File
advertisement

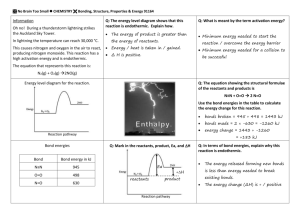
Endothermic and Exothermic Reactions Breaking bonds requires energy to be absorbed. When bonds form, energy is released. Chemical reactions must absorb energy to break the bonds of the reactants and release energy when the new bonds of the products form. Endothermic Reactions: Are chemical changes that ____________________ more energy than is released. Require a constant source of energy e.g. baking a cake Take the energy they absorb from the surrounding environment (air or water) The temperature of the surroundings _______________________ A + B + 500 kJ C Exothermic Reactions: Are chemical changes that ___________________ more energy than is absorbed. Only need energy to start Continue on their own using the energy they generate themselves No additional energy is required once the reaction begins Can be used as a source of energy to start other reactions The energy released is absorbed by the surrounding environment (air, water, food) The temperature of the surroundings _______________________ A + B C + 500 kJ Bond Energy: Is the same for breaking a bond or forming it Positive for breaking a bond/Negative for forming it Depends on whether the bond is single, double or triple Depends on the type of bond e.g. C to N or C to O (refer to chart on handout) Is measured in kJ per mol of bonds (kJ/mol) Calculating the difference between the total energy absorbed by breaking bonds and the total energy released when new bonds are formed determines whether the reaction is endo or exo. Reaction Energy = Energy absorbed by the Reactant bonds - Energy released by the Product bonds (ENTHALPY) Reaction Energy = BER - BEP ENTHALPY = BER - BEP Problem: How much energy is involved when 1 mole of methane, CH4, is burned? 1) Write the balanced chemical equation. _____________________________________________________________________________ 2) Sketch the Energy graph: 3) Sketch the molecules of the reactants and products using Lewis dot diagrams: Reactants Products 4) Calculate the total energy absorbed by breaking all the bonds of the reactants. 5) Calculate the total energy released by forming all the bonds of the products. 6) Subtract 5) from 4) = Reactant Energy 7) Write an English sentence for what happens when 1 mole of methane is burned.