Lecture 19 Slides
advertisement

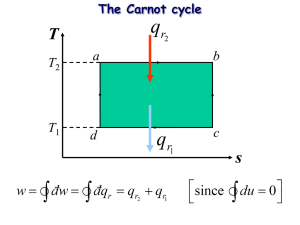
COP Refrigerators and Heat Pumps Reverse World http://www.youtube.com/watch?v=EahHTh BjDB0 Test 2 Starts Friday Oct 18 Ends Monday Oct 21 Late day Tuesday Oct 22 16 Multiple Choice 4 Free Response Give yourself plenty of time! Entropy is a measure of disorder… But what does that mean? Micro vs Macro States Entropy is about probability. It is very improbable that we will ever observe a process that decreases the entropy of a real physical process. In the limit as N goes to infinity, probability becomes fact. Consider the air in this room. Entropy – it’s different things to different people Information (compression) Parking a Car Clutter Thermodynamic 𝑠 = 𝑘𝐵 ln(𝑤) – Quantization Reversible vs. Irreversible Irreversible – Free expansion (adiabatic-ish) – Heat conduction – Scrambling Eggs, Bar room fight Reversible – Adiabatic expansion – Constant T expansion (at least for the gas) – Ice melting (sort of) Isolated vs Open Systems Water runs over a waterfall. What kind of process is this? A. B. Reversible Irreversible Reversible process - one that works equally well in reverse, one whose effect you can undo Irreversible process - one you cannot undo or will not spontaneously run in reverse Pretty much all spontaneous events in nature are irreversible irreversible scrambling eggs eating fish reversible ice liquid at 0oC liquid steam at 100oC Puff Balls Law of Increasing Disorder (2nd Law of Thermodynamics) Changes occurring in natural systems always proceed in such a way that the total amount of disorder in a closed system never decreases, and it increases in irreversible processes. http://www.youtube.com/watch?v=X4zd4Qpsbs8&NR=1 2 Ne 9:28 O that cunning plan of the evil one! O the vainness, and the frailties, and the foolishness of men! When they are learned they think they are wise, and they hearken not unto the counsel of God, for they set it aside, supposing they know of themselves, wherefore, their wisdom is foolishness and it profiteth them not. And they shall perish. Increase in Entropy = Increase in disorder f dQreversible S T i Ice Melting Isothermal Expansion Which path has a larger ∆S P A B V C. The same D. Not enough information Quick Quiz 22.6 The entropy change in a free expansion must be zero because Q=0 A. True B. False Non-reversible ideal gas process Any path will do. Pick one you can calculate! S ncV ln Tf Ti nR ln Vf Vi If an ideal gas undergoes a nonreversible process, which of the following will be true? A. B. C. D. The entropy will increase The entropy will decrease The entropy will stay the same It depends Free expansion The change in entropy for a free expansion is a) Positive b) Negative c) Zero d) I do not know Free expansion Free expansion followed by isothermal compression Conduction HOT COLD Ordered WARM Disordered Universal Heat Death “This is the way the world ends, not with a bang but a whimper.” T.S. Eliot Gospel perspective "every thing restored to its perfect frame" Alma 11:44