Density
advertisement

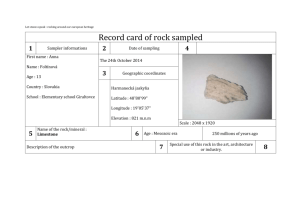
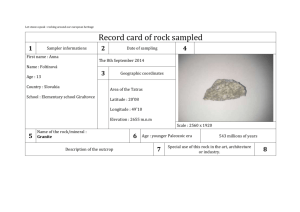
1. How can you compare the densities of liquids without calculations? 2. What is the difference between the mass and volume of an object? 3. What is density? 4. If you have 30.76 mL how many cubic centimeter are there? 5. What instrument is used to determine liquid volume? 6. A rectangular substance has a length of 4.0 cm, a width of 2.0 cm and a height of 3.0 cm. It has a mass of 10.5 g. What is the density? 7. What would be the density if the volume of the substance was 3 ml? Density • Mass per unit volume • Density = mass divided by volume: D=M/V • Units are g/mL or D= 3 g/cm • Density of water is 1 g/ml • Objects that are more dense than water will sink –Example: 2.78 g/ml • Objects that are less dense will float – Example: 0 .8 g/ml •Everything has its own density. •Density is not affected by the size of an object and can be used to identify what an object is made of – Ex. •Density of copper is 8.9 g/ml •Density of aluminum is 2.7 g/ml 1. A rectangular solid of unknown density is 5 cm long, 2 cm high and 4 cm wide. The mass of this solid is 300 g, calculate its density. 2. A rock occupies a volume of 20 cm3 and has a mass of 54 grams. Find the density of this rock. 3. A rock has a density of 4 g/ml. and a mass of 16 g. What is the volume this rock occupies? 4 An unknown substance from planet X has a density of 10 g/ml. It occupies a volume of 80 ml. What is the mass of this unknown substance? 5. What would be the density of 5 g of the substance from planet X in problem 3? 6. A cube made of an unknown material has a height of 2 cm and a mass of 9 g. Calculate the density of this cube given this information Actual Density Copper Brass Steel Aluminum Acrylic Nylon Pine PVC 8.9 g/ml 8.0-8.5 g/ml 7.6 g/ml 2.7 g/ml 1.16-1.19 g/ml 1.13 g/ml .35-.60 g/ml 1.39-1.42 g/ml