High Risk Medications
advertisement

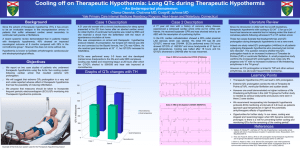
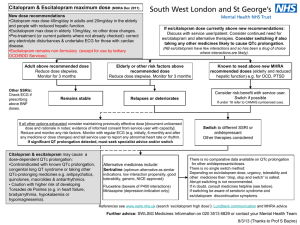
Regions Guidelines for Managing Medications and QTc prolongation Background: Prolongation of QTc interval is linked with the development of potentially lifethreatening ventricular arrhythmias such as torsades de pointes (TDP). Per European Committee for Proprietary Medicinal Products, the following are the ranges for normal, borderline and prolonged QTc intervals (after correction with Bazett’s formula). QTc Prolongation (msec) 1-15 years Men Female <440 < 430 <450 Borderline 440-460 430-450 450-470 Abnormal >460 > 450 > 470 Normal QTc is considered prolonged if the values are greater than 450 msec in males and greater than 470 msec in females. The clinical importance of QTc interval prolongation has prompted FDA to implement rigorous guidelines for evaluating the proarrhythmic potential of new drugs during the approval process. Based on FDA’s Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs document, an absolute QTc > 500 msec or a relative increase in the QTc interval of ≥ 60 msec above baseline during drug administration has been a threshold of particular concern. Medications with risk of prolonging the QT interval were identified and stratified according to a risk category based on FDA-approved product labeling and supporting literature. High risk category constitutes medications that have a known potential to prolong QT interval or carry a black box warning for potential to prolong QT interval and cause TDP. Medium risk medications have potential for QT prolongation in precautions/warnings section of product labeling or have been associated with welldefined cases of TDP reported in literature. Medications classified as low risk are associated with minimal QTc prolongation. 1 Antiarrhythmics: Phenothiazines: chlorpromazine fluphenazine mesoridazine perphenazine trifluoperazine Atypicals clozapine paliperidone quetiapine risperidone ziprasidone Others: haloperidol venlafaxine pimozide Psychotropics: TCAs amiodarone disopyramide dofetilide ibutilide procainamide quinidine sotalol Psychotropics: SSRIs: High Risk Medications Miscellaneous: citalopram fluoxetine paroxetine sertraline amitriptyline clomipramine desipramine doxepin imipramine nortriptyline protriptyline trimipramine arsenic cisapride droperidol thioridazine pentamidine Medium Risk Medications Antimicrobials: Tyrosine kinase inhibitors: Quinolones moxifloxacin dasatinib levofloxacin lapatinib ofloxacin nilotinib sunitinib Azoles: voriconazole Antimalarials: chloroquine halofantrine mefloquine Miscellaneous: alfuzosin flecainide fosphenytoin indapamide methadone ranolazine tacrolimus vardenafil Other Antibiotics: clarithromycin erythromycin telithromycin Low Risk Medications Antimicrobials: Antiemetics: Azoles: fluconazole itraconazole ketoconazole Miscellaneous: atazanavir azithromycin ciprofloxacin SMX/TMP 2 dolasetron palonosetron ondansetron granisetron Miscellaneous: mexiletine propafenone solifenacin Guidelines for QTc Prolongation Medication Monitoring A. Monotherapy with any “High-Risk” Medication 1. Chronic stable patients: Obtain 12 lead ECG for patients chronically being administered “High-Risk” QTc prolonging medication if ECG is not already available within last 30 days. (see attached list for “high risk meds) Monitor serum drug levels when appropriate Consult pharmacy to adjust medication doses based on renal/hepatic function if necessary 2. Initiation of Intravenous “High-Risk” drugs: Obtain 12 lead ECG prior to initiation of “high- risk” QTc prolonging medications (see attached list) Patient is required to have continuous telemetry monitoring during continuous intravenous infusion of “High-Risk” QTc prolonging medications Document QTc every 8 hours while patient is receiving therapy Monitor serum drug levels when appropriate Consult pharmacy to adjust medication doses based on renal/hepatic function if necessary 3. Initiation of PO scheduled “High-Risk” drugs: Obtain baseline 12-lead ECG Continuous telemetry or remote telemetry monitoring for the first 24 hours Document QTc every 8 hours for first 24 hours Then if QTc stable, 12 lead ECG daily to assess QTc for the next 48 hours, followed by QTc once daily q72hours (or until steady state levels achieved) Monitor serum drug levels when appropriate Consult pharmacy to adjust medication doses based on renal/hepatic function if necessary 4. For high risk PRN drug, e.g. droperidol: Obtain baseline 12-lead ECG Continuous telemetry or remote telemetry for first 24 hours Monitor QTc 3 hours after each dose for first 24 hours If continuing drug, monitor QTc once daily for next 48 hours (or until steady state levels achieved) See above #3 for further guidance as appropriate **Administer with caution the following PO medication in patients demonstrating a creatinine clearance <50 ml/min e.g. disopyramide, dofetilide, procainamide, quinidine, sotalol, droperidol, pentamidine** B. Concurrent therapy of 2 or more agents that can prolong QTc interval 1. For all patients, pharmacist will verbally suggest monitoring QTc interval (see part D) with prescriber in the following circumstances: If 2 or more high risk meds are prescribed concurrently If any high risk med is prescribed concurrently with any medium risk med 2. Pharmacist will at minimum leave a Pharmacist to Prescriber communication note under “Comments” in Epic to suggest monitoring QTc interval (see part D) in the following circumstances*: If 2 or more medium risk meds are prescribed concurrently If any high risk med is prescribed concurrently with any low risk med If any medium risk med is prescribed concurrently with any low risk med If 2 or more low risk meds are prescribed *call prescriber if unsure 3. Pharmacists are to use clinical judgment when suggesting either decreasing the dose, or to discontinue QTc prolonging drug and suggest alternative therapeutic options, e.g. alternative nonQTc prolonging antibiotics, when QTc is > 500 msec or if there is an increase of ≥ 60 msec over baseline. Pharmacists are to document interventions as a progress note. 3 C. Clinical Factors to Consider When Handling QTc Prolongation Drug Interactions 1. Does the patient have a history of cardiovascular disease, congenital long QT syndrome or bradycardia or arrhythmias? 2. Is the patient currently monitored on telemetry or does the patient have regular EKG readings? 3. Is the baseline QTc interval prolonged (> 500 msec) or does the patient have a history of QTc prolongation? 4. Are the interacting medications prescribed in high doses? 5. Is the patient hypokalemic or hypomagnesemic? 6. Is this a critically ill patient? 7. Does the patient have any liver dysfunction? **Answering yes to any of the above questions should prompt pharmacist to verbally discuss and suggest QTc monitoring instead of leaving a communication note in Epic. D. Monitoring of patients on multiple medications that may prolong the QTc interval: 1. Recommend MD to place patient on telemetry unit or remote telemetry with specifications for RN to monitor and document QTc for at least 24 hours 2. At minimum check QTc at baseline, then recheck subsequent QTc interval when drug regimen (accounting for dose changes or interacting drugs) reaches steady state levels 3. Make sure magnesium and potassium levels are adequately replaced 4 References: 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 2003 United Hospital, St Paul, MN QTc prolongation Guidelines www.torsades. Org accessed 4/2009 www.cmezone.com. “Drug Interactions and Drug-Induced QT Interval Prolongation: The Role of Pharmacists”. Accessed 1/2008 Cubeddu LX. QT Prolongation and Fatal Arrhythmias: A Review of Clinical Implications and Effects of Drugs. American Journal of Therapeutics 2003,10: 452-457 Gowda RM, et al. Torsade de pointes: the clinical considerations. International Journal of Cardiology 2004, 96: 1-6 Owens Jr RC, et al. Antimicrobial-Associated QT Interval Prolongation: Pointes of Interest. Clinical Infectious Diseases 2006; 43: 1603-1611 Al-Khatib SM, et al. What Clinicians Should Know About the QT Interval. JAMA 2003, 289 (16): 2120-2127 Yap YG, et al. Drug-Induced QT Prolongation and Torsades De Pointes. Heart 2003, 89: 1363-1372 Haddad PM, et al. Antipsychotic-Related QTc Prolongation, Torsades de Pointes and Sudden Death. Drugs 2002, 62 (11): 1649 Glassman AH, et al. Antipsychotic Drugs: Prolonged QTc Interval, Torsades de Pointes, and Sudden Death. Am J Psychiatry 2001, 158:1774-1782 Kao LW, et al. Drug-Induced Q-T Prolongation. Med Clin N Am 2005, 89: 11251144 Ng TMH, et al. Pharmacist Monitoring of QTc Interval-Prolonging Medications in Critically Ill Medical Patients: A Pilot Study. Annals of Pharmacotherapy 2008, 42: 475-481 Zemrak WR, et al. Association of Antipsychotic and Antidepressant Drugs with Q-T Interval Prolongation. American Journal of Health-System Pharmacy 2008, 65(11): 1029-1038 co-reviewed by Pam Triplett, RN-Cardiac Medicine Decentralized Nurse Educator; Helen Sullinger; Uyen Dinh, PharmD; Hollie Lawrence, PharmD 5