Diapositiva 1 - Universidad Autónoma de San Luis Potosí
advertisement

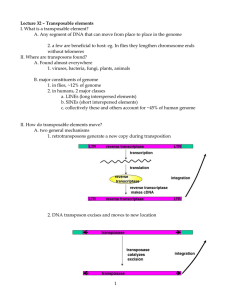
Transposons CA García Sepúlveda MD PhD Laboratorio de Genómica Viral y Humana Facultad de Medicina, Universidad Autónoma de San Luis Potosí 1 Session #25-26 Transposons Introduction • Genomes evolve both by rearranging existing sequences and by acquiring new sequences. • Rearrangements are sponsored by internal genomic events. – Unequal recombination (non-recirpocal) results from mispairing by the cellular mechanisms for homologous recombination. – Results in duplication or rearrangement of loci (Clusters & repeats). – Duplication of sequences within a genome gives rise to further duplication. 2 Session #25-26 Transposons Introduction • Genomes evolve both by rearranging existing sequences and by acquiring new sequences. • Results from the ability of vectors to carry information between genomes. Plasmids move by conjugation. Extrachromosomal elements move information horizontally by mediating the transfer of short lengths of genetic material. 3 Session #25-26 Transposons Introduction • Genomes evolve both by rearranging existing sequences and by acquiring new sequences. • Results from the ability of vectors to carry information between genomes. Phages spread by infection. Both plasmids and phages occasionally transfer host genes along with their own replicon. 4 Session #25-26 Transposons Introduction • Genomes evolve both by rearranging existing sequences and by acquiring new sequences. • Results from the ability of vectors to carry information between genomes. – Direct transfer of DNA occurs between some bacteria by means of transformation. 5 Session #25-26 Transposons Introduction • Genomes evolve both by rearranging existing sequences and by acquiring new sequences. • Results from the ability of vectors to carry information between genomes. In eukaryotes, some viruses (notably the retroviruses) can transfer genetic information during an infective cycle. 6 Session #25-26 Transposons Introduction • Genomes evolve both by rearranging existing sequences and by acquiring new sequences. • Another major cause of variation is provided by transposable elements or transposons: – these are discrete sequences in the genome that are mobile & able to transport themselves to other locations within the genome. – Found in both eukaryotes & prokaryotes. – Selfish DNA with the sole purpose of autoreplication. Visit: http://highered.mcgraw-hill.com/sites/0072556781/student_view0/chapter13/animation_quiz_5.html 7 Session #25-26 Transposons Introduction • Relationship of the transposon to the genome resembles that of a parasite with its host. • The propagation of an element by transposition is balanced by the harm done if a transposition event inactivates a necessary gene. • Any transposition event conferring a selective advantage will lead to preferential survival of the genome harboring the transposon ! 8 Session #25-26 Transposons Transposons • Transposons do not utilize an independent form (such as virus or plasmid DNA). • Move directly from one site of the genome to another. • Unlike other processes involved in genome restructuring, transposition does not rely on homology between donor and recipient sites. • Sometimes transfer contiguous host sequences to new sites elsewhere within the same genome as they move. • They are an internal counterpart to the vectors that can transport sequences from one genome to another. 9 Session #25-26 Transposons Transposons • Transposable elements can promote rearrangements of the genome, directly or indirectly: – Directly: The transposition event itself may cause deletions or inversions or lead to the movement of a host sequence to a new location. – Indirectly: Transposon sequences serve as substrates for cellular recombination systems by functioning as "portable regions of homology"; • two copies of a transposon at different locations (even on different chromosomes) may provide sites for reciprocal recombination resulting in deletions, insertions, inversions, or translocations. 10 Session #25-26 Transposons Transposons • They may provide the major source of mutations in the genome! • Two general classes of transposons: – DNA transposons Exist as sequences of DNA coding for proteins that are able directly to manipulate DNA so as to propagate themselves within the genome. – RNA transposons They are related to retroviruses and move as a consequence of their ability to make DNA copies of their RNA transcripts, the DNA copies then become integrated at new sites in the genome. 11 Session #25-26 Transposons Transposons • Transposons carry gene(s) that code for the enzyme activities required for their own transposition. • However, it may also require host machinery (DNA pol or DNA gyrase). 12 Session #25-26 Transposons Discovery • Transposable elements were first identified in the form of spontaneous insertions in bacterial operons. • Such an insertion prevents transcription and/or translation of the gene in which it is inserted. • The first transposons that were discovered were simple and called insertion sequences (IS). • Each type is given the prefix IS, followed by a number that identifies the type. The original classes were IS1-4, more classes have been discovered since. • Insertion into a particular site described with a double colon: l::IS1 = An IS1 element inserted into phage lambda. 13 Session #25-26 Transposons Insertion Sequences (IS) • IS elements are normal constituents of bacterial chromosomes & plasmids. • A standard strain of E. coli contains <10 copies of the more common IS elements. • The IS elements code only for the proteins needed to sponsor its own transposition. • Each IS element is different in sequence, but there are common organizational features. 14 Session #25-26 Transposons Insertion Sequences (IS) • An IS element ends in short inverted terminal repeats which are not identical but closely related. • Inverted repeats recognized by TRANSPOSASE. • Ensure that the same sequence is encountered proceeding toward the element from any direction. • Inverted repeat recognition is common to transposition events sponsored by all transposons. • Cis-acting mutations of these ends prevent transposition. 15 Session #25-26 Transposons Insertion Sequences (IS) • When an IS element transposes, a sequence of host DNA at the site of insertion is duplicated: DIRECT REPEATS. • IS DNA is always flanked by very short direct repeats with the same direction. • Pre-transposition genomic sequences exhibit only one of these “repeats” (i.e.: ATGCA). • Post-transposition sequence will have this sequence duplicated and flanking the transposon sequence. 16 Session #25-26 Transposons Insertion Sequences (IS) • IS display a characteristic structure in which its ends possess inverted terminal repeats while the adjacent ends of the flanking host DNA possess short direct repeats. • This type of organization is taken to be diagnostic and suggest that the sequence originated in a transposition event. • IS elements insert at a variety of sites within host DNA, some show preference for particular hotspots 17 Session #25-26 Transposons Insertion Sequences (IS) • All IS elements (except IS1) contain a single long coding region for transposase starting after the inverted repeat at one end and terminating before or within the inverted repeat at the other end. • IS1 is more complex, it employs two separate reading frames. • Frequency of transposition varies amongst the different elements. 18 Session #25-26 Transposons Insertion Sequences (IS) • What would happen if an IS transposed near the original position... or if two IS sequences were separated by genomic DNA? • These transposons are called COMPOSITE TRANSPOSONS (Tn). 19 Session #25-26 Transposons Composite Transposons (Tn) • Code for more than proteins involved in transposition. • A central genomic “core” flanked by two IS. 20 Session #25-26 Transposons Composite Transposons (Tn) • Central region discovered initially as carrying drug markers or drug resistance traits. • IS modules ("arms") may have same or inverted orientations (most common). 21 Session #25-26 Transposons Composite Transposons (Tn) • Central region discovered initially as carrying drug markers or drug resistance traits. • IS modules ("arms") may have same or inverted orientations (most common). • In some cases the modules are identical – Tn9 (direct repeats of IS1) – Tn903 (inverted repeats of IS903). In other cases, the modules are only closely related (Tn10, Tn5). 22 Session #25-26 Transposons Composite Transposons (Tn) • A functional IS module can transpose either itself or the entire transposon. • Either identical module of a composite transposon can sponsor movement (IS10L or IS10R). • In transposons with different modules transposition might depend entirely or principally on one of the modules (Tn10 or Tn5). • What is responsible for transposing a composite transposon instead of just the individual module? 23 Session #25-26 Transposons Composite Transposons (Tn) 1.- IS vs Tn equally feasible & useful from a “selfish point of view”. 2.- Selective pressure. 24 Session #25-26 Transposons Composite Transposons (Tn) 1.- IS vs Tn equally feasible & useful from a “selfish point of view”. 2.- Selective pressure. – Two IS elements can transpose any sequence residing between them just as well as themselves. – Exemplified by transposons in bacteria where the two modules can be considered to flank either the tetR gene of the original Tn10 or the genomic sequence in the other part of the circle. 25 Session #25-26 Transposons Composite Transposons (Tn) 1.- IS vs Tn equally feasible & useful from a “selfish point of view”. 2.- Selective pressure. – Selection for the trait(s) carried in the central region. – An IS10 module mobilizes an order of magnitude more frequently than Tn10. – But Tn10 is held together by selection for tetR; so that under selective conditions, the relative frequency of intact Tn10 transposition is higher. 26 Session #25-26 Transposons Composite Transposons (Tn) – The insertion of a transposon into a new site consists of: • Making staggered breaks in the target DNA • Joining the transposon to the protruding singlestranded ends • Filling in the gaps. The stagger between the cuts determines the length of the direct repeats and reflects the geometry of the enzyme involved in cutting target DNA. 27 Session #25-26 Transposons Composite Transposons (Tn) – The insertion of a transposon into a new site consists of: • Making staggered breaks in the target DNA • Joining the transposon to the protruding singlestranded ends • Filling in the gaps. 28 Session #25-26 Transposons Composite Transposons (Tn) – The insertion of a transposon into a new site consists of: • Making staggered breaks in the target DNA • Joining the transposon to the protruding singlestranded ends • Filling in the gaps. The generation and filling of the staggered ends explain the direct repeats of target DNA at the site of insertion. The use of staggered ends is common to all transposons! 29 Session #25-26 Transposons Composite Transposons (Tn) – Three different types of mechanism by which a transposon moves: – Replicative Transposition – Common Non-replicative Transposition – Conservative Non-replicative Transposition 30 Session #25-26 Transposons Composite Transposons (Tn) – Three different types of mechanism by which a transposon moves: – Replicative Transposition • The element is duplicated during the reaction, so that the transposing entity is a copy of the original element. • One copy remains at the original site, while the other inserts at the new site. • Transposition is accompanied by an increase in the number of copies. 31 Session #25-26 Transposons Composite Transposons (Tn) – Three different types of mechanism by which a transposon moves: – Replicative Transposition • Involves two types of enzymatic activity: – Transposase that acts on the ends of the original transposon. – Resolvase that acts on the duplicated copies. 32 Session #25-26 Transposons Composite Transposons (Tn) – Three different types of mechanism by which a transposon moves: – Non-Replicative Transposition • Two types: – Common Non-replicative Transposition. – Conservative transposition (now known as Episomal transposition or simply Episome). 33 Session #25-26 Transposons Composite Transposons (Tn) – Three different types of mechanism by which a transposon moves: – Non-Replicative Transposition • The transposing element moves as a physical entity directly from one site to another – without copies – without change – Requires only a Transposase • Tn10 & Tn5 34 Session #25-26 Transposons Composite Transposons (Tn) – Three different types of mechanism by which a transposon moves: – Common Non-Replicative Transposition • Disregards double strand cleavage of genomic DNA from which it originated. • Relies on host repair mechanisms to repair double strand breaks. 35 Session #25-26 Transposons Composite Transposons (Tn) – Three different types of mechanism by which a transposon moves: – Conservative Non-replicative Transposition • the element is excised from the donor site and inserted into a target site by a series of events in which every nucleotide bond is conserved. • Transposon looking after the health of its host. • Mechanism of lambda Phage integration. 36 Session #25-26 Transposons Composite Transposons (Tn) – Three different types of mechanism by which a transposon moves: – Conservative Non-replicative Transposition • The elements that use this mechanism are large, and can mediate transfer of donor DNA from one bacterium to another. • Although originaly transposons, more appropriate name is Episomes. 37 Session #25-26 Transposons Composite Transposons (Tn) – Transposons may use only one or different types of pathway for transposition. – Basic reactions involved in all classes of transposition event: • The ends of the transposon are disconnected from the donor DNA by cleavage reactions that generate 3’-OH ends. • The exposed ends are joined to the target DNA by trans-esterification in which the 3’-OH end directly attacks the target DNA. 38 Session #25-26 Transposons Composite Transposons (Tn) • Reactions take place within nucleoprotein complex (enzymes and both ends of the transposon). • Target site is chosen by transposase (random vs. specificity) – for a consensus sequence, – for a structure, such as bent DNA, – for inactive regions of the chromosome. 39 Session #25-26 Transposons Composite Transposon DNA rearrangements – Transposons promote other types of DNA rearrangements. – Some of these events are consequences of the multiple copies generated (gene duplications). – Others represent alternative outcomes of the transposition mechanism. 40 Session #25-26 Transposons Composite Transposon DNA rearrangements – Rearrangements of host DNA may result when a transposon inserts a copy at a second site near its original location. – Host (or transposon) systems may undertake reciprocal recombination between the two copies of the transposon. – The consequences are determined by whether the repeats are the same or in inverted orientation. 41 Session #25-26 Transposons Composite Transposon DNA rearrangements – Recombination between direct repeats will delete the material between them. – The intervening region is excised as a circle of DNA (which is lost from the cell). – The chromosome retains a single copy of the direct repeat. – A recombination between the directly repeated IS1 modules of the composite transposon Tn9 would replace the transposon with a single IS1 module… This doesn’t normally happen! Why? 42 Session #25-26 Transposons Composite Transposon DNA rearrangements – Excision is not supported nor coded by transposons themselves. – Mechanism is not known. – Excision is RecA-independent. – Might occur by some cellular mechanism that generates spontaneous deletions between closely spaced repeated sequences. 43 Session #25-26 Transposons Composite Transposon DNA rearrangements – Reciprocal recombination between a pair of inverted repeats. – The region between the repeats becomes inverted. – The repeats themselves remain available to sponsor further inversions. A composite transposon whose modules are inverted is a stable component of the genome, although the direction of the central region with regard to the modules could be inverted by recombination. Direction influences transcription and translation! 44 Session #25-26 Transposons Composite Transposon DNA rearrangements – Reciprocal recombination between a pair of inverted repeats. – The region between the repeats becomes inverted. – The repeats themselves remain available to sponsor further inversions. A composite transposon whose modules are inverted is a stable component of the genome, although the direction of the central region with regard to the modules could be inverted by recombination. Direction influences transcription and translation! 45 Session #25-26 Transposons Transposition Intermediates – Many mobile DNA elements transpose from one chromosomal location to another by a fundamentally similar mechanism. • IS elements • Prokaryotic & eukaryotic transposons • Bacteriophage Mu. • Retroviral RNA integration. • The first stages of immunoglobulin recombination. 46 Session #25-26 Transposons Transposition Intermediates – Transposon is nicked at both ends. – Target sequence is nicked at both ends. 47 Session #25-26 Transposons Transposition Intermediates – The nicked ends are joined crosswise to generate a covalent connection between the transposon and the target. – The two ends of the transposon are brought together in this process. – FIGURE NOTE: for simplicity in following the cleavages, the synapsis stage is shown after cleavage, but actually occurs BEFORE CLEAVAGE. 48 Session #25-26 Transposons Transposition Intermediates – A more realistic image – The strand transfer complex in which the transposon is connected to the target site through one strand at each end. – The next step of the reaction differs and determines the type of transposition. 49 Session #25-26 Transposons Transposition Intermediates – A more realistic image – The strand transfer complex can be a target for replication (leading to replicative transposition). – Or the strand transfer complex can be a target for repair (non-replicative transposition). 50 Session #25-26 Transposons Transposition Intermediates – A more realistic image – The strand transfer complex can be a target for replication (leading to replicative transposition). – Or the strand transfer complex can be a target for repair (non-replicative transposition). 51 Session #25-26 Transposons Bacteriophage Mu (μ) – Upon infecting a host cell, Mu integrates into the genome by nonreplicative transposition. 52 Session #25-26 Transposons Bacteriophage Mu (μ) – Upon infecting a host cell, Mu integrates into the genome by nonreplicative transposition. – During the ensuing lytic cycle, the number of copies is amplified by replicative transposition. 53 Session #25-26 Transposons Bacteriophage Mu (μ) – Upon infecting a host cell, Mu integrates into the genome by nonreplicative transposition. – During the ensuing lytic cycle, the number of copies is amplified by replicative transposition. – Both types of transposition involve the same type of reaction between the transposon and its target, but the subsequent reactions are different. 54 Session #25-26 Transposons Bacteriophage Mu (μ) – The initial manipulations of the phage DNA are performed by MuA transposase. 55 Session #25-26 Transposons Bacteriophage Mu (μ) – Three MuA-binding sites with a 22 bp consensus are located at each end of Mu DNA. – A monomer of MuA can bind to each site. – MuA also binds to an internal site in the phage genome (enhancer). – Binding of MuA at both the left and right ends and the internal site forms a strand transfer complex. 56 Session #25-26 Transposons Bacteriophage Mu (μ) – Joining the Mu transposon DNA to a target site passes through 3 stages. – This involves only the two sites closest to each end of the transposon. – MuA has two sites for manipulating DNA: • The consensus-binding site binds to the 22 bp sequences that constitute the L1, L2, R1, and R2 sites. 57 Session #25-26 Transposons Bacteriophage Mu (μ) – Joining the Mu transposon DNA to a target site passes through 3 stages. – This involves only the two sites closest to each end of the transposon. – MuA has two sites for manipulating DNA: • The active site cleaves the Mu DNA strands at positions adjacent to the MuA-binding sites L1 and R1. 58 Session #25-26 Transposons Bacteriophage Mu (μ) – Joining the Mu transposon DNA to a target site passes through 3 stages. – This involves only the two sites closest to each end of the transposon. – MuA has two sites for manipulating DNA: • The active site cannot cleave the DNA sequence that is adjacent to the consensus sequence it can ONLY DO SO on a different stretch of DNA (Trans acting). 59 Session #25-26 Transposons Bacteriophage Mu (μ) – MuB + ATP + Target – A second protein, MuB, assists the reaction. – It has an influence on the choice of target sites. – Mu has a preference for transposing to a target site >10-15 kb away from the original insertion (target immunity). – Not a very good use of energy to move only a couple of base pairs down the road! 60 Session #25-26 Transposons Replicative Transpositon – Replicative transposition by definition generates a copy of a transposon at a new site. – The process (in circular replicons) proceed through the formation of a COINTEGRATE or hybrid structure. – The process is explained by the Shapiro Model. 61 Session #25-26 Transposons Replicative Transpositon – A plasmid with a transposon is the donor molecule. • Note transposon in black • Note flanking direct repeats – A bacterial chromosome is the recipient. • Note the integration site • No repeats here yet – The process starts with the formation of the strand transfer complex (crossover complex). 62 Session #25-26 Transposons Replicative Transpositon – NICKING takes place creating single strand cuts in both the transposon (3'end) and recipient (5'end). – Nicking depends on TRANSPOSASE. 63 Session #25-26 Transposons Replicative Transpositon – NICKING takes place creating single strand cuts in both the transposon (3'end) and recipient (5'end). – Nicking depends on TRANSPOSASE. – 3'-end of the transposon is linked to 5'end of recipient forming a Cointegrate hybrid. 64 Session #25-26 Transposons Replicative Transpositon – NICKING takes place creating single strand cuts in both the transposon (3'end) and recipient (5'end). – Nicking depends on TRANSPOSASE. – 3'-end of the transposon is linked to 5'end of recipient forming a Cointegrate hybrid. – Sequence gaps are filled in by host replicative machinery (repair DNA Pols) using exposed 3'-OH as primers. • Generates integration site direct repeats 65 Session #25-26 Transposons Replicative Transpositon – Replicated transposon exists at two sites now. – As both transposons are similar they promote homologous recombination between the two copies. – The recombination reaction is called resolution and is catalyzed by an enzyme called the resolvase. 66 Session #25-26 Transposons Replicative Transpositon – Replicated transposon exists at two sites now. – As both transposons are similar they promote homologous recombination between the two copies. – The recombination reaction is called resolution and is catalyzed by an enzyme called the resolvase. – Resolution releases two individual replicons, each of which has a copy of the transposon. • One in the original donor replicon. • A second one in the target replicon. 67 Session #25-26 Transposons Non-Replicative Transpositon – In non-replicative transposition the cointegrate structure is never stabilized. – Tranposon is completely excised before transfer. – Donor molecule remains nicked (and perhaps repaired by host machinery). – Staggered cuts in the recipient after transposition are filled in by repair machinery. 68 Session #25-26 Transposons Non-Replicative Transpositon – Non-replicative transposition can also use double-strand breaks to completely excise the transposon before ligation. – Host repair machinery is still needed to fill in sequence gaps. 69 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – Certain strains of D. melanogaster encounter difficulties in interbreeding. – When flies from two of these different strains are crossed, the progeny display "dysgenic traits". – Dysgeneic Traits are a series of defects including: • mutations, • chromosomal aberrations, • distorted segregation at meiosis, and • sterility. 70 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – Hybrid Dysgenesis only appears when F1 is the product of specific matings: P Strain Males + M Strain Females – Nothing happens when F1 is the product of other types of matings: M Strain Males + P Strain Females • P Strains = Paternally contributing • M Strains = Maternally contributing 71 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – Dysgenesis is principally a phenomenon of the germ cells. – In crosses involving the P-M system, the F1 hybrid flies have normal somatic tissues. – However, their gonads do not develop. – The morphological defect in gamete development dates from the stage at which rapid cell divisions commence in the germline. 72 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – Transposition responsible for hybrid dysgenesis occurs by a non-replicative "cut and paste" mechanism: – It contributes to hybrid dysgenesis in two ways: • Insertion of the transposed element at a new site may cause mutations. • And the break that is left at the donor site has a deleterious effect. 73 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – Why is there a direction? P♂ + M♀ = HD M♂ + P♀ = Wt 74 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – Why is there a direction? P♂ + M♀ = HD M♂ + P♀ = Wt – P♂♀ have 30-50 P elements (transposons) – M♂♀ do not. – P elements code for P factors (transposase) in both P♂♀. 75 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – P elements code for P factors in both P♂♀. – P element has 4 ORFs (exons). – Somatic tissues only transcribe 3 exons. – Germline cells transcribe 4 exons. – Short polypeptide is 66KD and Represor. – Long polypeptide is 87 kD and Transposase 76 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – Presence of Represor in cytoplasm inhibits activity of Transposase in nucleous. – This explains why P♂♀ flies are normal. – P♂♀ flies can interbreed without problems because the function of cytoplasmic and nuclear transposon expression is balanced. – But P♂♀ flies encounter breeding difficulty with M♂♀ flies... in certain ways. 77 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – First, sperm differs from eggs in the amount of cytoplasm (cytotope). – Second, P♀ fly eggs possess both the translocon and enormous amounts of cytoplasm repressor > transposase – P♀ fly + M♂ fly = Progeny with P Cytotope Normal repressor > transposase 78 Session #25-26 Transposons Transposons & Hybrid Dysgenesis – P♂ fly sperm on the other hand possess little if any cytoplasm & the same translocon repressor < transposase – Yet P♂ fly somatic cells have maternal P cytotpe and are therby normal. – P♂ fly + M♀ fly = Progeny with M Cytotope Hybrid Dysgenesis repressor < transposase 79 Session #25-26 Transposons Transposons & Humans – The most common form of transposon in humans is the Alu sequence. – Classified as short interspersed nuclear elements (SINEs) amongst the class of repetitive DNA elements. – Named for its susceptibility to Alu restriction endonuclease. – Approximately 300 bases long and can be found between 300,000 & a million times in the human genome. – Alu sequences of different kinds occur in large numbers in primate genomes. 80 Session #25-26 Transposons Transposons & Humans What percentage of human genome consists of Alu sequence type transposons? 81 Session #25-26 Transposons Transposons & Humans What percentage of human genome consists of Alu sequence type transposons? 10.7% 82 Session #25-26 Transposons Transposons & Humans – NCBI Map Viewer BLAST for Alu. 83 Session #25-26 Transposons Transposons & Humans 84 Session #25-26 Transposons Transposons & Humans Go to Alu Jump Flash animation 85 Session #25-26 Transposons Transposons & Humans – The common SINE (a type of retroposon) family Alu probably originated from a duplication of a 7SL RNA gene, member of the SRP. 86 Session #25-26 Transposons Transposons & Humans – Transposons are mutagens. – They can damage the genome of their host cell in different ways: • It inserts itself into a functional gene commonly disabling it. • Replicative transposition leaves gaps. • Multiple copies of the same sequence (Alu repeats) can lead to unequal crossovers. 87 Session #25-26 Transposons Transposon related Hemophilia – Group of hereditary genetic disorders that impair the body's ability to control blood clotting or coagulation. – Hemophilia A, clotting factor VIII is absent, in B factor IX is deficient. – Type A occurs in about :5,000-10,000 male births, B occurs at about 1 in about 20,000-34,000. – Sex-linked, X chromosome disorder manifested almost entirely in males but inherited from the mother. – Major complications include hemarthrosis, hemorrhage, gastrointestinal bleeding, and menorrhagia. 88 Session #25-26 Transposons Transposons & Disease – Diseases that are often caused by transposons include: • Hemophilia A and B • Severe combined immunodeficiency • Porphyria • Predisposition to cancer • Duchenne muscular dystrophy. 89 Session #25-26 Transposons Conclusion Many changes in the genetic material are known to be deleterious. In general terms, the more dramatic the change the more catastrophic the outcome. Rearrangements of DNA have traditionally been viewed as unnatural and dangerous. Discovery of transposons has given us a far more dynamic perspective on genome evolution within which segments of DNA naturally move about. 90 Session #25-26 Transposons Conclusion The difference in genome size observed betwen the different lifeforms are thought to be the result (in large part) of transposon activity. 91 Session #25-26 Transposons Conclusion •Additionally, transposon-dependent genome rearrangements help explain a mechanism of speciation, which furthers natural diversity (as clearly demonstrated for D. melanogaster). • Flies isolated more than 30 years ago almost always M strains. • Flies isolated in the last 10 years mostly P strains. –Mechanism for reproductive isolation plays an important role in the evolution of novel species. 92 Session #25-26 Transposons Barbara McClintock (1983) One of the world's most distinguished cytogeneticists. First to document many fundamental genetic ideas under the microscope, including genetic recombination by chromosomal crossover during meiosis. Produced the first genetic map for maize. Demonstrated the role of the telomere & centromere. Discovered Dissociator & Activator Transposition (~Hybrid dysgenesis in Maize) Barbara McClintock (Jun 16, 1902 – Sept 2, 1992). 1983 Nobel laureate in Physiology & Medicine 93