AP Biology Review Unit 6: Energy Conversions & Enzymes and Cell
advertisement

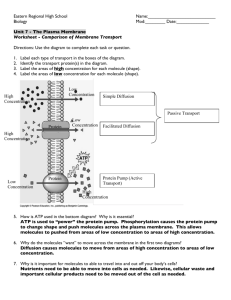
AP Biology Review Unit 6: Energy Conversions & Enzymes and Cell Cycle and Cell Communication 2A1: All living system require constant input of free energy. 2A2: Organisms capture free energy and store free energy for use in biological processes. 3A2: In eukaryotes, heritable information is passed to the next generation via processes that include the cell cycle and mitosis or meiosis plus fertilization. 3B2: A variety of intercellular and intracellular signal transmissions mediate gene expression. 3D1: Cell communication processes share common features that reflect a shared evolutionary history. 3D2: Cells communicate with each other through direct contact with other cells or from a distance via chemical signaling. 3D3: Signal transduction pathways link signal reception with cellular response. 3D4: Changes in signal transduction pathways can alter cellular response. 4B1: Interactions between molecules affect their structure and function 1. a. b. c. d. 2. a. b. c. d. e. f. g. h. i. j. k. l. Energy Organisms use free energy for organization, growth and reproduction. Loss of order or free energy flow results in death. More free energy (ex. Food) than needed will be stored for growth (roots, glycogen, fat, etc.). Matter and energy are not created but change form (1st law of thermo; ex. Sun energy to bond energy in glucose) and entropy is increasing in disorganization of energy (i.e. heat released by cell respiration). More organized or built up compounds have more free energy and less entropy (i.e. glucose) and less organized have less free energy and more entropy (i.e. carbon dioxide). Reactions can be coupled to maintain a system, ex. Photosynthesis and cell respiration Enzymes Biological catalysts (made of protein) that speed up rate of chemical reactions by lowering activation energy required for reaction to occur Enzyme has active site (exposed R groups) where reaction occurs Enzymes can break down substance (catabolic reaction) or build up substances (anabolic) Enzyme/substrate complex is formed Substrate is what enzyme acts on Rate is determined by collisions between substrate and enzyme Ends in –ase, named after substrate often Enzyme is specific to substrate; the substrate must be complementary to the surface properties (shape and charge) of the active site (which is made up of R groups with specific chemistry, i.e. hydrophobic). Enzyme rate is affected by: pH (optimal for each enzyme), temperature (optimal for each enzyme but in general increased temp means increased collisions so rate goes up initially; too much heat can denature enzyme), enzyme concentration (more enzyme faster rate or vice versa) substrate concentration (more substrate faster rate; vmax is fastest enzyme can work when saturated) Inhibition-competitive inhibition (something competes for active site; can be overcome with more substrate) Non-competitive inhibition- attaches at allosteric site and changes shape of enzyme so it is not functional; can not be overcome with more substrate Coenzymes (organic; NAD and vitamin B etc.) and cofactors (inorganic; zinc, magnesium etc.) interact with enzymes to put them into the right structure to do work. 3. Energy Transformations ATP- adenosine triphosphate- energy molecule for cells (recyclable) A. Photosynthesis 6CO2 + 6H2O C6H12O6 + 6O2 Photosynthetic organisms capture free energy present in sunlight and use water and carbon dioxide to make carbon products and free oxygen. Light-dependent reactions- photophosphorylation (1) Photosystems I and II (chlorophyll and proteins) are embedded in the internal membranes of chloroplasts (thylakoids of the grana). They pass electrons through an electron transport chain (ETC). When electrons are passed they allow hydrogen ions (protons) across the thykaloid membrane. The formation of the proton gradient powers the process of ATP synthesis to add a phosphate ADP to ATP (chemiosmosis). (2) Electrons are passed to NADP+ to make NADPH (electron carrier) (3) H2O is used and O2 released as by-product (4) Red and blue light works best (green is reflected typically) (5) Pigments= chlorophyll a and b; accessory pigments (6) Energy converted from sun into chemical energy of ATP and NADPH to be used in building of sugar (Calvin Cycle) Light-independent reactions- Calvin Cycle (1) carbon fixation occurs (2) occurs in stroma of chloroplasts (3) ATP and NADPH are used; rubisco is enzyme that fixes carbon (not picky and will fix O 2 toophotorespiration; “the ho”) Evolution of Photosynthesis (1) Photosynthesis first evolved in prokaryotic organisms; (bacterial) photosynthesis was responsible for the production of an oxygenated atmosphere (2) C4 photosynthesis- crabgrass, corn, drought resistance, uses new enzyme- PEP carboxylase which is specific for just CO2 and CAM- Crassulacean Acid metabolism used in dry climates, ex. Cacti stomates are closed during day and open at night B. Cellular respiration C6H12O6 + 6O26CO2 + 6H2O Makes ATP for cell use; uses glucose and oxygen makes waste products of carbon dioxide and water; occurs in mitochondria; NADH is electron carrier used Glycolysis (1) occurs in cytoplasm; anaerobic (2) rearranges the bonds in glucose molecules, releasing free energy to form ATP from ADP resulting in the production of pyruvate. Kreb’s cycle (1) occurs in mitochondrial matrix (2) also called the citric acid cycle (3) occurs twice (one for each acetyl co-a) (4) Pyruvate is oxidized further and carbon dioxide is released ; ATP is synthesized from ADP and inorganic phosphate via substrate level phosphorylation and electrons are captured by coenzymes (NAD+ and FAD). (5) (6) NADH and FADH2 carry them to the electron transport chain. The electron transport chain captures in a process similar to light dependent reactions to make ATP. At end 38 ATP made per glucose molecule- could be 36 if it cost ATP to get in mitochondria; some of the energy in bonds is lost as heat (not completely efficient but since it occurs in several reactions with enzymes it is more efficient than one combustion) Anaerobic Fermentation (1) No oxygen; cell only goes through glycolysis followed by fermentation (2) Fermentation recycles NAD needed to restart glycolysis (3) alcohol fermentation ex. yeast cells- glucose ethyl alcohol + CO2+ NAD (4) lactic acid fermentation ex. muscle cells- glucose lactic acid + NAD (5) Fermentation does not make ATP but glycolysis does- 2ATP; very inefficient; sufficient for microorganisms 4. a. b. c. d. e. f. g. h. i. j. k. l. m. o. 5. a. b. Cell cycle Reason for division- as cells increase in volume, the surface area decreases and demand for material resources increases which limits cell size Smaller cells have a more favorable surface area-to-volume ratio for exchange of materials with the environment (diffusion, etc.). High SA:V ratio is favorable. Ex. 6:1 is better than 6:5 Cell cycle switches between interphase and cell division. Interphase has three phases: growth (G1), synthesis of DNA (S) and preparation for mitosis (G2). During mitosis duplicated chromosomes line up in center with spindle fibers attached to help pull them apart. Duplicated chromosomes are pulled apart by spindle fibers. Cytokinesis-division of cytoplasm and reformation of cell membrane. Animal cell- pinches in (cleavage) using microfilaments; plant cell- form cell plate reforms cell wall (golgi deposits material). The cell cycle is directed by internal controls or checkpoints. Internal (enzymes and promoting factors) and external signals (growth factors) provide stop and- go signs at the checkpoints. Ex. Mitosispromoting factor (MPF); Platelet-derived growth factor (PDGF); p53 gene products Cancer results from disruptions in cell cycle control (too much division, defective tumor suppressor genes, overactive genes) which are a result of DNA damage to proto-oncogenes (regulatory genes)which make products like cyclins and cyclin-dependent kinases. Cells spend different amounts of time in interphase or division. Nondividing cells may exit the cell cycle; or hold at a particular stage in the cell cycle. Mitosis is used for growth and repair in animals; plants use mitosis to make gametes and for growth or repair. Mitosis usually begins with 1 cell, makes 2 identical cells or clones; maintains chromosome number; 1n1n or 2n2n. Meiosis (occurs after interphase) takes diploid cells and reduces the chromosome number to haploid. 2n1n. During meiosis, homologous chromosomes are paired (one from mom and one from dad) and line up in the center of the cell randomly. The homologues are pulled apart and separated in meiosis I. A second division occurs in which the duplicated chromosomes are pulled apart. Variation occurs in gametes during “crossing over,” and fertilization because of all possible combinations. Cell to Cell Communication Cells receive or send inhibitory or stimulatory signals from other cells, organisms or the environment. In single-celled organisms it is response to its environment. Ex. quorum sensing, d. e. f. g. h. i. j. k. In multicellular organisms, signal transduction pathways coordinate the activities within individual cells. Ex. Epinephrine stimulation of glycogen breakdown in mammals Cells communicate by cell-to-cell contact. Ex Immune cells interact by cell-cell contact, antigenpresenting cells (APCs), helper T-cells and killer T cells or plasmodesmata between plant cells that allow material to be transported from cell to cell. Cells communicate over short distances by using local regulators that target cells in the vicinity of the emitting cell. Ex. Neurotransmitters, plant immune response Signals released by one cell type can travel long distances to target cells of another cell type. Ex. Hormones. A receptor protein recognizes signal molecules, causing the receptor protein’s shape to change, which initiates transduction of the signal. Ex. G-protein linked receptors, ligand-gated ion channels, tyrosine kinase receptors. Signal transduction is the process by which a signal is converted to a cellular response. Signaling cascades relay signals from receptors to cell targets, often amplifying the incoming signals, with the result of appropriate responses by the cell. Second messengers are often essential to the function of the cascade. Ex. cyclic AMP calcium ions (Ca2+), and inositol triphosphate (IP3) Many signal transduction pathways include: Protein modifications or phosphorylation cascades in which a series of protein kinases add a phosphate group to the next protein in the cascade sequence. ------------------------------------------------------------------------------------------------------------------------------------------------AP Lab Investigation 5 Photosynthesis Overview: Spinach cut out disks were placed in two different syringes (bicarbonate and without) and those photosynthetic rate was calculated by measuring the number that floated over time. Students then designed their own experiment to see what factors affected photosynthesis. IV: presence of bicarbonate DV: number of disks floating Equations: ET50 = the point at which 50% of the leaf disks are floating (the median or ET50, the Estimated Time it takes 50% of the disks to float), inverse relationship between rate and ET 50 so we graphed 1/ET50 in this lab. AP Lab Investigation 6 Cell Respiration Overview: Germinating and non-germinating seeds (peas) were placed in different temperature water baths and cell respiration rate was determined based on oxygen consumption. Design your own experiment to determine what other factors affect cell respiration (type of seed, age of seed, etc.) IV: germinating or non-germinating and temperature DV: O2 consumption *volume was controlled with glass beads, CO2 gas was controlled with KOH, temperature was controlled with water bath Equations: dY/dt or product formed (dY) over time interval (dt) AP Lab Investigation 7 Cell Cycle Part I: Mitosis Overview: Two treatment groups of plant root tips were compared, one group was treated with lectin (increases cell division) and the other was a control group that had not been treated with lectin (we used cards for these). Chi-square analysis was used to determine if there was a significant difference between the two groups. IV: Lectin DV: Rate of Division Equations: , x2 value is compared to chart under .05 probability and correct degrees of freedom (number of groups -1). Numbers at critical value or above reject null hypothesis. Part II: Meiosis Overview: Spores of a fungus were evaluated for crossing over and cross over rates were calculated. Also karyotypes were evaluated for cancer and genetic diseases as a result of cross over mistakes. Equations: # of crossover/total number of spores = % cross over, % cross over /2 = map units AP Lab Investigation 13 Enzymes Overview: Rate of decomposition (breakdown) of hydrogen peroxide by the enzyme peroxidase was measured by measuring the amount of O2 gas produced in the reaction. An indicator called guaiacol was used to detect oxygen by changing a darker color which was measured by a colorimeter (measures transmittance of light through a sample). Designing your own experiment to determine what other factors affect enzyme reaction (light, temperature, pH or concentrations). IV: Time DV: color change (indicates oxygen production) Equations: rate dY/dt (change in transmittance of light over change in time) ------------------------------------------------------------------------------------------------------------------------------------------------Cell Division: anaphase cancer cell cycle cellular differentiation cell division centrioles chromosome crossing over crossover frequency cyclin-dependent kinase cytokinesis differentiation diploid (2N) DNA replication fertilization gamete haploid (1N) homologous chromosomes independent assortment interphase maternal chromosome meiosis metaphase mitosis nuclear division p53 paternal chromosome potency prophase recombination sex chromosome somatic cell specialized cell synapsis telophase Communication communication cyclic AMP (cAMP) G-protein linked receptor phosphorylation cascade protein kinase quorum sensing receptor second messenger signal cascade signal transduction signal transduction pathway Cell Energy absorption spectrum accessory pigment acetyl coA action spectrum activation energy active site anabolism anaerobic metabolism allosteric regulation ATP autotroph Calvin cycle catabolism catalyst cellular respiration chemiosmosis chemoautotroph chlorophyll chloroplast citric acid cycle coenzyme cofactor compartmentalization consumer cyclic electron flow denaturation electron transport chain entropy endergonic reaction enzyme exergonic reaction feedback inhibition fermentation glycolysis heterotroph induced fit model light dependent reactions light independent reactions metabolic pathway mitochondrion NAD NADP negative feedback non-cyclic electron flow oxidative phosphorylation photolysis photosynthesis positive feedback ribulose bisphosphate substrate-level phosphorylation thylakoid membrane Questions and Practice 1. How do the laws of thermodynamics relate to the biochemical processes that provide energy to living systems? 2. How do enzymes regulate the rate of chemical reactions? 3. How does the specificity of an enzyme depend on its structure? 4. How is the activity of an enzyme regulated? 5. How does the cell cycle assure genetic continuity? 6. How does mitosis allow for the even distribution of genetic information to new cells? 7. What are the mechanisms of cytokinesis? 8. How is the cell cycle regulated? 9. How can aberrations in the cell cycle lead to tumor formation? 10. Why is meiosis important in heredity? 11. How is meiosis related to gametogenesis? 12. What are the similarities and differences between gametogenesis in animals and plants? 13. What is the role of ATP in coupling the cell’s anabolic and catabolic processes? 14. How does chemiosmosis function in bioenergetics? 15. How are organic molecules broken down by catabolic pathways? 16. What is the role of oxygen in energy-yielding pathways? 17. How do cells generate ATP in the absence of oxygen? 18. How does photosynthesis convert light energy into chemical energy? 19. How are the chemical products of the light-trapping reactions coupled to the synthesis of carbohydrates? 20. What kinds of photosynthetic adaptations have evolved in response to different environmental conditions? 21. What interactions exist between photosynthesis and cellular respiration? 22. How was photosynthetic rate measured in the photosynthesis lab? 23. What was the IV in the photosynthesis lab? DV? Control? Controlled variables? 24. How was respiration rate measured in respiration lab? 25. What was the IV in the lab? DV? Control? Controlled variables? 26. Make a concept map to relate the following terms: high free energy, low free energy, entropy, enzymes, photosynthesis, light dependent reaction, light independent reaction, cell respiration, glycolysis, krebs, etc, and fermentation. 27. Make a concept map to relate the following terms: cell cycle, interphase, growth, dna replication, mitosis, meiosis, homologous chromosomes, separation of chromosomes, cancer, checkpoints, regulatory proteins. 28. Make a concept map to relate the following terms: unicellular, multicellular, local regulators, long distance regulation, contact, receptor, signal transduction, enzyme cascade, response Membranes Plasma Membrane The plasma membrane surrounds the cell and functions as an interface between the living interior of the cell and the nonliving exterior. All cells have one. It regulates the movement of molecules into and out of the cell. Membrane Structure The fluid-mosaic model states that membranes are phospholipid bilayers with protein molecules embedded in the bilayer. Phospholipids Most of the lipids in a membrane are phospholipids. Phospholipids contain glycerol, two fatty acids, and a phosphate group. The phosphate group is polar (hydrophilic), enabling it to interact with water. The fatty acid tails are nonpolar (hydrophobic) and do not interact with water. Phospholipid Bilayers Phospholipids spontaneously form a bilayer in a watery environment. They arrange themselves so that the polar heads are oriented toward the water and the fatty acid tails are oriented toward the inside of the bilayer (see the diagram below). In general, nonpolar molecules do not interact with polar molecules. This can be seen when oil (nonpolar) is mixed with water (polar). Polar molecules interact with other polar molecules and ions. For example table salt (ionic) dissolves in water (polar). The bilayer arrangement shown below enables the nonpolar fatty acid tails to remain together, avoiding the water. The polar phosphate groups are oriented toward the water. Flexibility The fatty acid tails are flexible, causing the lipid bilayer to be fluid. This makes the cells flexible. At body temperature, membranes are a liquid with a consistency that is similar to cooking oil. Cholesterol In animals, cholesterol is a major membrane lipid. It may be equal in amount to phospholipids. It is similar to phospholipids in that it one end is hydrophilic, the other end is hydrophobic. Cholesterol makes the membrane less permeable to most biological molecules. Proteins Embedded in the Membrane Proteins are scattered throughout the membrane. They may be attached to inner surface, embedded in the bilayer, or attached to the outer surface. Hydrophilic (polar) regions of the protein project from the inner or outer surface. Hydrophobic (nonpolar) regions are embedded within the membrane. Membrane proteins are capable of lateral movement. Diffusion Diffusion is the movement of particles from an area of higher concentration to an area of lower concentration. The movement is due to collisions, which occur more frequently in areas of higher concentration. The dots on the diagram above represent molecules or ions. After a period of time, the particles becoming dispersed (below). Overall, the movement is from the area of initial high concentration to areas that have a lower concentration. Temperature and the Rate of Diffusion Larger particles move slower, due to their larger mass and may be influenced by numerous collisions with many nearby smaller particles. Smaller particles move faster. The overall energy of movement is proportional to the square root of the temperature. Hotter particles move faster because they have more energy. The rate of diffusion increases as temperature increases because the particles move faster. As temperature increases, the collisions among particles become more energetic, causing particles to move from areas of higher concentration to lower concentration at a faster rate. Membranes are Differentially Permeable The plasma membrane is differentially permeable because some particles can pass through, others cannot. It can control the extent to which certain substances pass through. Nonpolar molecules pass through cell membranes more readily than polar molecules because the center of the lipid bilayer (the fatty acid tails) is nonpolar and does not readily interact with polar molecules. The following substances can pass through the cell membrane: Nonpolar molecules (example: lipids) Small polar molecules such as water The following substances cannot pass through the cell membrane: Ions and charged molecules (example: salts dissolved in water) Large polar molecules (example: glucose) Macromolecules Osmosis Osmosis is the diffusion of water across a differentially permeable membrane (see "Diffusion" above). It occurs when a solute (example: salt, sugar, protein, etc.) cannot pass through a membrane but the solvent (water) can pass through. In areas where the solute concentration is high, the concentration of water molecules is low. In areas where the solute concentration is low, the concentration of water molecules is high. If there is no solute, the water is 100% water (high water concentration). Water moves areas where the concentration of water molecules is high (low solute concentration) to areas where the concentration of water molecules is low (high solute concentration). In general, water moves toward the area with a higher solute concentration because it has a lower water concentration. In the container on the left side of the diagram, water will enter the cell because it is more concentrated on the outside. In the center drawing, water is more concentrated inside the cell, so it will move out. If the solute concentration is the same inside as it is out, the amount of water that moves out will be approximately to the amount that moves in. Osmotic pressure is the force of osmosis. In the diagram above, the cell on the left will swell. The pressure within the cell is osmotic pressure. Tonicity Tonicity refers to the relative concentration of solute on either side of a membrane. Isotonic In an isotonic solution, the concentration of solute is the same on both sides of the membrane (inside the cell and outside). A cell placed in an isotonic solution neither gains or loses water. Most cells in the body are in an isotonic solution. Hypotonic A hypotonic solution is one that has less solute (more water). Cells in hypotonic solution tend to gain water. Animal cells can lyse (rupture) in a hypotonic solution due to the osmotic pressure. Freshwater organisms live in a hypotonic solution and have a tendency to gain water. The contractile vacuole in freshwater protozoans removes water that enters the cell. The cell wall of plant cells prevents the cell from rupturing. The osmotic pressure, called turgor pressure, helps support the cell. A cell in which the contents are under pressure is turgid. Hypertonic solution A hypertonic solution is one that has a high solute concentration. Cells in a hypertonic solution will lose water. The marine environment is a hypertonic solution for many organisms. They often have mechanisms to prevent dehydration or to replace lost water. Animal cells placed in a hypertonic solution will undergo crenation, a condition where the cell shrivels up as it loses water. Plant cells placed in a hypertonic solution will undergo plasmolysis, a condition where the plasma membrane pulls away from the cell wall as the cell shrinks. The cell wall is rigid and does not shrink. Left: These Elodea cells were placed in a 10% NaCl solution. The contents of the cells was reduced but the cell walls remained intact. Compare these cells to normal cells in the photograph below. Click on the image to view an enlargement. Left: Normal Elodea cells X 400 Click on the image to view an enlargement. Functions of Membrane Proteins Enzymes Some enzymes are embedded within membranes. Cell Identification Markers Lipids and proteins within the membrane may have a carbohydrate chain attached. These glycolipids and glycoproteins often function as cell identification markers, allowing cells to identify other cells. This is particularly important in the immune system where cells patrolling the body's tissues identify and destroy foreign invaders such as bacteria or viruses. Cell Adhesion - Junctions Proteins associated with the cell membranes of animal cells may bind to proteins of adjacent cells. These connections, called junctions may serve to bind cells together, to prevent the movement of material between the cells, or to allow cells to communicate with each other. Attachment of the Cytoskeleton Receptors Receptors enable cells to detect hormones and a variety of other chemicals in their environment. The binding of a molecule and a receptor initiates a chemical change within the cell. In the diagram above, the binding of hormone and receptor initiates the conversion of chemical A to chemical B. Hormones are molecules that cells use to communicate with one another. For example, cells in the pancreas produce the hormone insulin when glucose levels in the blood become elevated. The hormone travels within the blood to other parts of the body. It stimulates liver and muscle cells to begin removing the glucose and storing it as glycogen. Vesicle Trafficking Vesicles may follow microtubules to their destination. Proteins within the membrane of the vesicle recognize and attach to proteins in other membranes. This allows vesicles to attach to the membranes of other organelles such as the endoplasmic reticulum, golgi apparatus, or lysosomes. Transport of Materials Across Cell Membranes Facilitated Diffusion Facilitated diffusion involves the use of a protein to facilitate the movement of molecules across the membrane. In some cases, molecules pass through channels within the protein. In other cases, the protein changes shape, allowing molecules to pass through. As can be seen below, the protein changes shape and releases the molecule to the side of the membrane that has the lower concentration. Additional energy is not required because the molecule is traveling down a concentration gradient (high concentration to low concentration). The energy of movement comes from the concentration gradient. Active Transport Active transport is used to move ions or molecules against a concentration gradient (low concentration to high concentration). Active transport is like a water pump; it uses energy to pump water uphill where a siphon cannot. Facilitated diffusion (see above) is like a siphon in that additional energy is not required but it can only allow movement downhill. Movement against a concentration gradient requires energy. The energy is supplied by ATP which is released by breaking a phosphate bond to produce ADP: ATP ADP + Pi + energy Cells that use a lot of active transport have many mitochondria to produce the ATP needed. The Sodium-Potassium Pump The sodium-potassium pump uses active transport to move 3 sodium ions to the outside of the cell for each 2 potassium ions that it moves in. It is found in all human cells, especially nerve and muscle cells. One third of the body’s energy expenditure is used to operate the sodium-potassium pump. Mechanism of operation of the Sodium-Potassium Pump The diagrams below illustrate the mechanism of operation of the sodium-potassium pump. In these diagrams, orange is used to represent the pump protein. Circles are used to represent sodium ions and squares are used to represent potassium ions. Notice that the pump has three sodium binding sites and two potassium binding sites. Three sodium ions enter the pump. ATP bonds to the pump. One phosphate bond in the ATP molecule breaks, releasing its energy to the pump protein. The pump protein changes shape, releasing the sodium ions to the outside. The two potassium binding sites are also exposed to the outside, allowing two potassium ions to enter the pump. When the phosphate group detaches from the pump, the pump returns to its original shape. The two potassium ions leave and three sodium ions enter. The cycle then repeats itself. Examples of Active Transport Plants move minerals (inorganic ions) into their roots by active transport. The gills of marine fish have cells that can remove salt from the body by pumping it into the salt water. The thyroid gland cells bring in iodine for use in producing hormones. Cells in the vertebrate kidney reabsorb sodium ions from urine. Cotransport Active transport uses energy to pump materials across a membrane. A concentration gradient of ions or molecules therefore is a high-energy condition. The ions or molecules will attempt to move back across the membrane under pressure (osmotic pressure). This energy can be used to transport other molecules across the membrane. In the diagram below, energy from ATP is used to produce a concentration gradient of H+. Sucrose can be pumped into cells where the concentration of sucrose is already high by using the energy of a high concentration of hydrogen ions on the outside of a cell. Active transport pumps the hydrogen ions out and certain proteins in the cell membrane allow the hydrogen ions to reenter the cell. As the hydrogen ions force their way through this protein, the energy of reentry is used to pump sucrose into the cell. Endocytosis and Exocytosis These processes are used for materials that are too big to pass through the plasma membrane via protein transport. Endocytosis The process by which a cell engulfs material to bring it into the cell is called endocytosis. Two major forms of endocytosis described below. Phagocytosis Phagocytosis refers to the process of engulfing large particles. A vacuole is formed that contains the material that has been engulfed. Pinocytosis Pinocytosis refers to engulfing macromolecules. As in phagocytosis, a vesicle is formed which contains the molecules that were brought into the cell. Vacuoles and vesicles produced by phagocytosis and pinocytosis can fuse with lysosomes (lysosomes are vesicles that contain digestive enzymes). Phagocytosis and pinocytosis remove membrane from cell surface to form vacuoles that contain the engulfed material. Receptor-Mediated endocytosis Macromolecules bind to receptors on the surface of the cell. Receptors with bound macromolecules aggregate in one area and are brought into the cell by endocytosis. The vesicle containing the macromolecules can release the macromolecules into the cell directly or they can be processed by chemicals contained within lysosomes after fusing with the lysosomes. The vesicle (and receptors) then returns to the cell surface. Example: Hypercholesterolemia Cholesterol is carried by LDL (low-density lipoprotein), which binds to LDL receptors on the cell surface. Normally cholesterol (and LDL) is brought into the cell by receptor-mediated endocytosis as described above. A faulty gene for the LDL receptor results in LDL not binding to the cells. The Cholesterol remains in the blood and becomes deposited on arteries. Reduced blood flow in arteries that supply the heart causes heart attacks in patients as early as 6 years. Exocytosis Exocytosis moves material to the outside. A vesicle fuses with the plasma membrane and discharges its contents outside. This allows cells to secrete molecules. The fusion of vesicles to the plasma membrane adds membrane to the cell surface. CONCEPT 6 – REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes, returning the changing condition back to its target set point. b. Positive feedback mechanisms amplify responses and processes in biological organisms. The condition initiating the response is moved farther away from the initial set-point. Amplification occurs when the stimulus is further activated which, in turn, initiates an additional response that produces system change. 2. Cell-to-cell communication a. Cells receive or send inhibitory or stimulatory signals from other cells, organisms or the environment. b. In single-celled organisms it is response to its environment. c. In multicellular organisms, signal transduction pathways coordinate the activities within individual cells. Ex. Epinephrine stimulation of glycogen breakdown in mammals d. Cells communicate by cell-to-cell contact. Ex Immune cells interact by cell-cell contact, antigenpresenting cells (APCs), helper T-cells and killer T cells or plasmodesmata between plant cells that allow material to be transported from cell to cell. e. Cells communicate over short distances by using local regulators that target cells in the vicinity of the emitting cell. Ex. Neurotransmitters, plant immune response f. Signals released by one cell type can travel long distances to target cells of another cell type. Ex. Hormones g. A receptor protein recognizes signal molecules, causing the receptor protein’s shape to change, which initiates transduction of the signal. Ex. G-protein linked receptors, ligand-gated ion channels, tyrosine kinase receptors. h. Signal transduction is the process by which a signal is converted to a cellular response. Signaling cascades relay signals from receptors to cell targets, often amplifying the incoming signals, with the result of appropriate responses by the cell. i. Second messengers inside of cells are often essential to the function of the cascade. j. Many signal transduction pathways include: Protein modifications or phosphorylation cascades in which a series of protein kinases add a phosphate group to the next protein in the cascade sequence. 3. Gene Regulation a. Prokaryotes (1) Inducers (turn genes on) and repressors (turn genes off) are small molecules that interact with regulatory proteins and/or regulatory sequences. (2) Regulatory proteins inhibit gene expression by binding to DNA and blocking transcription (negative control). (3) Regulatory proteins stimulate gene expression by binding to DNA and stimulating transcription (positive control) or binding to repressors to inactivate repressor function. b. Eukaryotes (1) Transcription factors bind to DNA sequences and other regulatory proteins (2) Some of these transcription factors are activators (increase expression), while others are repressors (decrease expression). (3) The combination of transcription factors binding to the regulatory regions at any one time determines how much, if any, of the gene product will be produced. 4. Immunity a. Plants, invertebrates and vertebrates have multiple, nonspecific immune responses, ex: phagocytes engulf and digest pathogens with the help of lysosomes b. Mammals use specific immune responses triggered by natural or artificial agents that disrupt dynamic homeostasis. (1) The mammalian immune system includes two types of specific responses: cell mediated and humoral. (2) In the cell-mediated response, cytotoxic T cells, a type of lymphocytic white blood cell, target‖intracellular pathogens when antigens are displayed on the outside of the cells. (3) In the humoral response, B cells, a type of lymphocytic white blood cell, produce antibodies against specific antigens. (4) Antigens are recognized by antibodies to the antigen. (5) Antibodies are proteins produced by B cells, and each antibody is specific to a particular antigen. (6) A second exposure to an antigen results in a more rapid and enhanced immune response. 5. Viruses a. Replication (1) Viruses inject DNA or RNA into host cell (2) Viruses have highly efficient replicative capabilities that allow for rapid evolution (3) Viruses replicate via the lytic cycle, allowing one virus to produce many progeny simultaneously (4) Virus replication allows for mutations to occur through usual host pathways. (5) RNA viruses lack replication error-checking mechanisms, and thus have higher rates of mutation. (6) Related viruses can combine/recombine information if they infect the same host cell. (7) Some viruses are able to integrate into the host DNA and establish a latent (lysogenic) infection (8) HIV is a well-studied system where the rapid evolution of a virus within the host contributes to the pathogenicity of viral infection. (9) Genetic information in retroviruses is a special case and has an alternate flow of information: from RNA to DNA, made possible by reverse transcriptase, an enzyme that copies the viral RNA genome into DNA. This DNA integrates into the host genome and becomes transcribed and translated for the assembly of new viral progeny. Vocabulary Antibody Antigen B-cell Cell-mediated immunity communication cyclic AMP (cAMP) cytotoxic T-cell G-protein linked receptor Helper T-cell Hormone Humoral immunity Inducer Lytic cycle Lysogenic cycle Negative feedback Operon Operator Phagocyte phagocytosis phosphorylation cascade positive feedback protein kinase receptor Thinking Practice 1. Refer to the diagram at the right to respond to the following questions. a. Is the hormone hydrophobic or hydrophilic? How do you know? repressor retrovirus reverse transcriptase second messenger signal cascade signal transduction signal transduction pathway transcription factor virus white blood cell b. Explain how the action of the hormone might be different if it could move through the cell membrane. c. Explain what is happening in this picture and make a prediction about what will be the end result in the cell to which this hormone has bound. 2. Lactose digestion in E. coli begins with its hydrolysis by the enzyme b-galactosidase. The gene encoding bgalactosidase, lacZ, is part of a coordinately regulated operon containing other genes required for lactose utilization. Use the legend below to draw the gene and its interaction with RNA polymerase, the repressor protein, and lactose when lactose is being digested. 3. Based on the information provided, propose a possible mechanism for a drug to resist HIV infection. 4. Describe the processes occurring at each of the numbered positions (I, II, III, and IV) in the diagram to the right. 5. Refer to the images at the right to answer the following: a. Which immune response in shown: cell mediated or humoral? Explain how you know. b. What are the “Y” shaped molecules called? What is their role in the immune response? c. Describe how the “Y” shaped molecules relate to the graph displayed. 6. One student described an action potential in a neuron by saying “As more gates open the concentration of sodium inside the cell increases and this causes even more gates to open.” Is this an example of a positive or negative feedback loop? Justify your reasoning. 7. The figure to the right shows the feedback mechanism for regulating blood glucose. a. Is this a positive or negative feedback loop? Explain your answer. b. Individuals that suffer from Type I diabetes do not have functional insulin-producing cells. Describe how their blood will differ from that of a healthy individual after a glucose-rich meal. 8. In a molecular biology laboratory, a student obtained competent E. coli cells and used a common transformation procedure to induce the uptake of plasmid DNA with a gene for resistance to the antibiotic kanamycin. The results below were obtained. a. What is the purpose of Plate IV? b. Explain the growth you see and the type of bacteria (transformed vs. non-transformed or both) that would be on Plate 1. c. Explain the growth you see and the type of bacteria (transformed vs. non-transformed or both) that would be on Plate II. d. If the student repeated the experiment, but the heat shock was unsuccessful and the plasmid was unable to be transformed, for which plates would growth be expected? Explain your answer.