Involvement of Non-VCU Institutions in VCU Human Subjects
advertisement

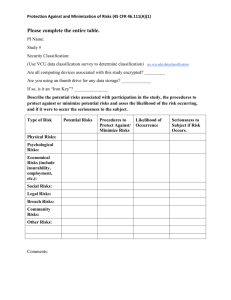
Involvement of Non-VCU Institutions in VCU Human Subjects Research Prepared for the 5th Thursday for Human Subjects Protection Program 3/07 1 What is a Non-VCU Institution? An institution (or an employee or agent of the institution) that is not under the authority of VCU or VCU Health Systems and is located within the United States or a United States territory. Examples include clinics, schools, other universities, consulting firms, etc. 2 VCU Responsibilities Review each institution’s role in the human subjects research, adequacy of the facility (in order to ensure human subject safety in the case of an unanticipated emergency), responsibilities of its agents/employees, and oversight that our PI will provide in order to ensure adequate and ongoing protection of the human subjects. 3 VCU Responsibilities (Continued) Ensure that all collaborating institutions “engaged” in non-exempt VCU human subjects research operate under an appropriate OHRP-approved or other federally-approved assurance for the protection of human subjects when the research: (a) is funded by a direct federal grant, contract, or cooperative agreement awarded to VCU or (b) involves serving as the coordinating center for federally-conducted or –supported research. 4 When is a Collaborating Institution Engaged? In general, the collaborating institution becomes engaged whenever: (a) the institution’s employees or agents intervene or interact with human subjects for purposes of non-exempt federally-conducted or supported research; or (b) the institution’s employees or agents obtain individually identifiable private information about human subjects for purposes of non-exempt federally-conducted or supported research. 5 Examples of Engagement Examples of when an institution is considered “engaged” in human subjects research can be found at http://www.hhs.gov/ohrp/humansubjects/assurance/engage.htm Institutions whose employees intervene with living individuals by performing invasive or noninvasive procedures for research purposes (e.g., drawing blood; dispensing drugs; administering other treatments. 6 Examples of Engagement (Continued) Institutions whose employees or agents interact with living individuals for research purposes (e.g., conducting research interviews; obtaining informed consent). Institutions whose employees obtain, receive or possess private information that is individually identifiable (either directly or indirectly through coding systems) for research purposes (e.g., obtaining private information for data analysis purposes). 7 What is a Federalwide Assurance? A Federalwide Assurance is "documentation of an institutional commitment to comply with the Department of Health and Human Services regulations for the protection of human subjects". The Terms of the Federalwide Assurance can be found at http://www.hhs.gov/ohrp/humansubjects/assurance/filasurt.htm. 8 Steps to Take if You Plan to Involve Non-VCU Institutions Plan for their involvement as you are designing your study and submitting grant applications. Review the VCU IRB Policy on Involvement of NonVCU Institutions in VCU Human Subjects Research http://www.research.vcu.edu/irb/wpp/flash/wpp_guide.htm#XVII-6.htm 9 Steps to Take if You Plan to Involve Non-VCU Institutions (Continued) Determine whether the institutions will be “engaged” in the research project. If they are not engaged, or the project comprises exempt review status, or the project does not involve a direct federal award or application for such: Be prepared to thoroughly describe their involvement in your protocol. 10 Steps to Take if You Plan to Involve Non-VCU Institutions (Continued) If they are engaged and the research project is non-exempt and involves a direct federal award or application for such: Determine whether the institutions have a Federalwide Assurance (FWA). • If they have an FWA, you will need to include their Assurance number in Section XII: Involvement of Non-VCU Institutions in VCU Human Subjects Research of your protocol/research synopsis. 11 Steps to Take if You Plan to Involve Non-VCU Institutions (Continued) • If they do not have an FWA, discuss with them the need to obtain one if they intend to be involved in your research project. 12 Steps to File a Federalwide Assurance Review Steps to File a Domestic Federalwide Assurance at http://www.hhs.gov/ohrp/assurances/assurances_in dex.html The collaborating institutions will need to identify an IRB of record for their institution when they file. 13 Steps to File a Federalwide Assurance If these institutions do not routinely engage in human subjects research, they are likely to prefer relying on the VCU Institutional Review Board (IRB) when engaged in VCU human subjects research. VCU would need to agree to the arrangement and such an arrangement must be documented in writing between the two institutions. If VCU agrees to the arrangement, the collaborating institution will list the VCU IRB registration number for each designated IRB and the name of the IRB(s) on its FWA application. 14