Atomic Theory (Part 1)
advertisement

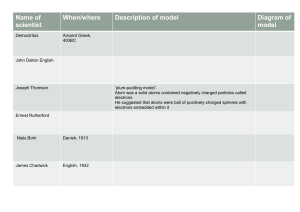
Name: Date: Period: Atomic Theory and Bohr Model – Review Worksheet Due: Friday 4/11/14 Name that Scientist: Write the letter on the line matching the correct scientist to the described contribution listed below that they played in the history of the Atomic Theory. A) Democritus B) John Dalton C) Dmitri Mendeleev D) JJ Thomson E) Ernest Rutherford F) Niels Bohr 1. Proposed that atoms had a dense positively charged nucleus surrounded by electrons. 2. His model of the atom was called the “Plum Pudding” model. 3. Was the first to propose the existence of atoms. 4. Said that electrons could only exist in certain orbits and at certain energy levels. 5. Came up with the theory that all substances were made of atoms. 6. Known as the father of the modern day Periodic Table Answer the questions below 7. Today’s model of the atom is a) “Plum Pudding” Model b) Cloud Model 8. The word “Atomos” means not to be c) Bohr Model . d) Billiard Ball Model