The Science of Chemistry
advertisement

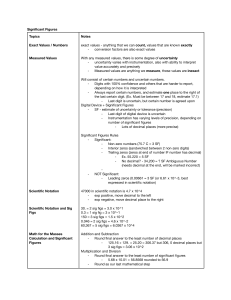
The Science of Chemistry Measurement Introduction When you hear the term chemistry, what comes to mind??? What do you think we are going to study?? Choose two class recorders - Brainstorm concepts on the board…. What is chemistry about?? The study of matter and the changes it undergoes Chemical reactions involve energy Involves physical and chemical changes Measurement – number and unit Metric Base Units Gram – mass Sec – time Meter – length Mole - amount Kelvin – temperature Metric prefixes Large Tera – 1012 times larger Giga – 109 times larger Mega – 106 times larger Kilo – 103 times larger Small Centi – 102 times smaller Milli – 103 times smaller Micro – 106 times smaller Nano – 109 times smaller Pico – 1012 times smaller Conversions using metric prefixes Create a metric prefix line Practice: How many liters are in 525 milliliters? How many centimeters are in 0.15 meters? How many micrograms are in 3 grams? Taking Measurements Mass – triple beam balance or electronic balance Volume – graduated cylinder Length – ruler Temperature – thermometer Uncertainty in Measurement Accuracy – how close measured values are to the true (accepted) value Precision – how close measured values are to each other Error Random Error Caused by unknown and unpredictable changes in the experiment – corrected by doing many experiments Experimenter’s interpretation of measurement reading Systematic Error Produce a result that is different from the actual value – cannot be corrected by repeating the experiment Significant Figures The measured figures and one additional estimated digit are counted as significant digits. Always use significant figures in labs and workout problems!!!! Rules for counting sig figs 1. 2. 3. 4. All nonzero figures are significant. Zeros count if the number contains a decimal AND the zero is preceded by a nonzero digit. Zeros between nonzero figures are significant. Exact numbers contain an infinite number of sig figs. Examples How many sig figs are in the measurements? a) b) c) d) e) f) g) h) i) j) 1.000 70 m 100 890 L 0.005 4 cm 17.10 kg 3.29 x 103 μg 3 200 000 g 6 000. cg 0.000 350 24 hrs = 1 day 60 s = 1 min a. 6 b. 5 c. 2 d. 4 e. 3 f. 2 g. 4 h. 3 i. infinite j. infinite Rules for Calculating with Sig Figs 1. 2. Multiplication/Division – Number of sig figs in result equals the total number of figures in the least precise measurement. Addition/Subtraction – Number of decimal places in the result equals the number of decimal places in the least precise measurement. Examples Complete the following calculations. Write answers with correct number of significant figures. a) b) c) d) 4.00 + 3.0 = 2.5 - 0.5 = 3.2 x 2 = 10. / 2.0 = a. 7.0 b. 2.0 c. 6 d. 5.0 Rounding 1. 2. 3. 4. Round 357 g to 2 significant figures. Round 65.233 cm to 3 significant figures. Round 0.000 684 L to 1 significant figures. Round 1.256 x 102 mL to 2 significant figures. Scientific Notation Shorthand way of writing very large or small measurements Must include all significant figures Examples Write the following numbers in scientific notation. 1. 2. 3. 4. 65 000 0.000 000 85 0.000 008 00 340. 1. 2. 3. 4. 6.5 x 104 8.5 x 10-7 8.00 x 10-6 3.40 x 102 More Examples Write in decimal notation 1. 2. 3. 4. 2 500 3.400 0.000 000 85 0.000 000 960