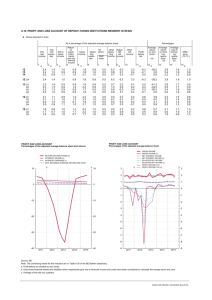
Ajay J. Kirtane and Gregg W. Stone, 2008 Mean f/u 1.5 yrs MI
advertisement

Comprehensive Meta-Analysis of DES vs. BMS Randomized Trials and Registries Ajay J. Kirtane, M.D., S.M. Gregg W. Stone, M.D. Columbia University Medical Center The Cardiovascular Research Foundation Conflict of Interest Disclosure • Ajay J. Kirtane Past honorarium from Boston Scientific Corporation (modest) Consultant/Speaker: Medtronic Vascular, Abbott Vascular (modest) • Gregg W. Stone Research grants from Boston Scientific and Abbott Vascular Background: DES vs. BMS RCTs • In most individual RCTs, DES have reduced the rates of TLR and TVR compared to BMS, with no significant differences in death or MI • However, individual RCTs are underpowered to assess low frequency endpoints • RCTs, particularly the pivotal RCTs leading to regulatory approval, have been criticized for not reflecting “real-world” DES use • RCT outcomes may vary based upon differences in enrollment criteria (e.g. “on-label” vs. “offlabel”), the amount of routine angiographic FU, and with the duration of clinical FU Background: DES vs. BMS Registries • In order to address issues of both sample size as well as generalizability to the “real-world”, numerous observational and registry comparisons of DES vs. BMS have been undertaken • The outcomes from these studies have varied • While more generalizable than RCTs, the DES vs. BMS registries are heterogeneous, with differences in design and analysis methodology (e.g. adjusted vs. unadjusted, type of adjustment) • Registry outcomes may also vary based upon the types of patients enrolled (e.g. all comers vs. just ACS or high risk), and the duration of clinical FU Persistent Questions: DES vs. BMS • While some of the alarm generated after ESC 2006 has been mitigated by analyses of patient-level data from the “on-label” RCTs*, there remains concern regarding DES outcomes in “off-label” patients and lesions, and with uncontrolled use Are DES safe in higher risk off-label pts and in the unregulated environment of real-world use? Are the benefits of DES in reducing TVR as robust in the real-world as in the RCTs, given the impact of routine angio FU and the oculostenotic reflex in many RCTs? *Stone et al, Kastrati et al, Spaulding et al, Mauri et al N Engl J Med 2007; 356(10). Methods: Goals and Objectives (1) • We therefore sought to perform a systematic review and meta-analysis of DES vs. BMS studies To derive summary estimates of all-cause mortality, MI, and TVR in studies with ≥1 year of follow-up To specifically assess differences between RCT and registry safety and effiacy with regard to these endpoints Methods: Goals and Objectives (2) • Randomized Trials To assess differences between RCTs according to “on-label” vs. “off-label” use, duration of FU, and baseline risk • Registries / Observational Analyses To assess differences in the estimates derived from registries using unadjusted and adjusted analyses (and according to the types of adjustment) To assess differences between registries according to duration of FU, and baseline risk • To assess differences in effect size estimates between the RCTs and registries for each endpoint Methods: Inclusion Criteria • English language RCTs or registries which reported a direct comparison of DES (commercialized formulations of SES and PES only) vs. BMS. • Criteria for each study: ≥100 patients total Mortality reported (± MI and/or TVR) ≥1 year of cumulative follow-up reported, with the outcome assessed at the same time point in both comparator arms Methods: Exclusion Criteria • “DES era” vs. pure “BMS era” studies in which the DES era group did not exclude BMS pts Excepting intent-to-treat RCTs • Study used a control group from another study already in the meta-analysis • Study was itself a meta-analysis (although data abstracted from individual studies in a published meta-analysis could be used) Methods: Search Strategy (1) • 2 PUBMED searches Focused: (eluting stent OR DES OR drug-eluting stent) AND (bare OR uncoated OR standard OR BMS) AND (("2002"[PDat] : "2008"[PDat]) AND (Humans[Mesh]) AND (English[lang])) AND (coronary) NOT (cost-effectiveness) NOT review[pt] NOT case reports[pt] NOT editorial[pt] NOT comment[pt] Broad: stent AND bare AND (eluting OR sirolimus OR paclitaxel) • Cochrane database • Eurointervention journal Methods: Search Strategy (2) • Abstracts/presentations from 2007 meetings: ACC SCAI/I2 Summit ESC TCT AHA • Data requested from study PI’s for large registries For most updated data Where not publicly available or to clarify methodology Methods: Analysis • Pre-specified separate analysis of RCTs and Registries performed given clinical heterogeneity • RCTs: Direct randomization to DES vs. BMS • Registries: Non-rand comparison of DES vs. BMS (including non-rand comparisons within a RCT) • All analyses cumulative • No landmarks • Single time point estimate for each study assuming constant hazard of DES vs. BMS through study period (hence use of HR or RR as the estimate) • Higher quality estimate picked for primary analyses (adjusted > unadjusted) Methods: Statistical Analysis • All analyses were performed at The Cardiovascular Research Foundation/Columbia University • Models (both reported): • Fixed effects (Inverse-Variance weighted) • Random effects (DerSimonian and Laird)* • Fixed effects model was considered the primary model if significant heterogeneity was not present; otherwise random effects was considered primary • Formal heterogeneity testing was performed using the I2 statistic; heterogeneity was defined as I2 ≥ 25% *Weights displayed in figures are based upon the primary model used Selected Excluded Studies • GRACE Non-landmark data not available (PI contacted as well) Unequal follow-up in comparator arms (data not presented at fixed timepoint) • RRISC Less than 100 patients • Medicare Data Comparison of pre-DES era with post-DES rather than DES vs. BMS All-Cause Mortality: All RCTs 8,867 patients, 21 trials % Weight ES (95% CI) CI) Weight (I-V) (%) Estimate (95% Study ID SCORPIUS SESAMI Typhoon Passion BASKET (SES only) STRATEGY SES-SMART Seville HAAMU-STENT MISSION! PRISON II Pache et al Ortolani et al DIABETES RAVEL SIRIUS C-SIRIUS E-SIRIUS TAXUS II TAXUS IV TAXUS V I-V Overall Effects (I-squared = 0.0%, p = 0.918) Random D+L Overall *Fixed Effects (I2=0.0%) 1.28 (0.35, 4.61) 0.43 (0.11, 1.63) 1.01 (0.38, 2.65) 0.70 (0.36, 1.36) 0.82 (0.37, 1.84) 0.84 (0.36, 1.96) 0.21 (0.02, 1.71) 1.35 (0.23, 7.78) 2.00 (0.63, 6.38) 0.48 (0.09, 2.59) 0.50 (0.09, 2.67) 1.40 (0.45, 4.35) 2.00 (0.19, 21.38) 1.44 (0.48, 4.33) 1.75 (0.73, 4.16) 1.02 (0.67, 1.54) 0.68 (0.11, 4.04) 1.08 (0.25, 2.24) 1.61 (0.57, 4.53) 0.89 (0.63, 1.25) 0.97 (0.57, 1.65) 0.97 (0.81, 1.15) 0.97 (0.81,1.15) 0.97 (0.81, 1.15) 0.97 (0.81,1.15), p=0.72 Favors DES .1 1.86 1.70 3.27 6.99 4.80 4.30 0.62 1.00 2.30 1.09 1.07 2.40 0.55 2.55 4.08 17.82 0.95 2.57 2.87 26.29 10.92 100.00 Favors BMS 1 10 Ajay J. Kirtane and Gregg W. Stone, 2008 Mean f/u 2.9 yrs All-Cause Mortality: RCTs (On-Label) % 4,818 patients, 10 trials Study ID Weight ES (95% CI) (I-V) Estimate (95% CI) Weight (%) SCORPIUS 1.28 (0.35, 4.61) 2.84 Pache et al 1.40 (0.45, 4.35) 3.67 Ortolani et al 2.00 (0.19, 21.38) 0.85 RAVEL 1.75 (0.73, 4.16) 6.23 SIRIUS 1.02 (0.67, 1.54) 27.25 C-SIRIUS 0.68 (0.11, 4.04) 1.45 E-SIRIUS 1.08 (0.25, 2.24) 3.93 TAXUS II 1.61 (0.57, 4.53) 4.39 TAXUS IV 0.89 (0.63, 1.25) 40.20 TAXUS V - Simple 1.09 (0.53, 2.22) 9.20 I-V Overall (I-squared = 0.0%, p = 0.927) Random Effects 1.05 (0.84, 1.30) (0.84,1.30) 100.00 *Fixed Effects (I2=0.0%) D+L Overall (0.84,1.30), 1.05 (0.84, 1.30) p=0.69 Favors DES .1 Favors BMS 1 Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 4.0 yrs All-Cause Mortality: RCT’s (Off-Label) % 4,049 patients, 12 trials Study ID Weight ES (95% CI) (I-V) Estimate (95% CI) Weight (%) SESAMI 0.43 (0.11, 1.63) 4.90 Typhoon 1.01 (0.38, 2.65) 9.44 Passion 0.70 (0.36, 1.36) 20.16 BASKET (SES only) 0.82 (0.37, 1.84) 13.84 STRATEGY 0.84 (0.36, 1.96) 12.40 SES-SMART 0.21 (0.02, 1.71) 1.80 Seville 1.35 (0.23, 7.78) 2.87 HAAMU-STENT 2.00 (0.63, 6.38) 6.64 MISSION! 0.48 (0.09, 2.59) 3.16 PRISON II 0.50 (0.09, 2.67) 3.10 DIABETES 1.44 (0.48, 4.33) 7.36 TAXUS V - complex 0.84 (0.38, 1.84) 14.32 Random I-V OverallEffects (I-squared = 0.0%, p = 0.798) *Fixed Effects (I2=0.0%) D+L Overall 0.84 (0.62,1.13) (0.62, 1.13) 100.00 0.84 (0.62,1.13), (0.62, 1.13) p=0.24 Favors DES .1 Favors BMS 1 Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 1.5 yrs All-Cause Mortality: All Registries 161,232 patients, 28 registries Study ID ES (95% CI) Estimate (95% CI) NHLBI (off label, adjusted) NHLBI (on label, adjusted) Germany Metabolic Syndrome Ontario (matched) Mayo FFR Substudy Italian Diabetic Multivessel (adjusted) McMaster STEMI (adjusted) Rotterdam Off-Label Washington Hosp Center (matched) Asan Korea (adjusted) SCAAR (adjusted) Wake Forest (adjusted) Western Denmark (adjusted) NY State (adjusted, unmatched) MIDAS (adjusted) Massachusetts (matched) STENT (adjusted) Liverpool (matched) GHOST (adjusted) DEScover (unadjusted) Cedars Acute MI REAL (adjusted) Melbourne Multicenter SVG (adjusted) ACUITY (from RCT) RESTEM ARTS II (from RCT) ERACI III (from RCT) D+L Overall (I-squared =270.1%, p = 0.000) *Random Effects (I =70.1%) I-V Overall % Weight (D+L) Weight (%) 0.94 (0.64, 1.38) 3.40 1.47 (0.87, 2.48) 2.31 1.47 (0.65, 3.35) 1.15 0.71 (0.59, 0.84) 5.98 1.00 (0.21, 4.75) 0.36 1.22 (0.36, 4.10) 0.57 0.17 (0.03, 0.97) 0.29 0.98 (0.85, 1.13) 6.44 1.16 (0.78, 1.75) 3.21 0.60 (0.46, 0.79) 4.70 1.03 (0.94, 1.14) 6.98 0.72 (0.55, 0.95) 4.66 1.00 (0.86, 1.17) 6.29 0.84 (0.72, 0.97) 6.35 0.66 (0.59, 0.74) 6.80 0.79 (0.71, 0.89) 6.80 0.69 (0.55, 0.87) 5.25 0.45 (0.24, 0.84) 1.78 0.55 (0.36, 0.83) 3.09 0.53 (0.35, 0.80) 3.13 0.82 (0.37, 1.83) 1.20 0.83 (0.70, 0.98) 6.10 0.67 (0.23, 1.94) 0.73 1.33 (0.47, 3.76) 0.76 0.63 (0.49, 0.82) 4.87 0.73 (0.51, 1.05) 3.63 0.74 (0.41, 1.35) 1.92 1.18 (0.54, 2.58) 1.25 0.80 (0.72, 0.88) 100.00 0.80 (0.72,0.88), p<0.001 0.83 (0.79, 0.86) Fixed Effects 0.83 (0.79,0.86) NOTE: Weights are from random effects analysis .1 Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 2.5 yrs All-Cause Mortality: Unadjusted Registries 122,989 patients, 22 registries % Weight Study ID ES (95% CI) (D+L) (%) Estimate (95% CI) Weight NHLBI (off label, unadjusted) 0.58 (0.42, 0.80) 4.86 NHLBI (on label, unadjusted) 1.04 (0.68, 1.58) 3.70 Germany Metabolic Syndrome 1.47 (0.65, 3.35) 1.43 Mayo FFR Substudy 1.00 (0.21, 4.75) 0.45 McMaster STEMI (unadjusted) 0.27 (0.06, 1.24) 0.47 Rotterdam Off-Label 0.98 (0.85, 1.13) 7.53 Asan Korea (unadjusted) 0.71 (0.58, 0.87) 6.63 Wake Forest (unadjusted) 0.71 (0.54, 0.92) 5.65 Western Denmark (unadjusted) 0.81 (0.70, 0.94) 7.46 NY State (unadjusted, unmatched) 0.79 (0.69, 0.92) 7.51 MIDAS (unadjusted) 0.67 (0.60, 0.75) 7.94 Massachusetts (unadjusted) 0.54 (0.49, 0.59) 8.16 STENT (unadjusted) 0.52 (0.43, 0.62) 6.93 Liverpool (unadjusted) 0.49 (0.30, 0.80) 3.08 DEScover (unadjusted) 0.53 (0.35, 0.80) 3.79 Cedars Acute MI 0.82 (0.37, 1.83) 1.49 REAL (unadjusted) 0.74 (0.66, 0.82) 7.98 Melbourne 0.67 (0.23, 1.94) 0.90 ACUITY (from RCT) 0.63 (0.49, 0.82) 5.78 RESTEM 0.73 (0.51, 1.05) 4.37 ARTS II (from RCT) 0.74 (0.41, 1.35) 2.35 ERACI III (from RCT) 1.18 (0.54, 2.58) 1.54 2=75.3%) D+L Overall (I-squared p = 0.000) *Random Effects =(I75.3%, 0.70 (0.63, 0.78) 100.00 0.70 (0.63,0.78), p<0.001 I-V Overall Fixed Effects 0.69 (0.66, 0.72) 0.69 (0.66,0.72) NOTE: Weights are from random effects analysis .1 Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 2.1 yrs All-Cause Mortality: Adjusted Registries % 134,534 patients, 18 registries Study ID Weight ES (95% CI) (D+L) Estimate (95% CI) Weight (%) NHLBI (off label, adjusted) 0.94 (0.64, 1.38) 4.59 NHLBI (on label, adjusted) 1.47 (0.87, 2.48) 3.15 Ontario (matched) 0.71 (0.59, 0.84) 7.90 Italian Diabetic Multivessel (adjusted) 1.22 (0.36, 4.10) 0.79 McMaster STEMI (adjusted) 0.17 (0.03, 0.97) 0.40 Washington Hosp Center (matched) 1.16 (0.78, 1.75) 4.34 Asan Korea (adjusted) 0.60 (0.46, 0.79) 6.27 SCAAR (adjusted) 1.03 (0.94, 1.14) 9.14 Wake Forest (adjusted) 0.72 (0.55, 0.95) 6.22 Western Denmark (adjusted) 1.00 (0.86, 1.17) 8.29 NY State (adjusted, unmatched) 0.84 (0.72, 0.97) 8.37 MIDAS (adjusted) 0.66 (0.59, 0.74) 8.92 Massachusetts (matched) 0.79 (0.71, 0.89) 8.92 STENT (adjusted) 0.69 (0.55, 0.87) 6.98 Liverpool (matched) 0.45 (0.24, 0.84) 2.44 GHOST (adjusted) 0.55 (0.36, 0.83) 4.18 REAL (adjusted) 0.83 (0.70, 0.98) 8.05 Multicenter SVG (adjusted) 1.33 (0.47, 3.76) 1.05 D+L OverallEffects (I-squared = 76.6%, *Random (I2=76.6%) p = 0.000) 0.80 (0.72, 0.90) p<0.001 100.00 0.80 (0.72,0.90), 0.82 (0.79,0.86) 0.82 (0.79, 0.86) Fixed Effects I-V Overall NOTE: Weights are from random effects analysis .1 Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 2.7 yrs All-Cause Mortality: Registries Begg’s Funnel Plot p=0.92 log(Hazard Ratio) 2 1 0 -1 -2 0 .5 Standard Error of log(Hazard Ratio) Ajay J. Kirtane and Gregg W. Stone, 2008 1 MI: All RCTs % Weight ES (95% CI) (I-V) Estimate (95% CI) Weight (%) 8,850 patients, 20 trials StudyIDID Study SCORPIUS SCORPIUS SESAMI SESAMI Typhoon Typhoon Passion Passion BASKET(All) (All) BASKET STRATEGY STRATEGY SES-SMART SES-SMART HAAMU-STENT HAAMU-STENT MISSION! MISSION! PRISONII II PRISON Ortolanietetalal Ortolani DIABETES DIABETES SCANDSTENT SCANDSTENT RAVEL RAVEL SIRIUS SIRIUS C-SIRIUS C-SIRIUS E-SIRIUS E-SIRIUS TAXUSII II TAXUS TAXUSIVIV TAXUS TAXUSVV TAXUS I-V OverallEffects D+L (I-squared = 3.0%, p = 0.420) Random D+L Overall I-V Overall *Fixed Effects (I2=3.0%) .1.1 Favors DES 0.82 (0.23, 2.95) 2.02 1.00 (0.20, 4.88) 1.29 0.80 (0.22, 2.97) 1.94 0.83 (0.26, 2.69) 2.40 1.15 (0.64, 2.08) 9.45 0.82 (0.31, 2.40) 3.13 0.16 (0.04, 0.67) 1.65 0.25 (0.03, 2.19) 0.71 0.62 (0.28, 1.39) 5.11 0.83 (0.26, 2.64) 2.44 1.50 (0.26, 8.61) 1.07 0.60 (0.20, 1.50) 3.23 0.33 (0.09, 1.18) 1.98 1.24 (0.49, 3.14) 3.80 0.96 (0.59, 1.55) 14.07 0.59 (0.14, 2.47) 1.59 1.94 (0.93, 4.02) 6.13 0.63 (0.23, 1.72) 3.24 0.99 (0.66, 1.48) 20.13 1.27 (0.79, 2.04) 14.59 0.94 (0.79, 1.13) 100.00 0.94 (0.78,1.13) 0.94 (0.78, 1.13) 0.94 (0.79,1.13), p=0.54 11 Favors BMS 10 10 Ajay J. Kirtane and Gregg W. Stone, 2008 Mean f/u 2.9 yrs MI: RCTs (On Label) % 4,318 patients, 9 trials Study ID Weight ES (95% CI) (I-V) Estimate (95% CI) Weight (%) SCORPIUS 0.82 (0.23, 2.95) 3.33 Ortolani et al 1.50 (0.26, 8.61) 1.77 RAVEL 1.24 (0.49, 3.14) 6.29 SIRIUS 0.96 (0.59, 1.55) 23.26 C-SIRIUS 0.59 (0.14, 2.47) 2.63 E-SIRIUS 1.94 (0.93, 4.02) 10.13 TAXUS II 0.63 (0.23, 1.72) 5.36 TAXUS IV 0.99 (0.66, 1.48) 33.28 TAXUS V - Simple 0.98 (0.52, 1.81) 13.95 Random I-V OverallEffects (I-squared = 0.0%, p = 0.761) *Fixed Effects (I2=0.0%) D+L Overall 1.03(0.81,1.30) (0.81, 1.30) 1.03 100.00 1.03 1.03(0.81,1.30), (0.81, 1.30) p=0.82 Favors DES .1 Favors BMS 1 Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 4.4 yrs MI: RCT’s (Off Label) Study Study IDID %% 4,532 patients, 12 trialsESES(95% (95% CI) CI) Weight Weight (I-V) (I-V) Estimate (95% CI) Weight (%) SESAMI SESAMI 1.00 1.00 (0.20, (0.20, 4.88) 4.88)3.30 3.30 Typhoon Typhoon 0.80 0.80 (0.22, (0.22, 2.97) 2.97)4.97 4.97 Passion Passion 0.83 0.83 (0.26, (0.26, 2.69) 2.69)6.16 6.16 BASKET BASKET (All) (All) 1.15 1.15 (0.64, (0.64, 2.08) 2.08)24.22 24.22 STRATEGY STRATEGY 0.82 0.82 (0.31, (0.31, 2.40) 2.40)8.03 8.03 SES-SMART SES-SMART 0.16 0.16 (0.04, (0.04, 0.67) 0.67)4.24 4.24 HAAMU-STENT HAAMU-STENT 0.25 0.25 (0.03, (0.03, 2.19) 2.19)1.83 1.83 MISSION! MISSION! 0.62 0.62 (0.28, (0.28, 1.39) 1.39)13.11 13.11 PRISON PRISON II II 0.83 0.83 (0.26, (0.26, 2.64) 2.64)6.26 6.26 DIABETES DIABETES 0.60 0.60 (0.20, (0.20, 1.50) 1.50)8.29 8.29 SCANDSTENT SCANDSTENT 0.33 0.33 (0.09, (0.09, 1.18) 1.18)5.08 5.08 TAXUS TAXUS VV - complex - complex 1.84 1.84 (0.86, (0.86, 3.94) 3.94)14.52 14.52 I-VI-V Overall Overall (I-squared (I-squared= = 25.5%, 25.5%, p p= = 0.194) 0.194) Random Effects 0.83 0.83 (0.62, (0.62, 1.10) 1.10)100.00 100.00 0.77 (0.54,1.10) *Fixed Effects (I2=25.5%) D+L D+L Overall Overall 0.83 (0.62,1.10), p=0.19 0.77 0.77 (0.54, (0.54, 1.10) 1.10) Favors DES .1.1 .1 Favors BMS 111 Ajay J. Kirtane and Gregg W. Stone, 2008 10 10 10 Mean f/u 1.5 yrs MI: All Registries 129,955 patients, 24 registries Study ID % Weight ES (95% CI) (D+L) NHLBI (off label, adjusted) 0.71 (0.50, 1.00) 5.01 NHLBI (on label, adjusted) 0.71 (0.47, 1.05) 4.21 Germany Metabolic Syndrome 0.23 (0.07, 0.78) 0.70 Ontario (matched) 1.10 (0.91, 1.32) 8.26 Mayo FFR Substudy 0.67 (0.12, 3.84) 0.35 Brazil Large Vessels 1.50 (0.25, 8.90) 0.33 Italian Diabetic Multivessel (adjusted) 1.02 (0.46, 2.25) 1.50 McMaster STEMI (adjusted) 0.28 (0.04, 1.71) 0.30 Washington Hosp Center (matched) 0.51 (0.29, 0.88) 2.69 Asan Korea (adjusted) 0.66 (0.42, 1.05) 3.55 SCAAR (adjusted) 1.01 (0.91, 1.11) 10.17 Wake Forest (adjusted) 0.84 (0.60, 1.18) 5.15 Western Denmark (adjusted) 1.29 (1.06, 1.57) 8.02 Massachusetts (matched) 0.92 (0.83, 1.02) 10.10 STENT (adjusted) 0.69 (0.52, 0.92) 6.10 GHOST (adjusted) 1.12 (0.74, 1.70) 4.03 DEScover (unadjusted) 0.69 (0.40, 1.18) 2.80 Cedars Acute MI 0.25 (0.06, 1.16) 0.48 REAL (adjusted) 0.92 (0.76, 1.11) 8.18 Melbourne 1.00 (0.39, 2.58) 1.10 ACUITY (from RCT) 1.07 (0.91, 1.25) 8.90 RESTEM 0.80 (0.52, 1.23) 3.86 ARTS II (from RCT) 0.53 (0.32, 0.88) 3.09 ERACI III (from RCT) 2.30 (0.91, 5.96) 1.11 Estimate (95% CI) Weight (%) D+L Overall (I-squared p = 0.000) *Random Effects =(I257.9%, =57.9%) 0.89 (0.80,0.98), (0.80, 0.98) 100.00 0.89 p=0.023 I-V Overall Fixed Effects 0.96 (0.91,1.01) (0.91, 1.01) 0.96 *MI is NOTE: QWMI in Washington Hospital Center, RESTEM Weights are from random effects analysis .1 Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 2.5 yrs MI: Unadjusted Registries % 88,221 patients, 18 registries Weight ES (95% CI) Study ID (D+L) Estimate (95% CI) Weight (%) NHLBI (off label, unadjusted) 0.75 (0.55, 1.01) 8.11 NHLBI (on label, unadjusted) 0.80 (0.56, 1.16) 7.22 Germany Metabolic Syndrome 0.23 (0.07, 0.78) 1.56 Mayo FFR Substudy 0.67 (0.12, 3.84) 0.82 Brazil Large Vessels 1.50 (0.25, 8.90) 0.77 McMaster STEMI (unadjusted) 0.29 (0.04, 1.86) 0.67 Asan Korea (unadjusted) 0.78 (0.55, 1.11) 7.41 Western Denmark (unadjusted) 1.19 (0.99, 1.43) 9.89 Massachusetts (unadjusted) 0.75 (0.69, 0.82) 10.97 STENT (unadjusted) 0.62 (0.49, 0.78) 9.19 DEScover (unadjusted) 0.69 (0.40, 1.18) 5.02 Cedars Acute MI 0.25 (0.06, 1.16) 1.09 REAL (unadjusted) 1.15 (1.02, 1.30) 10.65 Melbourne 1.00 (0.39, 2.58) 2.34 ACUITY (from RCT) 1.07 (0.91, 1.25) 10.22 RESTEM 0.80 (0.52, 1.23) 6.31 ARTS II (from RCT) 0.53 (0.32, 0.88) 5.40 ERACI III (from RCT) 2.30 (0.91, 5.96) 2.36 p = 0.000) (I-squared(I=277.8%, D+L Overall Effects *Random =77.8%) I-V Overall Fixed Effects 100.00 0.97) 0.83 (0.70, 0.83 (0.70,0.97), p=0.023 0.93) 0.88 (0.83, 0.88 (0.83,0.93) *MI is QWMI in Washington RESTEM effects analysis random Center, are from Hospital Weights NOTE: .1 Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 2.0 yrs MI: Adjusted Registries Study ID % 107,294 patients, 14 registries ES (95% CI) Weight (D+L) Estimate (95% CI) Weight (%) NHLBI (off label, adjusted) 0.71 (0.50, 1.00) 6.18 NHLBI (on label, adjusted) 0.71 (0.47, 1.05) 5.11 Ontario (matched) 1.10 (0.91, 1.32) 10.94 Italian Diabetic Multivessel (adjusted) 1.02 (0.46, 2.25) 1.72 McMaster STEMI (adjusted) 0.28 (0.04, 1.71) 0.34 Washington Hosp Center (matched) 0.51 (0.29, 0.88) 3.16 Asan Korea (adjusted) 0.66 (0.42, 1.05) 4.25 SCAAR (adjusted) 1.01 (0.91, 1.11) 14.06 Wake Forest (adjusted) 0.84 (0.60, 1.18) 6.37 Western Denmark (adjusted) 1.29 (1.06, 1.57) 10.56 Massachusetts (matched) 0.92 (0.83, 1.02) 13.94 STENT (adjusted) 0.69 (0.52, 0.92) 7.70 GHOST (adjusted) 1.12 (0.74, 1.70) 4.87 REAL (adjusted) 0.92 (0.76, 1.11) 10.81 D+L OverallEffects (I-squared = 60.8%, p = 0.002) *Random (I2=60.8%) 0.91 (0.81, 1.01) p=0.083 100.00 0.91 (0.81,1.01), Fixed Effects I-V Overall 0.96 (0.91, 1.01) 0.96 (0.91,1.01) *MI is QWMI in Washington Hospital Centereffects NOTE: Weights are from random .1 analysis Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 2.8 yrs Study ID TVR: All RCTs 7,291 patients, 16 trials % Weight ES (95% CI) Estimate (95% CI) (D+L) Weight (%) SESAMI 0.36 (0.17, 0.79) 4.36 Typhoon 0.42 (0.25, 0.69) 7.20 STRATEGY 0.34 (0.16, 0.77) 4.22 HAAMU-STENT 0.33 (0.09, 1.19) 1.91 MISSION! 0.38 (0.17, 0.85) 4.08 PRISON II 0.37 (0.19, 0.69) 5.49 Pache et al 0.38 (0.23, 0.64) 7.14 Ortolani et al 0.58 (0.25, 1.36) 3.78 SCANDSTENT 0.17 (0.09, 0.33) 5.44 RAVEL 0.51 (0.25, 1.04) 4.83 SIRIUS 0.48 (0.37, 0.62) 11.51 C-SIRIUS 0.30 (0.10, 0.93) 2.45 E-SIRIUS 0.35 (0.21, 0.56) 7.45 TAXUS II 0.61 (0.35, 1.08) 6.44 TAXUS IV 0.57 (0.45, 0.72) 11.94 TAXUS V 0.77 (0.60, 0.98) 11.75 D+L OverallEffects (I-squared = 53.2%, p = 0.006) *Random (I2=53.2%) Fixed Effects I-V Overall 0.45 (0.37, 0.54) p<0.001 100.00 0.45 (0.37,0.54), 0.51 (0.45, 0.57) 0.51 (0.45,0.57) NOTE: Weights are from random effects analysis .1 Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 3.2 yrs TVR: RCTs *Hazard Ratio .4 .6 .8 Meta-Regression on Percent Angiographic F/U .2 p=0.73 20 40 60 80 perangfu Percentage of Patients with Angiographic F/U 100 tvr_hr Fitted *log(HR) regressed on percentage of pts with angiographic f/u; figurevalues displayed on exponentiated scale Ajay J. Kirtane and Gregg W. Stone, 2008 Study ID TVR: All Registries 73,819 patients, 17 registries % Weight ES (95% CI) Estimate (95% CI) (D+L) Weight (%) Ontario (matched) 0.69 (0.60, 0.80) 9.88 Mayo FFR Substudy 0.18 (0.04, 0.78) 0.68 Brazil Large Vessels 0.43 (0.17, 1.10) 1.57 McMaster STEMI (adjusted) 0.32 (0.05, 1.92) 0.46 Washington Hosp Center (matched) 0.65 (0.49, 0.85) 7.35 Asan Korea (adjusted) 0.32 (0.24, 0.43) 7.05 Wake Forest (adjusted) 0.63 (0.48, 0.83) 7.38 NY State (adjusted, unmatched) 0.54 (0.50, 0.60) 10.70 STENT (adjusted) 0.58 (0.47, 0.71) 8.70 GHOST (adjusted) 0.28 (0.20, 0.39) 6.31 Montevergine 0.51 (0.39, 0.68) 7.30 DEScover (adjusted) 0.58 (0.40, 0.83) 5.81 Cedars Acute MI 0.22 (0.08, 0.62) 1.34 REAL (adjusted) 0.67 (0.59, 0.76) 10.17 Multicenter SVG (adjusted) 0.58 (0.28, 1.18) 2.41 RESTEM 0.62 (0.47, 0.80) 7.53 ERACI III (from RCT) 0.58 (0.39, 0.86) 5.35 D+L Overall Effects (I-squared 71.2%, p = 0.000) *Random (I2==71.2%) I-V Overall Fixed Effects 0.53 (0.47, 0.61) 100.00 0.53 (0.47,0.61), p<0.001 0.57 (0.54, 0.60) 0.57 (0.54,0.60) NOTE: Weights are from random effects analysis .1 Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 2.2 yrs Study ID TVR: Unadjusted Registries 55,531 patients, 12 registries ES (95% CI) Estimate (95% CI) % Weight (D+L) Weight (%) Mayo FFR Substudy 0.18 (0.04, 0.78) 1.82 Brazil Large Vessels 0.43 (0.17, 1.10) 3.80 McMaster STEMI (unadjusted) 0.29 (0.04, 1.86) 1.16 Wake Forest (unadjusted) 0.62 (0.48, 0.80) 11.08 NY State (unadjusted, unmatched) 0.56 (0.51, 0.62) 12.76 STENT (unadjusted) 0.74 (0.63, 0.87) 12.21 Montevergine 0.51 (0.39, 0.68) 10.77 DEScover (unadjusted) 0.63 (0.46, 0.87) 10.20 Cedars Acute MI 0.22 (0.08, 0.62) 3.32 REAL (unadjusted) 0.99 (0.91, 1.08) 12.84 RESTEM 0.62 (0.47, 0.80) 10.93 ERACI III (from RCT) 0.58 (0.39, 0.86) 9.11 *Random D+L Overall Effects (I-squared(I2==88.9%) 88.9%, p = 0.000) 0.60 (0.48, 0.74) 100.00 0.60 (0.48,0.74), p<0.001 Fixed Effects I-V Overall 0.73 (0.69, 0.77) 0.73 (0.69,0.77) NOTE: Weights are from random effects analysis .1 Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 2.2 yrs TVR: Adjusted Registries 63,456 patients, 11 registries Study ID % Weight ES (95% CI) Estimate (95% CI) (D+L) Weight (%) Ontario (matched) 0.69 (0.60, 0.80) 12.72 McMaster STEMI (adjusted) 0.32 (0.05, 1.92) 0.66 Washington Hosp Center (matched) 0.65 (0.49, 0.85) 9.74 Asan Korea (adjusted) 0.32 (0.24, 0.43) 9.38 Wake Forest (adjusted) 0.63 (0.48, 0.83) 9.78 NY State (adjusted, unmatched) 0.54 (0.50, 0.60) 13.65 STENT (adjusted) 0.58 (0.47, 0.71) 11.34 GHOST (adjusted) 0.28 (0.20, 0.39) 8.46 DEScover (adjusted) 0.58 (0.40, 0.83) 7.83 REAL (adjusted) 0.67 (0.59, 0.76) 13.05 Multicenter SVG (adjusted) 0.58 (0.28, 1.18) 3.39 D+L Overall (I-squared = 79.4%, p = 0.000) *Random Effects (I2=79.4%) 0.54 (0.46,0.63), (0.46, 0.63) p<0.001 100.00 0.54 0.58 0.58 (0.54,0.61) (0.54, 0.61) I-V Overall Fixed Effects NOTE: Weights are from random effects analysis .1 Favors DES 1 Favors BMS Ajay J. Kirtane and Gregg W. Stone, 2008 10 Mean f/u 2.2 yrs Summary: DES vs. BMS Treatment Effect Estimates RCTs - Fixed effects - Random effects Registries - Fixed effects - Random effects Mortality MI TVR 8,867 pts, 21 trials 8,850 pts, 20 trials 7,291 pts, 16 trials 0.97 0.97 0.94 0.94 0.51 0.45* 161,232 pts, 28 studies 129,955 pts, 24 studies 73,819 pts, 17 studies 0.83 0.80* 0.96 0.89* 0.57 0.53* <1.0 DES better Study Limitations • Randomized trial analyses are still underpowered to assess these clinical endpoints • Registry analyses are based upon observational, non-randomized analyses • Potential for residual confounding • Significant heterogeneity, despite attempts to address this through random effects models, meta-regression and sensitivity analyses • Analysis was primarily of summary-level data and included unpublished studies • Use of hazard ratio / relative risk assumes constant hazards throughout the FU period Conclusions (1) In 22 RCTs in which 9,470 pts were randomized to DES or BMS and followed for ≥1 yr, DES resulted in: • Non significant 3% and 6% reductions in mortality and MI respectively • A highly significant 55% reduction in TVR In 30 registries in which 174,302 pts were treated with either DES or BMS (non-randomized) and followed for ≥1 yr, DES was associated with: • A highly significant 20% reduction in mortality • A significant 11% reduction in MI • A highly significant 47% reduction in TVR Conclusions (2) The favorable results of DES from the RCT and registry analysis populations were robust and consistent for both on-label and off-label use, and for clinical f/u extending to 3-4 years These findings, derived from more than 180,000 pts treated in 52 studies, strongly suggest that DES are safe for both on-label and off-label use, and have comparable efficacy in both RCTs and in the “real-world”