Hot and concentrated sodium hydroxide solution in ethanol. Reflux. • Opposite to addition reaction • 2 kinds; E1 and E2 • If the conditions are different: A substitution reaction CH3CH2-Br + OH- CH3CH2-OH + BrWill happen in warm dilute solution of sodium hydroxide, the hydroxide ion will work as a nucleophile. The E1 mechanism (similar to SN1) • Involves heterolytic fission of C-X bond • Tertiary halogenalkane • Via a carbocation intermediate The E2 mechanism (similar to SN2) • Concerted bimolecular reaction • Primary halogenalkane • Via transition state Dehydration reaction of alcohol to form alkene • E1 or E2? • Excess of H2SO4, ~170oC 20.4 Condensation reaction • 2 Organic molecules 1 Organic molecule + water • Acid catalysed • Esterification, etherification • Common in biochemistry, formation of polysaccharides, proteins, nucleic acids Esterification H O H H H H C C H H O H C C H+ H C C H H E th a n o l OH HO C C H E t a n o ic a c id H H O E t y le t h a n o a t e Name: R-yl R’-oate Esters: flavouring agents (food, perfume), solvents, plastics (polyesters) H H + H2O W a te r Amide condensation H H H C C H H HO N H2 O H C C H H H Amine Carboxylic acid H H C C H H C H3 A la n in e COH HN C C H + H H 2O H Water O O CH H Amide H H2N N O CH H G ly c in e COOH H2N CH C H3 C N C H2 H D ip e p t id e o f a la n in e a n d g ly c in e COOH + H2O Polycondensations Plastics • Polyesters: polyethylene terphtalate (PET) • Polyamides; nylone Benzene-1,4-dicarboxylic acid + Ethane-1,2-diol Can condense with a diol Can condense with a dicarboxylic acid Monomers with two functional groups are required Polyamides HOOC-R-COOH + H2N-R’-NH2 H2N-R’-NH-OC-R-COOH Di-amine amide bond HOOC-(CH2)4-COOH + H2N-(CH2)6-NH2 HOOC-(CH2)4-CONH-(CH2)6-NH2 + water hexanedioic acid + 1,6-diaminohexane Nylon 20.6 Reaction pathways 1. Elimination reaction. Hot, concentrated and reflux 2. Substitution reaction. SN1 or SN2 3. Substitution reaction. SN1 or SN2. (Can be substituted up 4 times to a quarternary ammonum salt) 4. Condensation reaction. Acid catalyst (or alkaline catalyst, but more common when hydrolysis of ester). Equilibrium reaction. 5. Condensation reaction. Acid catalyst (or alkaline catalyst). Equilibrium reaction. 6. Nitrile to amin: Reduction with H2 and Ni-catalyst 20.5 Stereoisomerism • Different location in space of atoms or groups Structural isomers • Chain: e.g. n-butane : methylpropane • Positional: e.g. 1-propanol : 2-propanol • Functional groups: Ethanoic acid, CH3-COOH : Methyl methanoate HCOOCH3 Propanal, CH3-CH2-CHO : Propanone, CH3-CO-CH3 Geometric • Double bond = p and s bonds. No free rotation around a double cis-2-buten trans-2-buten • cis-1,2-dichloroethane: bp = 60.3oC • trans-1,2-dichloroethane: b.p. = 47.5oC cis-but-2-ene-1,4.dioic 1,4.dioic trans-but-2-ene- m.p = 286oC m.p.= 130oC with decomposition. • Strong H-bond between molecules in trans. Strong H-bond in the molecule in cis. Cyclic compounds can also give geometric isomers • Cis and trans Optical mirror plane enantiomers to each other Stereo isomers Different location in space. Geometric cis-2-buten trans-2-buten Optical mirror plane enantiomers to each other • A carbon with 4 different substituents is said to have an asymmetric centre or to be chiral. • Chiral can also mean the whole molecule. • Enantiomers have the same physical properties except that they rotate the plane of polarised light in different directions. • Very important which form of the enantiomer in biology and medicine. Polarimeter • Light: Electromagnetic radiation. Normal light oscillating in all directions. • Plane-polarized light: When normal light is sends throuhg a polarizing filter only waves in the same plan can pass. If two polarizing filters places 90o to each other the light will be compleataly blocked. • Polarimeter: Measure how much the light will rotate when pass through a solution with optically avtive substance. A sample holder between two filter