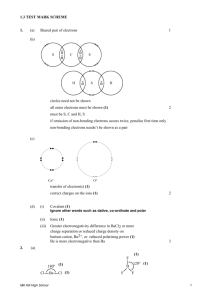
hydrogen bond
advertisement

Chapter 4 Nanomaterials & Bonding NANO 101 Introduction to Nanotechnology 1 Quiz Wednesday • • • • • MC/Short Answer (10 questions, 30 pts) Free Response (3 questions, 24 pts) Calculations (3 questions, 21 pts) Formula sheet, similar to extra practice Bring a scientific calculator, not a cellphone, graphing calculators are okay • You will have 2 hrs, shouldn’t take the entire time. 2 Materials • Matter with a useful purpose Matter is… • Anything that has mass and occupies space • Made up of atoms and/or molecules • To make molecules and solids – need chemical bonds 3 Models of the Atom J.J. Thomson Rutherford Proton Neutron Current model Electron Charge Mass (kg) Location ~1.673*10-27 ~1.675*10-27 ~9.11*10-31 nucleus nucleus “clouds” 4 Covalent Bonding • Minimum energy: Energy • Attractive and repulsive forces cancel out • Binding Energy • Separation between atoms when bonded • Interatomic distance Interatomic Distance • Bond length http://voh.chem.ucla.edu/vohtar/winter02/20A/lecture5.htm 5 Covalent Compounds Atoms share electrons to form molecules H + H H H H H H2 Bonding electrons F + F F F F F F2 Lone pair electrons Covalent bonds usually between nonmetal + nonmetal 6 Ionic Bonding • • • • Forms salts Transfer of electrons Electrostatic attraction No sharing of electrons Non-Metal: Accepts electron anion Metal: Donates electrons Fluorine Sodium cation 7 Ionic solids • Examples: NaCl (table salt), (NH₄)₂CO₃ • Properties: – High melting point NaCl – Very hard – Poor conductivity – Water soluble 8 Organic Compounds • • • • C-C bond length ~ 0.15 nm C-H bond length ~ 0.11 nm Simplest organic molecule: methane CH4 Octane end H to end H = 1.6 nm Covalent Solids • Examples: Diamond, Graphene, Silicon carbide, Boron Nitride SiC in bulletproof vests • Properties: Diamond = carbon molecular solid – Hard – High melting point – Low conductivity (graphene is exception) 10 Ionic/Covalent Scale • Electronegativity – How much an atom wants to gain electrons. Same electronegativity Very different electronegativity 11 Metallic Bonding Atom cores • Electrical Conductors • Thermal Conductors • Ductile materials Sea of valence electrons 12 Intermolecular Forces Van der Waals forces • dipole-dipole • if between H & N, O, F hydrogen bond H H O H O H O H H • dipole-induced dipole • induced dipole-induced dipole (London Dispersion forces) 13 Graphite • Intramolecular vs intermolecular – Solid lines = covalent bonds = intramolecular foce – Dotted lines = Van der Waals forces = intermolecular force 14 Strength of London Dispersion • Propane • Octane • Paraffin 15 Quiz Wednesday • • • • • MC/Short Answer (10 questions, 30 pts) Free Response (3 questions, 24 pts) Calculations (3 questions, 21 pts) Formula sheet, similar to extra practice Bring a scientific calculator, not a cellphone, graphing calculators are okay • You will have 2 hrs, shouldn’t take the entire time. 16 Small Structures • Particles • Wires • Films, Layers, Coatings • Porous Materials • Small Grained Materials • Molecules 17 Particles • Small particles are mostly surface • Bulk solids typically < 1% surface atoms • Small nanoparticles can have ~90% surface atoms • Van der Waals forces dominates • Mostly interparticle interactions (fewer bonds) • More reactive • Useful as catalysts Source: Younan Xia, Washington University Source: Seoul National University 18 Wires / Tubes Source: Evans Group, University of Leeds • • • • Source: Science Buzz, Science Museum of Minn. Electronics Optics Strength-based applications Can characterize by aspect ratio (length/(width or diameter)) 19 Films, Layers, Coatings Surface is where all interactions and reactions take place. Self-Assembled Monolayers: Ordered arrangement of molecules that occurs spontaneously tail http://phys.org/news85328131.html#nRlv backbone head 20 Molecular Self Assembly Mechanisms for much chemistry Happens due to: • Intermolecular interactions – Hydrogen bonding – hydrophobicity and hydrophilicity – Solvency “like dissolves like” • Specific covalent reactions – Functionalization – Ex. Thiols and coinage metals Looking Ahead • Lab #1 due Thursday 4/23 • Homework #3 due Monday 4/27 22 Pre-Lab for Thursday • Bottom – up Nanoparticle Synthesis – Starting with molecular precursors – Chemistry! Will build the nanoparticles • Comprehensive study on synthesis and properties of colloidal gold published by Faraday (1857) • Classic method (1985, Turkevich) – Precursor: dilute chlorauric acid (HAuCl4) – Reducing agent: sodium citrate (NaC6H5O7) – Reaction temperature: 100 °C – Product: stable, uniform, ~20 nm particles 23 Gold NP Biosensors 24 Make a salt or sugar sensor • What type of bonds hold the citrate to the gold nanoparticle? • What is the difference between sugar dissolved in water and salt dissolved in water? Dalton Trans., 2014,43, 5054-5061 • Observe color visually • Observe color with spectrometer http://www.cytodiagnostics.com/store/pc/Gold-Nanoparticle-Properties-d2.htm 25