File carboxylic acids-Chap 10 & 11
advertisement

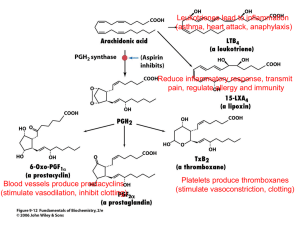
Chapters 10 and 11 -Carboxyl Group since a combination of carbonyl and hydroxyl groups. (see below) -nomenclature: common names usually used. -for IUPAC, simply add "oic acid" as suffix Nomenclature • Suffix is “oic” • COOH always at end of chain and always the #1 carbon • carbon understood to be #1 and therefore not stated as part of the name • • • • Examples: 4-Bromopentanoic acid heptanoic acid 2-ethyl-3-hydroxy-5-methyl-hexanoic acid Common Carboxylic Acids and where they are found: Formic Acid---red ants and bee stings Acetic Acid ---Vinegar Butyric Acid-- Rancid Butter, Locker Rooms Caproic, Caprylic,Capric - Goats Lactic Acid (Hydroxy acid) - Muscles, anaerobic respiration Citric Acid (Hydroxy acid) - citrus fruits Oxidation of Alcohol and Carboxylic Acids • Alcohol • Ex: ethanol Aldehyde ethanal Carboxylic Acid ethanoic acid -Fat + NaOH = soap -if vegetable fat, softer soap -hydrophobic portion dissolves dirt (if dirt non-soluble in water) and hydrophilic portion attaches to water to be washed away -in hard water (Mg and Ca), minerals will precipitate out to leave "ring" around tub. -detergents will not allow precipitation of minerals HOW SOAPS AND DETERGENTS WORK • These types of energy interact and should be in proper balance. Let's look at how they work together. Let's assume we have oily, greasy soil on clothing. Water alone will not remove this soil. One important reason is that oil and grease present in soil repel the water molecules. Now let's add soap or detergent. The surfactant's water-hating end is repelled by water but attracted to the oil in the soil. At the same time, the water-loving end is attracted to the water molecules. These opposing forces loosen the soil and suspend it in the water. Warm or hot water helps dissolve grease and oil in soil. Washing machine agitation or hand rubbing helps pull the soil free. Soap Diagram Soap and Detergents -Acid + Base water and a salt Formic Acid + Sodium Hydroxide -important preservatives -examples: Sodium Benzoate: inhibits mold and bacteria Calcium/Sodium Propionate: cheese, cakes, bread -relate to preservatives…see website on additives H2O Food additives • • • • • Artificial additives and behavior website Artificial colors website List of additives and preservative website Safety of food additives/list website http://www.cspinet.org/reports/chemcuisine. htm#Alphabetical Carboxylic Esters and Flavors • • • • • Ethyl Formate Ethyl Butyrate Methyl Butyrate Octyl Acetate Methyl Salicylate Rum Pineapple Apple Orange Wintergreen Esters are produced when carboxylic -Flavoring Agents acids are heated with alcohols in the -Alcohol + Carboxylic Acid = Ester presence of an acid catalyst. The catalyst is usually concentrated -Ethanol + Formic acid sulphuric acid. -Methanol + Butyric Acid Methyl Salicylate = Wintergreen Wintergreen Lab website-natural wintergreen equation website • Procedure: – – – – – 3 mLs. Methanol into test tube .5 grams Salicylic Acid into test tube Mix thoroughly Add 10 drops of H2SO4(sulfuric acid) to test tube (this acts as a catalyst) Place test tube into a beaker of water that is at 60-700C at keep test tube in this beaker for approx. 10 minutes – After 10 minutes, remove test tube and add hot water to fill test tube (do not add more than 20 mLs. of hot water…use water from 600 beaker) – Mix and detect odor…pour small amount onto a paper towel and detect odor • Discussion: – Thoroughly explain and show chemical equation for the formation of a carboxylic ester and methyl salicylate in particular. – Explain how/why esters are used in the food industry – Explain the difference between “all natural” and “natural flavors” as it relates to foods • • • • • • • teaberry Ericaceae Gaultheria procumbens L. Leaf: Alternate, simple, evergreen, oval to elliptical, 1 to 2 inches long, minutely serrated, thickened with a wintergreen odor when crushed, leaves appear whorled since they cluster at tips of plant; dark shiny green above, much paler below often with black dots. Flower: Monoecious; small (1/4 inch), white, urn-shaped, hanging from short stems from leaf axils, appearing in mid to late summer. Fruit: Red, round, 1/4 to 1/2 inch in diameter, hanging beneath the leaves, mild wintergreen taste, ripen in late summer and persist into winter. Twig: Slender, green turning brown with age. Bark: Light brown. Form: Low plant with a height of only 3 to 5 inches; stems shoot out of the ground and end in a tight cluster of leaves. • Drug patents • • • • A generic drug (pl. generic drugs, short: generics) is a drug which is produced and distributed without patent protection. A generic must contain the same active ingredients as the original formulation. In most cases, it is considered bioequivalent to the brand name counterpart with respect to pharmacokinetic and pharmacodynamic properties. By extension, therefore, generics are assumed to be identical in dose, strength, route of administration, safety, efficacy, and intended use. In most cases, generic products are not available until the patent protections afforded to the original developer have expired. When generic products become available, the market competition often leads to substantially lower prices for both the original brand name product and the generic forms. The time it takes a generic drug to appear on the market varies. Drug patents give twenty years of protection, but they are applied for before clinical trials begin, so the effective life of a drug patent tends to be between seven and twelve years. Generic Drugs website Pricing of Generic vs. Patent Drugs article US vs. Canada and Drug laws article OTC Analgesics (over the counter) -aspirin (acetylsalicyclic acid) -salicylic acid and willow tree bark -1893 Bayer Company -anti-coagulant -anti-inflammatory -anti-pyretic -Reye's syndrome (NOT for children!) -medline website -Ibuprofen -Advil, Motrin, Nuprin -anti-inflammatory, fever reduction -allergic reaction mimics Meningitis -stomach upset side effect medline website -acetaminophen -Tylenol -fever reduction -liver damage if alcohol use -medline website -naproxen sodium -longer lasting than ibuprofen -”Aleve” -anti-inflammatory -jaundice as side-effect -medline website Non-steroidal anti-inflammatory drugs • Salycilates: Aspirin (Acetylsalicylic Acid), Diflunisal, Ethenzamide • Arylalkanoic acids: Diclofenac, Indometacin, Sulindac • 2-Arylpropionic acids (profens): Carprofen, Flurbiprofen, Ibuprofen, Ketoprofen, Ketorolac, Loxoprofen, Naproxen, Tiaprofenic acid, • N-Arylanthranilic acids (fenamic acids): Mefenamic acid – Pyrazolidine derivatives: Phenylbutazone • Oxicams: Meloxicam, Piroxicam -- Coxibs: Celecoxib, Etoricoxib, Parecoxib, Rofecoxib, Valdecoxib -Sulphonanilides: Nimesulide OTC Analgesics Aspirin Ibuprofen Acetaminophen Naproxen Sodium Ketoprofen Aspirin (acetylsalicylic acid) • • • • analgesic (against minor pains and aches) antipyretic (against fever) anti-inflammatory antiplatelet (“blood-thinning”) effect and is used in long-term lowdoses to prevent heart attacks and cancer (high doses right after an attack) (anticoagulant) • Its primary undesirable side effects, especially in higher doses, are gastrointestinal distress (including ulcers and stomach bleeding) and tinnitus (ringing in ear) • It is generally recommended that one seek medical help if symptoms do not improve after a few days of therapy • Children, including teenagers, are discouraged from using aspirin in cold or flu symptoms as this has been linked with Reye's syndrome. Aspirin (acetylsalicylic acid) • coined by the Bayer company of Germany • Also known as ASA, ASS, AAS (depending on language) • bitter powder extracted from willow bark eases aches and pains and reduce fevers • active extract of the bark, called salicin (when oxidized becomes salicylic acid) 2-(acetyloxy)benzoic acid Formula C9H8O4 C6H4(OCOCH3)CO OH Reye’s Syndrome • Website-Aspirin and Reye’s Syndrome • What is Reye's Syndrome? • Reye's Syndrome is a disease which affects all organs of the body, but most lethally the liver and the brain. Reye's Syndrome is a two-phase illness because it is almost always associated with a previous viral infection, such as influenza, cold, or chicken pox. Scientists do know that Reye's Syndrome is not contagious and the cause is unknown. Reye's Syndrome is often misdiagnosed as encephalitis, meningitis, diabetes, drug overdose, poisoning, Sudden Infant Death Syndrome, or psychiatric illness. Aspirin and Salicylic Acid • Aspirin or acetylsalicylic acid, is a derivative of salicylic acid that is a mild, nonnarcotic analgesic useful in the relief of headache and muscle and joint aches. The drug works by inhibiting the production of prostaglandins, body chemicals that are necessary for blood clotting and which also sensitize nerve endings to pain. • The father of modern medicine was Hippocrates, who lived sometime between 460 B.C and 377 B.C. Hippocrates was left historical records of pain relief treatments, including the use of powder made from the bark and leaves of the willow tree to help heal headaches, pains and fevers. • By 1829, scientists discovered that it was the compound called salicin in willow plants which gave you the pain relief. • Website on history of aspirin • Aspirin info website Ibuprofen • nonsteroidal anti-inflammatory drugs (NSAIDs) • works by reducing hormones that cause inflammation and pain in the body(prostaglandins) • marketed under various trademarks including Act-3, Advil, Brufen, Herron Blue, Motrin, Nuprin, Dorival and Nurofen • used for relief of symptoms of arthritis, primary dysmenorrheal, fever, and as an analgesic, especially where there is an inflammatory component • believed to work through inhibition of cyclooxygenase (COX), thus inhibiting prostaglandin synthesis • Severe allergic reactions mimic meningitis symptoms • Ibuprofen Ibuprofen has a dosedependent duration of action of approximately 4–8 hours, which is longer than suggested by its short half-life • recommended dose varies with body mass •made available under prescription in the United Kingdom in 1969 yet now is an over the counter drug 2-[4-(2-methylpropyl)phenyl]propanoic acid Formula C13H18O2 Acetaminophen (Paracetamol) • a common analgesic and antipyretic drug that is used for the relief of fever, headaches, and other minor aches and pains • major ingredient in numerous cold and flu medications • Tylenol • Minimizes side-effects when combined with opioid analgesics • compounds contained in cinchona bark, similar to the willow bark in aspirin (same salicylic acid) • Liver damage has been linked to use of acetaminophen and alcohol in combination Acetaminophen (Paracetamol) • Often combined with codeine (an opioid) and caffeine • paracetamol acts by reducing production of prostaglandins, which are involved in the pain and fever processes, by inhibiting the cyclooxygenase (COX) enzyme • important differences between the effects of aspirin and those of paracetamol - less side effects - does not reduce blood clotting - no detrimental side effects to the lining of the stomach N-(4-hydroxyphenyl)acetamide Formula C8H9NO2 Naproxen Sodium • marketed under various trade names including: Aleve, • • • • Anaprox, Naprogesic, Naprosyn, Naprelan non-steroidal anti-inflammatory drug (NSAID) commonly used for the reduction of mild to moderate pain, fever, inflammation and stiffness caused by conditions such as arthritis naproxen is capable of producing disturbances in the gastrointestinal tract. Increases risk of cardiovascular problems. not recommended for use with NSAIDs of the salicylate family prescription drug Naprosyn in 1976, remains a prescription drug is most of the world. However is OTC in USA under trademark Aleve. Naproxen Sodium • member of the 2arylpropionic acid (profen) family of NSAIDs • It is an odorless, white to off-white crystalline substance • Prescribed for people suffering from: Formula C14H14O3 (+)-(S)-2-(6-methoxynaphthalen-2-yl) propanoic acid osteoarthritis, rheumatoid arthritis, psoriatic arthritis, gout, ankylosing spondylitis, injury (like fractures), menstrual cramps, tendonitis, bursitis, and the treatment of primary dysmenorrhea Ketoprofen • one of the propionic acid class of non-steroidal antiinflammatory drug (NSAID) with analgesic and antipyretic effects. It acts by inhibiting the body's production of prostaglandin • Brand names in the US are Orudis and Oruvail • 12.5 mg. OTC, greater amounts are prescription 2-(3-benzoylphenyl)propanoic acid Formula C16H14O3 NSAIDS • • • • What is an NSAID? Non-steroidal anti-inflammatory drug How does it work? Reduces inflammation, that leads to pain. Works by inhibition of enzyme COX-1 or COX2…cyclooxygenase, which is needed to produce Prostaglandins…chemical that increases swelling and therefore pain NSAIDS continued… Why do NSAIDS bother the stomach? • One type of prostaglandin is needed to make the protective lining of the stomach. So, if this chemical is reduced, the stomach physiology is altered…pain or ulcer can result. How are COX-2 inhibitors different than COX-1? • COX-1 related to stomach, COX-2, is not. So, new class of COX-2 inhibitors on market…Celebrex (Vioxx,Bextra removed. linked to increase cardiovascular risk) Cox-1 • Cox-1 inhibitor: An agent that inhibits the action of the enzyme cox-1 (cyclooxygenase-1). The common anti-inflammatory drugs such as aspirin, ibuprofen, and naproxen block the action of both cox-1 and cox-2. Cox-1 inhibitors can reduce inflammation, but they may also decrease the natural protective mucus lining of the stomach. Therefore, these medications can cause stomach upset, intestinal bleeding, and ulcers. In some cases, using a buffered form of a cox-1 inhibitor can eliminate or reduce these adverse effects. Cox-2 problems? • As many of our readers know, Vioxx is a drug that belongs to a category of drugs called the Cox-2 inhibitors since these drugs target an enzyme in the stomach called Cox-2. Vioxx was recalled because it increased the risk of heart attacks (tens of thousands of Vioxx patients died from these heart attacks and others have suffered irreparable damage to their cardiovascular systems). • There are two other drugs on the market right now that belong to the Cox-2 category: Celebrex and Bextra (they are sometimes sold by other brand names outside the United States). When it was found that Bextra was causing many cardiovascular complications, FDA directed its maker, Pfizer, to add a warning to Bextra label. More research has shown that Celebrex also has adverse side effects but the data is not conclusive yet. Pfizer is insisting that while both Celebrex and Bextra are Cox-2 drugs their molecular structure is different from that of Vioxx, and hence, they are not as dangerous as Vioxx. This is their main line of argument against recalling Celebrex and Bextra though most medical experts think that in the interest of patient safety, both these drugs should be recalled. (Related article: FDA issues an advisory against Celebrex and Bextra) Cox-2 problems cont. • • • • • • • A private research team claims to have identified a unique action of the painkiller Vioxx (search), which is independent of its anti-inflammatory action, that might explain why it’s linked to heart attack and stroke and why other similar drugs might not be. The researchers say the findings should reassure patients about the cardiovascular safety of other Cox-2 drugs (search), but two cardiologists contacted by WebMD remain unconvinced. Vioxx was withdrawn from the market by manufacturer Merck & Co. a month and a half ago after research showed that long-term use doubled the risk of heart attack and stroke. While some clinical trials have shown that another widely prescribed Cox-2, Pfizer’s Celebrex (search), is more heart friendly than Vioxx, The Cleveland Clinic cardiologist Steven E. Nissen, MD, says the jury is still out. Pfizer is a WebMD sponsor. “The clinical evidence to date has not implicated [Celebrex],” he tells WebMD. “But this drug has also has not been studied as carefully in patients with a high risk of cardiovascular disease.” Molecular Evidence The newly reported study was conducted by researchers with Elucida Research of Beverly, Mass., and published in the latest issue of the journal Atherosclerosis. Elucida founder R. Preston Mason, PhD, says the two-year study uncovered important differences in the action of the Cox-2 drugs. These important differences may be key to linking Vioxx to cardiovascular disease risk. Vioxx Websites • Recent news • Legal information Celebrex in the news • Website