Chem - Lakewood City Schools
advertisement

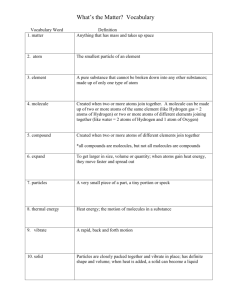
Chemical Foundations for Cells Chapter 6 Chemical Benefits and Costs • Understanding of chemistry provides fertilizers, medicines, etc. • Chemical pollutants damage ecosystems Elements • Fundamental forms of matter • Can’t be broken apart by normal means • 92 occur naturally on Earth Most Common Elements in Living Organisms Oxygen = 65% Hydrogen = 9.5% Carbon = 18.5% Nitrogen = 3.3% What Are Atoms? • Smallest particles that retain properties of an element • Made up of subatomic particles: – Protons (+) – Electrons (-) – Neutrons (no charge) Hydrogen and Helium Atoms electron proton neutron HYDROGEN HELIUM Atomic Number • Number of protons • All atoms of an element have the same atomic number • Atomic number of hydrogen = 1 • Atomic number of carbon = 6 Mass Number Number of protons + Number of neutrons Isotopes vary in mass number Isotopes • Atoms of an element with different numbers of neutrons (different mass numbers) • Carbon 12 has 6 protons, 6 neutrons • Carbon 14 has 6 protons, 8 neutrons What Determines Whether Atoms Will Interact? The number and arrangement of their electrons Atoms seek to be more stable – complete orbitals Electrons • Carry a negative charge • Repel one another • Are attracted to protons in the nucleus • Move in orbitals - volumes of space that surround the nucleus y Z X When all p orbitals are full Electron Orbitals • Orbitals can hold up to two electrons • Atoms differ in the number of occupied orbitals • Orbitals closest to nucleus are lower energy and are filled first Shell Model • First shell – Lowest energy – Holds 1 orbital with up to 2 electrons • Second shell – 4 orbitals hold up to 8 electrons CALCIUM 20p+ , 20e- Electron Vacancies • Unfilled shells make atoms likely to react • Hydrogen, carbon, oxygen, and nitrogen all have vacancies in their outer shells CARBON 6p+ , 6e- NITROGEN 7p+ , 7e- HYDROGEN 1p+ , 1e- Chemical Bonds, Molecules, & Compounds • Bond is union between electron structures of atoms • Atoms bond to form molecules • Molecules may contain atoms of only one element - O2 • Molecules of compounds contain more than one element - H2O Chemical Bookkeeping • Use symbols for elements when writing formulas • Formula for glucose is C6H12O6 – 6 carbon atoms – 12 hydrogen atoms – 6 oxygen atoms Chemical Bookkeeping • Chemical equation shows reaction Reactants ---> Products • Equation for photosynthesis: 6CO2 + 6H2O ---> + C6H12O12 + 6H2O Important Bonds in Biological Molecules Ionic Bonds Covalent Bonds Hydrogen Bonds Covalent Bonding Atoms share a pair or pairs of electrons to fill outermost shell •Single covalent bond •Double covalent bond •Triple covalent bond Nonpolar Covalent Bonds • Atoms share electrons equally • Nuclei of atoms have same number of protons • Example: Hydrogen gas (H-H) Polar Covalent Bonds • Number of protons in nuclei of participating atoms is NOT equal • Electrons spend more time near nucleus with most protons • Water - Electrons more attracted to O nucleus than to H nuclei Ion Formation • Atom has equal number of electrons and protons - no net charge • Atom loses electron(s), becomes positively charged ion • Atom gains electron(s), becomes negatively charged ion Ionic Bonding • One atom loses electrons, becomes positively charged ion • Another atom gains these electrons, becomes negatively charged ion • Charge difference attracts the two ions to each other Formation of NaCl • Sodium atom (Na) – Outer shell has one electron • Chlorine atom (Cl) – Outer shell has seven electrons • Na transfers electron to Cl forming Na+ and Cl• Ions remain together as NaCl Formation of NaCl 7mm electron transfer SODIUM ATOM 11 p+ 11 e- CHLORINE ATOM 17 p+ 17 e- SODIUM ION 11 p+ 10 e- CHLORINE ION 17 p+ 18 eFig. 2.10a, p. 26 Hydrogen Bonding • Molecule held together by polar covalent bonds has no NET charge • However, atoms of the molecule carry different charges • Atom in one polar covalent molecule can be attracted to oppositely charged atom in another such molecule Examples of Hydrogen Bonds one large molecule another large molecule a large molecule twisted back on itself Fig. 2.12, p. 27 Hydrogen Ions: H+ • Unbound protons • Have important biological effects • Form when water ionizes The pH Scale • Measures H+ concentration of fluid • Change of 1 on scale means 10X change in H+ concentration Highest H+ Lowest H+ 0---------------------7-------------------14 Acidic Neutral Basic Acids & Bases • Acids – Donate H+ when dissolved in water – Acidic solutions have pH < 7 • Bases – Accept H+ when dissolved in water – Acidic solutions have pH > 7 Properties of Water Polarity Temperature-Stabilizing Cohesive Solvent Water Is a Polar Covalent Molecule • Molecule has no net charge • Oxygen end has a slight negative charge • Hydrogen end has a slight positive charge O H H Liquid Water H + + _ O H + H + + _ O H + Water Cohesion • Hydrogen bonding holds molecules in liquid water together • Creates surface tension • Allows water to move as continuous column upward through stems of plants Temperature-Stabilizing Effects • Liquid water can absorb much heat before its temperature rises • Why? • Much of the added energy disrupts hydrogen bonding rather than increasing the movement of molecules Why Ice Floats • In ice, hydrogen bonds lock molecules in a lattice • Water molecules in lattice are spaced farther apart then those in liquid water • Ice is less dense than water Water Is a Good Solvent • Ions and polar molecules dissolve easily in water • When solute dissolves, water molecules cluster around its ions or molecules and keep them separated Spheres of Hydration Diffusion • Brownian motion – molecules are in constant motion • Diffusion – movement from area of high concentration to area of low concentration – Affected by • Concentration • Temperature or agitation • Pressure Dynamic Equilibrium • Molecules are still in motion • No net gain or loss of molecules • Living systems seek to achieve Organic Compounds Hydrogen and other elements covalently bonded to carbon Carbohydrates Lipids Proteins Nucleic Acids Carbon’s Bonding Behavior • Outer shell of carbon has 4 electrons; can hold 8 • Each carbon atom can form covalent bonds with up to four atoms Bonding Arrangements • Carbon atoms can form chains or rings • Other atoms project from the carbon backbone Condensation Reactions • Form polymers from subunits • Enzymes remove -OH from one molecule, H from another, form bond between two molecules • Discarded atoms can join to form water CONDENSATION enzyme action at functional groups Fig. 3.4a, p. 37 Hydrolysis • A type of cleavage reaction • Breaks polymers into smaller units • Enzymes split molecules into two or more parts • An -OH group and an H atom derived from water are attached at exposed sites HYDROLYSIS enzyme action at functional groups Fig. 3.4b, p. 37 Carbohydrates – energy source Monosaccharides (simple sugars) Disaccharides (two simple sugars) Polysaccharides (complex carbohydrates) Monosaccharides • Simplest carbohydrates • Most are sweet tasting, water soluble • Most have 5- or 6-carbon backbone Glucose (6 C) Fructose (6 C) Ribose (5 C) Deoxyribose (5 C) Two Monosaccharides glucose fructose Disaccharides • Two monosaccharides covalently bonded • Formed by condensation reaction glucose fructose + H2O sucrose Polysaccharides • Straight or branched chains of many saccharides • Most common are composed entirely of glucose – Cellulose • tough, indigestible • structural material in plants – Starch • easily digested • storage form in plants – Glycogen • sugar storage form in animals – Chitin • structural material for hard parts of invertebrates • cell walls of many fungi Lipids • Most include fatty acids – Fats – Phospholipids – Waxes • Tend to be insoluble in water • Energy source, insulation & protection Fatty Acids • Carboxyl group (-COOH) at one end • Carbon backbone (up to 36 C atoms) – Saturated - Single bonds between carbons – Unsaturated - One or more double bonds Three Fatty Acids What difference does the double bond make? stearic acid oleic acid linolenic acid Fats • Fatty acid(s) attached to glycerol • Triglycerides are most common Proteins • Carbon, hydrogen, oxygen, nitrogen & sulfur • Amino acid building blocks • AA linked by peptide bonds • Enzymes • Build tissue Enzymes • Protein • Act as catalyst – Helps reaction happen faster or at lower temperatures • Substrate specific shapes – Lock & key system – Recycled; not used up Denaturation • Disruption of threedimensional shape • Breakage of weak bonds • Causes of denaturation: – pH – Temperature • Destroying protein shape disrupts function Nucleic Acids • Carbon, hydrogen, oxygen, nitrogen & phosphorus • Nucleotides – building blocks • DNA, RNA • Genetic information