Experiment 3
advertisement

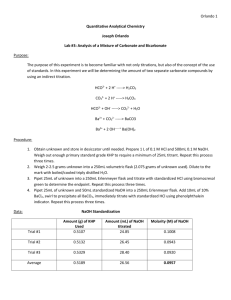
Experiment 3: Analysis of Carbonate and Bicarbonate Objective: The objective of this experiment is to determine the amount of carbonate and bicarbonate in an unknown sample using direct and indirect titration techniques. The acid-base titrations will involve the use of primary and secondary standards. The percentages of carbonate and bicarbonate in the unknown will be analyzed using statistical analysis. Background: Acid-base titrations involve an acid or base titrant with a known concentration to measure the concentration of an unknown solution. When the titration reaches the equivalence point there are stoichiometrically equivalent amounts of titrant and analyte. Direct titrations involve adding the titrant directly to the analyte until the equivalence point is reached. Indirect titrations involve adding a standardized reagent to the analyte and the added reagent is titrated to the equivalence point. Procedure: Unknowns A, B, and C were obtained and stored in the desiccator. HCl (1 L, 0.1 M) was prepared by diluting HCl (17.00 mL, 6 M) with distilled water. NaOH was prepared by dissolving solid NaOH (4.00 g) in distilled water. KHP (0.500 g) was used to standardize the NaOH. The standardized NaOH was used to standardize the HCl solution. An Unknown A solution (250 mL) was made by dissolving Unknown A (2.0000 g) in carbon dioxide free water. The Unknown A solution (25.00 mL) was pipetted in a 250 mL Erlenmeyer flask and titrated with HCl and bromocresol green indicator. Unknown A (25.00 mL) and standardized NaOH (50.00 mL) were pipetted into an Erlenmeyer flask. Barium Chloride (10 mL, 10% wt/v) was added to the flask and immediately titrated with HCl and phenolphthalein indicator. The aforementioned method was used to analyze Unknowns B and C. Data: Chemical equations 𝐻𝐶𝑂3 − (𝑎𝑞) + 𝐻𝐶𝑙(𝑎𝑞) → 𝐻2 𝐶𝑂3 (𝑎𝑞) → 𝐶𝑙 − Total Alkalinity (𝑎𝑞) + 𝐻2 𝑂(𝑙) + 𝐶𝑂2 (𝑔) 𝐶𝑂32− (𝑎𝑞) + 2 𝐻𝐶𝑙(𝑎𝑞) → 𝐻2 𝐶𝑂3 (𝑎𝑞) → 2 𝐶𝑙 − (𝑎𝑞) + 𝐻2 𝑂(𝑙) + 𝐶𝑂2 (𝑔) Bicarbonate conversion to Carbonate 𝐻𝐶𝑂3 − (𝑎𝑞) + 𝑁𝑎𝑂𝐻(𝑎𝑞) → 𝑁𝑎𝐶𝑂3 − (𝑎𝑞) + 𝐻2 𝑂(𝑙) Carbonate Precipitation 𝐵𝑎𝐶𝑙2 (𝑎𝑞) + 𝐶𝑂32− (𝑎𝑞) → 𝐵𝑎𝐶𝑂3 (𝑠) + 2 𝐶𝑙 − (𝑎𝑞) Table 1: Reaction equations for the carbonate and bicarbonate analysis Mass of KHP Trial 1 Trial 2 Trial 3 Mass KHP Standardization 1 (g) 0.5100 0.5098 0.5097 Mass KHP Standardization 2 (g) 0.5015 0.5008 0.5012 Table 2: Mass of KHP used in NaOH standardizations Mass of Unknown Samples Unknown A Unknown B Unknown C 2.0094 g 2.0010 g 2.0038 g Table 3: Mass of Unknowns A, B, and C Standardization of NaOH Standardization 1 Initial Volume NaOH (mL) Trial 1 Trial 2 Trial 3 0.72 0.22 0.89 Final Volume NaOH (mL) 26.92 26.12 26.82 Table 4: Titration data for NaOH standardization Standardization 2 Volume NaOH used (mL) 26.20 25.90 25.93 Initial Volume NaOH (mL) 0.00 0.00 0.00 Final Volume NaOH (mL) 26.80 26.75 26.85 Volume NaOH used (mL) 26.80 26.75 26.85 Standardization of HCl Standardization 1 Initial Volume NaOH (mL) Trial 1 Trial 2 Trial 3 Final Volume NaOH (mL) 0.01 10.01 0.11 Standardization 2 Volume NaOH used (mL) 10.01 20.02 10.13 Initial Volume NaOH (mL) 10.00 10.01 10.02 Final Volume NaOH (mL) 0.00 10.45 20.92 Volume NaOH used (mL) 10.45 20.92 31.35 10.45 10.47 10.43 Table 5: Titration data for HCl standardization Total Alkalinity Titration Unknown A Trial 1 Trial 2 Trial 3 Unknown B Unknown C Initial Volume HCl (mL) Final Volume HCl (mL) Volume HCl used (mL) Initial Volume HCl (mL) Final Volume HCl (mL) Volume HCl used (mL) 0.31 0.21 0.67 32.51 32.13 32.71 32.20 31.92 32.04 0.11 0.10 0.22 29.37 29.31 29.50 29.26 29.21 29.28 Initial Final Volume Volume Volume HCl HCl HCl used (mL) (mL) (mL) 0.32 0.69 0.21 26.79 27.11 26.67 26.47 26.42 26.46 Table 6: Carbonate/Bicarbonate titration with HCl Moles of Excess NaOH Unknown A Trial 1 Trial 2 Trial 3 Unknown B Unknown C Initial Volume HCl (mL) Final Volume HCl (mL) Volume HCl used (mL) Initial Volume HCl (mL) Final Volume HCl (mL) Volume HCl used (mL) 0.00 0.00 0.00 42.78 42.80 42.82 42.78 42.80 42.82 0.30 0.00 0.00 37.4 37.15 37.12 37.10 37.15 37.12 Table 7: Indirect titration of NaOH with HCl Initial Final Volume Volume Volume HCl HCl HCl used (mL) (mL) (mL) 0.00 0.00 0.00 26.95 27.00 26.90 26.95 27.00 26.90 Calculations: Mass of NaOH Needed for Solution Equation 0.1 𝑀 𝑁𝑎𝑂𝐻 39.9971 𝑔 𝑁𝑎𝑂𝐻 1𝐿× × = 3.99971 𝑔 𝑁𝑎𝑂𝐻 𝐿 1 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 Calculation 1: Mass of NaOH needed to make 1 L of a 0.1 M solution Volume of HCl Needed for Solution Equation 𝑀1 𝑉1 = 𝑀2 𝑉2 Example 0.1 𝑀 × 1 𝐿 = 6 𝑀 × 𝑉2 𝑉2 = 16.667 𝑚𝐿 Calculation 2: Volume of 6 M HCl needed for 0.1 M dilution Mass of KHP Needed for Titration Equation 0.1 𝑀 𝑁𝑎𝑂𝐻 1 𝑚𝑜𝑙 𝐾𝐻𝑃 204.22 𝑔 𝐾𝐻𝑃 0.025 𝐿 × × × = 0.5100 𝑔 𝐾𝐻𝑃 𝐿 1 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 1 𝑚𝑜𝑙 𝐾𝐻𝑃 Calculation 3: Mass of KHP needed for 25.00 mL titration Moles of NaOH Equation 𝑔 𝐾𝐻𝑃 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 × = 𝑚𝑜𝑙𝑒𝑠 𝑁𝑎𝑂𝐻 𝑀𝑊 𝐾𝐻𝑃 𝑚𝑜𝑙 𝐾𝐻𝑃 Example Trial 1 0.5100 𝑔 𝐾𝐻𝑃 1 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 × 204.22 𝑔 𝐾𝐻𝑃 1 𝑚𝑜𝑙 𝐾𝐻𝑃 = 0.00249731 𝑚𝑜𝑙𝑒𝑠 𝑁𝑎𝑂𝐻 Calculation 4: Moles of NaOH calculation with Standardization 1 Trial 1 data Molarity of NaOH Equation 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 = 𝑀 𝑁𝑎𝑂𝐻 𝑉 Example Trial 1 0.00249731 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 = 0.09532 𝑀 𝑁𝑎𝑂𝐻 0.02620 𝐿 Calculation 5: Molarity of NaOH calculation with Standardization 1 Trial 1 data Moles of HCl Equation 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 𝑚𝑜𝑙 𝐻𝐶𝑙 𝑉 𝑁𝑎𝑂𝐻 × × 𝐿 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 = 𝑚𝑜𝑙 𝐻𝐶𝑙 Example Trial 1 0.09598 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 1 𝑚𝑜𝑙 𝐻𝐶𝑙 0.0100 𝐿 𝑁𝑎𝑂𝐻 × × 𝐿 1 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 = 0.000959844 𝑚𝑜𝑙 𝐻𝐶𝑙 Calculation 6: Moles of HCl calculation with Standardization 1 Trial 1 data Molarity of HCl Equation 𝑚𝑜𝑙 𝐻𝐶𝑙 = 𝑀 𝐻𝐶𝑙 𝑉 Example Trial 1 0.000959844 𝑚𝑜𝑙 𝐻𝐶𝑙 = 0.09608 𝑀 𝐻𝐶𝑙 0.010 𝐿 Calculation 7: Molarity of HCl calculation with Standardization 1 Trial 1 data Total Alkalinity Equation 𝑚𝑜𝑙 𝐻𝐶𝑙 𝑚𝑜𝑙 𝐶𝑂3 2− /𝐻𝐶𝑂3 − 𝑉 𝐻𝐶𝑙 × × 𝐿 𝑚𝑜𝑙 𝐻𝐶𝑙 = 𝑚𝑜𝑙 𝐶𝑂3 2− /𝐻𝐶𝑂3 − Unknown A Example Trial 1 0.09608 𝑚𝑜𝑙 𝐻𝐶𝑙 1 𝑚𝑜𝑙𝐶𝑂3 2− /𝐻𝐶𝑂3 − 0.03220 𝐿 𝐻𝐶𝑙 × × 𝐿 1 𝑚𝑜𝑙 𝐻𝐶𝑙 = 0.003093776 𝑚𝑜𝑙 𝐶𝑂3 2− /𝐻𝐶𝑂3 − Calculation 8: Total alkalinity calculation of Unknown A Moles of NaOH to react with Bicarbonate Equation 𝑉 × 𝑀 = 𝑚𝑜𝑙𝑒𝑠 𝑜𝑓 𝑁𝑎𝑂𝐻 Unknown A Example Trial 1 0.050 𝐿 𝑁𝑎𝑂𝐻 × 0.09598 𝑀 = 0.004799 𝑚𝑜𝑙𝑒𝑠 𝑜𝑓 𝑁𝑎𝑂𝐻 Calculation 9: Total moles of NaOH to react with Bicarbonate before the titration. Unknown C used Standardization 2 data. Moles of NaOH in Excess Equation 1 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 𝑉×𝑀× = 𝑚𝑜𝑙𝑒𝑠 𝑜𝑓 𝑁𝑎𝑂𝐻 1 𝑚𝑜𝑙 𝐻𝐶𝑙 Unknown A Example Trial 1 1 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 0.04278 𝐿 𝐻𝐶𝑙 × 0.09608𝑀 𝐻𝐶𝑙 × 1 𝑚𝑜𝑙 𝐻𝐶𝑙 = 0.0041103 𝑚𝑜𝑙𝑒𝑠 𝑜𝑓 𝑁𝑎𝑂𝐻 Calculation 10: Moles of NaOH in excess. Unkown C used standardization 2 data Moles of Bicarbonate in Titration Equation 𝑇𝑜𝑡𝑎𝑙 𝑀𝑜𝑙𝑒𝑠 𝑁𝑎𝑂𝐻 − 𝑀𝑜𝑙𝑒𝑠 𝑁𝑎𝑂𝐻 𝑖𝑛 𝐸𝑥𝑐𝑒𝑠𝑠 = 𝑚𝑜𝑙 𝐻𝐶𝑂3 − Unknown A Example 0.004799 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 − 0.0041103 𝑁𝑎𝑂𝐻 𝑖𝑛 𝐸𝑥𝑐𝑒𝑠𝑠 = 0.0006887 𝑚𝑜𝑙 𝐻𝐶𝑂3 − Calculation 11: Moles of bicarbonate reacted with NaOH Moles of Bicarbonate in Unknown Solution Equation 𝑚𝑜𝑙 𝐻𝐶𝑂3 − × 𝑉𝑓𝑙𝑎𝑠𝑘 = 𝑀 𝐻𝐶𝑂3 − 𝑉𝑝𝑖𝑝𝑒𝑡 Calculation 12: Moles of bicarbonate in Unknown A solution Unknown A Example 0.0006887 𝑚𝑜𝑙 𝐻𝐶𝑂3 − × 0.250 0.025 𝐿 = 0.006887 𝑀 𝐻𝐶𝑂3 − Mass of Bicarbonate in Unknown A Equation 𝑀𝑊 𝐻𝐶𝑂3 − − 𝑚𝑜𝑙 𝐻𝐶𝑂3 × = 𝑚𝑎𝑠𝑠 𝐻𝐶𝑂3 − 𝑚𝑜𝑙 𝐻𝐶𝑂3 − Unknown A Example 84.0066 𝑔 𝐻𝐶𝑂3 − − 0.006687 𝑚𝑜𝑙 𝐻𝐶𝑂3 × 1 𝑚𝑜𝑙 𝐻𝐶𝑂3 − = 0.5786 𝑔 𝐻𝐶𝑂3 − Calculation 13: Mass of bicarbonate in Unknown A Mass Percent of Bicarbonate in Unknown A Equation 𝑚𝑎𝑠𝑠 𝐻𝐶𝑂3 −− × 100 = % 𝐻𝐶𝑂3 − 𝑔 𝑠𝑎𝑚𝑝𝑙𝑒 Unknown A Example 0.5786 𝑔 𝐻𝐶𝑂3 − × 100 = 28.91 % 𝐻𝐶𝑂3 − 2.0015 𝑔 𝑠𝑎𝑚𝑝𝑙𝑒 Calculation 14: Mass percent of bicarbonate in Unknown A Moles of Carbonate in Titration 𝑚𝑜𝑙 𝐶𝑂3 2− /𝐻𝐶𝑂3 2 − Equation − 𝑚𝑜𝑙 𝐻𝐶𝑂3 − = 𝑚𝑜𝑙 𝐶𝑂3 2− Unknown A Example 𝐶𝑂3 2− − 0.00307968 𝑚𝑜𝑙 𝐻𝐶𝑂 − − 0.0006887𝑚𝑜𝑙 𝐻𝐶𝑂3 3 2 = 0.0011955 𝑚𝑜𝑙 𝐶𝑂3 2− Calculation 15: Moles of carbonate in Unknown A titration Moles of Carbonate in Unknown Solution Equation 𝑚𝑜𝑙 𝐶𝑂3 2− × 𝑉𝑓𝑙𝑎𝑠𝑘 = 𝑀 𝐶𝑂3 2− 𝑉𝑝𝑖𝑝𝑒𝑡 Unknown A Example 0.0011955 𝑚𝑜𝑙 𝐻𝐶𝑂3 − × 0.250 0.025 𝐿 = 0.0119549 𝑀 𝐶𝑂3 2− Calculation 16: Moles of carbonate in Unknown A solution Mass of Carbonate in Unknown A 𝑚𝑜𝑙 𝐶𝑂3 2− × Equation 𝑀𝑊 𝐶𝑂3 2− 𝑚𝑜𝑙 𝐶𝑂3 2− = 𝑚𝑎𝑠𝑠 𝐶𝑂3 2− Unknown A Example 105.988 𝑔 𝐶𝑂3 2− 0.0119549 𝑚𝑜𝑙 𝐶𝑂3 2− × 1 𝑚𝑜𝑙 𝐶𝑂3 2− = 1.2671 𝑔 𝐶𝑂3 2− Calculation 17: Mass of carbonate in Unknown A Mass Percent of Carbonate in Unknown A Equation 2− − 𝑚𝑎𝑠𝑠 𝐶𝑂3 𝑔 𝑠𝑎𝑚𝑝𝑙𝑒 × 100 = % 𝐶𝑂3 2− Calculation 18: Mass percent of carbonate in Unknown A Unknown A Example − 1.2671 𝑔 𝐶𝑂3 2− × 100 = 63.31 % 𝐶𝑂3 2− 2.0015 𝑔 𝑠𝑎𝑚𝑝𝑙𝑒 Mean (x̅) Example NaOH Molarity Equation x 0.09532 M 0.09638 M 0.09625 M 3 x 0.09595 M x 1 x 2 x 3 ... x n n x Calculation 19: Mean calculation Standard Deviation (S) Equation n S (x i 1 1 x) 2 n 1 Example EDTA Molarity S=√ (0.09532 M-0.09598 M)2 + (0.09638 M-0.09598 M)2 + (0.09625 M-0.09598 M)2 2 S = 4.749×10−4 Calculation 20: Standard deviation calculation Results: NaOH Standardization Average NaOH Molarity Standardization1 0.09598 M±0.0004749 Average NaOH Molarity Standardization 2 0.09157 M±0.0001177 Table 8: Average molarity for NaOH standardization HCl Standardization Average HCl Molarity Standardization1 0.09608 M±0.00010 Average HCl Molarity Standardization 2 0.10030 M±0.00019 Table 9: Average molarity for HCl standardization Total Alkalinity Unknown A Unknown B Unknown C Average Moles of CO32-/HCO30.00308 mol ±1.102E-5 0.00281 mol ±2.829E-6 0.00265 mol ±2.167E-6 Table 10: Average moles of carbonate and bicarbonate from HCl titration. Mass Percent of Carbonate and Bicarbonate in Unknown Sample Unknown A Unknown B Unknown C Average Mass % HCO328.91% 50.42% 78.61% Average Mass % CO363.31% 42.58% 20.57% Table 11: Mass percent of carbonate and bicarbonate in unknown samples Discussion: Primary standards like KHP are extremely pure (≥99.9%) can be weighed and directly used to standardize the titrant. Secondary standards like the NaOH and HCl solutions are prepared in the laboratory and standardized against the primary standard. A titrant is a solution used to react with the analyte during the titration. The NaOH is the titrant in the HCl standardization. HCl is the titrant in the unknown titrations. Indirect titrations or back titrations involve the excess reagent being titrated after the reaction with the analyte. In this experiment the excess NaOH is determined after the carbonate is precipitated with barium chloride. The percentage of bicarbonate and carbonate found in Unknown A was 28.91% and 63.31%, respectively. There is a discrepancy in the data, the percentages should add up to 100%; however, the calculated percentage adds up to 92.22%. In Unknown B, the experimental percentage of bicarbonate and carbonate were 50.42% and 42.58% respectively. Again there is a discrepancy in the data; the total percentage is 93%. Unknown C has experimental bicarbonate and carbonate percentages of 78.61% and 20.57% respectively (Table 11), and a total percentage of 99.18%. Unknown C yielded the most accurate results because the overall percentage is closer to 100%. Conclusion: The indirect titration to determine the excess NaOH is essential to the experiment because you cannot determine the individual amount of sodium bicarbonate using a direct titration. In the future a more distinctive indicator would be helpful to determine the end point of the indirect titration. The phenolphthalein indicator used for the indirect titration has a color change from bright pink to a translucent light pink. Determining the actual end point of the titration was very ambiguous. Overall, the mass percentage of sodium bicarbonate and sodium carbonate were able to be determined for all three unknown samples.